Article contents
Prevention of Candida biofilm formation over polystyrene by plasma polymerization technique
Published online by Cambridge University Press: 15 October 2020
Abstract
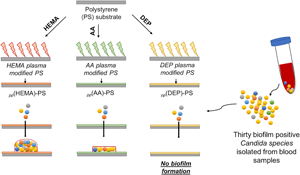
This work investigates the antifungal effect of plasma polymer films produced by low-pressure RF-generated plasma system using acrylic acid, 2–hydroxyethyl methacrylate, and diethyl phosphite (DEP). Unmodified and plasma-modified polystyrene (PS) microplate wells were tested by 30 biofilm-positive Candida spp. isolated from blood samples and two control strains using a quantitative plaque assay method. Regardless of the precursors and plasma parameters, biofilm formation was inhibited for all plasma-modified microplate wells. The most significant anti-biofilm effect was observed on PS modified by DEP at 90 W plasma power with the inhibition of all Candida species’ biofilm formation.
- Type
- Research Letters
- Information
- Copyright
- Copyright © The Author(s), 2020, published on behalf of Materials Research Society by Cambridge University Press
References
A correction has been issued for this article:
- 5
- Cited by
Linked content
Please note a has been issued for this article.



