Recently, photoelectrochemical (PEC) water splitting using semiconductor photoanode has received great attention due production of hydrogen through clean energy. The alpha hematite (α Fe2O3) is one of the candidate amongst photoanodic materials, which is chemically stable, abundant in nature with a band gap of 2.0-2. 2eV allowing to be harvesting in the visible light. However, it has also drawn back due to high recombination rate of electron–hole pair revealing the low concentration of charges and lower device performance. In common with α-Fe2O3, the titanium dioxide (TiO2) has been known as one of the most explored photoanode electrode material due to its physical and chemical stability in aqueous and non-toxicity. However, TiO2 has large bandgap (3.0-3.2 eV) that results in absorbing UV light and very small part of visible region. Incorporation of TiO2 in α-Fe2O3 could achieve better efficiencies as photoanode materials by enhancing the electric conductivity, limited hole diffusion length, and both materials can absorb light in both UV and visible spectrum range. However, the photoanodic properties of α-Fe2O3 with different concentrations of TiO2 are mostly unknown. Under this work, α-Fe2O3-TiO2 nanomaterial was synthesized using a hydrothermal method. The α-Fe2O3-TiO2 nanomaterials containing different weight percentage (2.5, 5, 16, 25, and 50) of TiO2 to α-Fe2O3 were characterized using SEM, XRD, UV-Vis, FTIR and Raman techniques, respectively. The electrochemical properties of α-Fe2O3-TiO2 nanomaterials were investigated by cyclic voltammetry and chronoamperometry techniques, respectively.
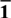 ${{\rm{\bar{1}}}}$2} surfaces of hematite for water splitting
${{\rm{\bar{1}}}}$2} surfaces of hematite for water splitting