Introduction
In recent years, coastal Rapid Assessment Surveys (RAS) have proven valuable to assess the current distribution of non-native species, to detect new introductions, and to assess changes in our knowledge of the distribution of native species. In the New England-New York region, these surveys began in 2000 focusing on sampling biofouling communities on floating docks in marinas (Pederson et al., Reference Pederson, Carlton, Bastidas, David, Grady, Green-Gavrielidis, Hobbs, Kennedy, Knack, McCuller, O'Brien, Osborne, Pankey and Trott2021). Seven surveys have been held, generally every two to three years, between 2000 and 2019, largely including sites from Maine to Connecticut (with New York being sampled as well in 2003 and 2019) (Pederson et al., Reference Pederson, Bullock, Carlton, Dijkstra, Dobroski, Dyrynda, Fisher, Harris, Hobbs, Lambert, Lazo-Wasem, Mathieson, Miglietta, Smith, Smith and Tyrrell2005, Reference Pederson, Carlton, Bastidas, David, Grady, Green-Gavrielidis, Hobbs, Kennedy, Knack, McCuller, O'Brien, Osborne, Pankey and Trott2021; Mathieson et al., Reference Mathieson, Pederson, Neefus, Dawes and Bray2008; McIntyre et al., Reference McIntyre, Pappal, Bryant, Carlton, Cute, Dijkstra, Erickson, Garner, Gittenberger, Grady, Haram, Harris, Hobbs, Lambert, Lambert, Lambert, Marques, Mathieson, McCuller, Mickiewicz, Pederson, Rock-Blake, Smith, Sorte, Stefaniak and Wagstaff2013; Wells et al., Reference Wells, Pappal, Cao, Carlton, Currimjee, Dijkstra, Edquist, Gittenberger, Mathieson, McCuller, Osborne, Pederson, Ros, Smith, Stefaniak and Stevens2014; Kennedy et al., Reference Kennedy, Pappal, Carlton, David, Dijkstra, Duffey, Gibson, Grady, Green-Gavrielidis, Harris, Hobbs, Mauk, McCuller, Neefus, O'Brien, Osborne, Pederson, Robidoux, Tyler and Van Volkom2020). The success of these surveys has resulted in similar surveys being deployed in European waters (Arenas et al., Reference Arenas, Bishop, Carlton, Dyrynda, Farnham, Gonzalez, Jacobs, Lambert, Lambert, Nielsen, Pederson, Porter, Ward and Wood2006; Ashton et al., Reference Ashton, Boos, Shucksmith and Cook2006; Nall et al., Reference Nall, Guerin and Cook2015; O'Shaughnessy et al., Reference O'Shaughnessy, Lyons, Ashelby, Counihan, Pears, Taylor, Davies and Stebbing2023) and elsewhere. This type of repeated targeted sampling across an extensive geographic region coupled with the participation of taxon-specific experts has proven to be a successful method for biomonitoring.
The polychaetous annelids (Polychaeta) are among the most diverse marine organisms and are integral components of marine ecosystems, often dominating benthic communities in terms of abundance and species richness (Rouse et al., Reference Rouse, Pleijel and Tilic2022). In addition, many commensal species have been implicated as introduced pests to commercially reared shellfish, thereby posing a threat to the global aquaculture trade (Simon and Sato-Okoshi, Reference Simon and Sato-Okoshi2015; Davinack et al., Reference Davinack, Strong and Brennessel2024; Martinelli et al., Reference Martinelli, Considine, Casendino, Tarpey, Jimenez-Hidalgo, Padilla-Gamino, King, Hauser, Rumrill and Wood2024). However, identifying polychaetes poses significant challenges, even for expert polychaetologists. These difficulties stem in part from the increasing reports of cryptic species complexes and the constantly evolving taxonomy of these organisms, which can lead to uncertainty with regard to their status as native, introduced or cryptogenic species (Nygren, Reference Nygren2014; Darling and Carlton, Reference Darling and Carlton2018; Hutchings and Kupriyanova, Reference Hutchings and Kupriyanova2018). A recent review by Darling and Carlton (Reference Darling and Carlton2018), proposed the category of ‘pseudocosmopolitan’ for species whose perceived cosmopolitan status might be a misleading result of overly conservative taxonomic practices. This can be applied as a sub-category for cryptogenic species that are members of known species complexes that have yet to be resolved taxonomically and biogeographically. In one of the most recent rapid assessment surveys in New England, David and Krick (Reference David and Krick2019) incorporated DNA barcoding for the first time into the cataloging process that resulted in increased resolution of species diversity. These challenges underscore the importance of a comprehensive and nuanced approach to understanding polychaete diversity and distribution.
In this study, we present the first annotated checklist of polychaetes collected from floating dock communities in the New England – New York regions, which for centuries have had a long maritime history. Consequentially this has resulted in the potential historical movement of marine species from other biogeographic regions. While previously published technical and peer-reviewed articles have summarized the findings of these surveys, granular details of the polychaete records were not previously assessed comprehensively and quantitatively. In addition, with the recent explosion in DNA barcoding, some previous identifications are no longer valid due to recent synonymizations. The polychaete checklist presented here is the product of almost 20 years of rapid biodiversity assessment surveys, reflecting a substantial effort in understanding marine biodiversity in New England. In addition to cataloguing these polychaetes, we delve into their biogeographic status, categorizing each species as native, introduced, or cryptogenic – the latter denoting species that cannot be definitively classified as either native or introduced (Carlton, Reference Carlton1996). Finally, we statistically analyse polychaete composition across different marinas and across different years. Altogether, this information is pivotal for understanding the changing dynamics of marine communities.
Materials and Methods
Description of study region and RAS protocol
Between 2000 and 2019, seven rapid assessment surveys were conducted at 56 marinas, largely along the New England coast (Figure 1). The names and GPS coordinates of the specific sites where at least one polychaete species was found (40 sites in total) are found in Table 1. These surveys were carried out in 2000, 2003, 2007, 2010, 2013, 2018 and 2019, with the 2019 survey specifically targeting Rhode Island and Connecticut (southern New England) and marinas in New York City (Brooklyn and Staten Island). Many of the marinas were sampled repeatedly across multiple years using the same methodology. The typical rapid assessment protocol involved sampling each marina for one hour by a team of taxonomists and research assistants. Over the past two decades, the number of workers has varied between 12–15 individuals with the sampling period occurring during summer months (either July or August). Sampling involved the scraping of material off docks and pilings and the amount can differ depending on the individual sampling. Team members sample broadly across any given marina, attempting to capture both protected sites in inner marina areas as well as more exposed outer sites. Biological material examined from these communities included mostly marine invertebrates and algae that were associated with subtidal structures such as submerged docks, pilings, ropes and buoys. While some organisms were identified on site, some groups such as polychaetes were stored alive in coolers with ice and transported to nearby university laboratories for further identification on the same day.
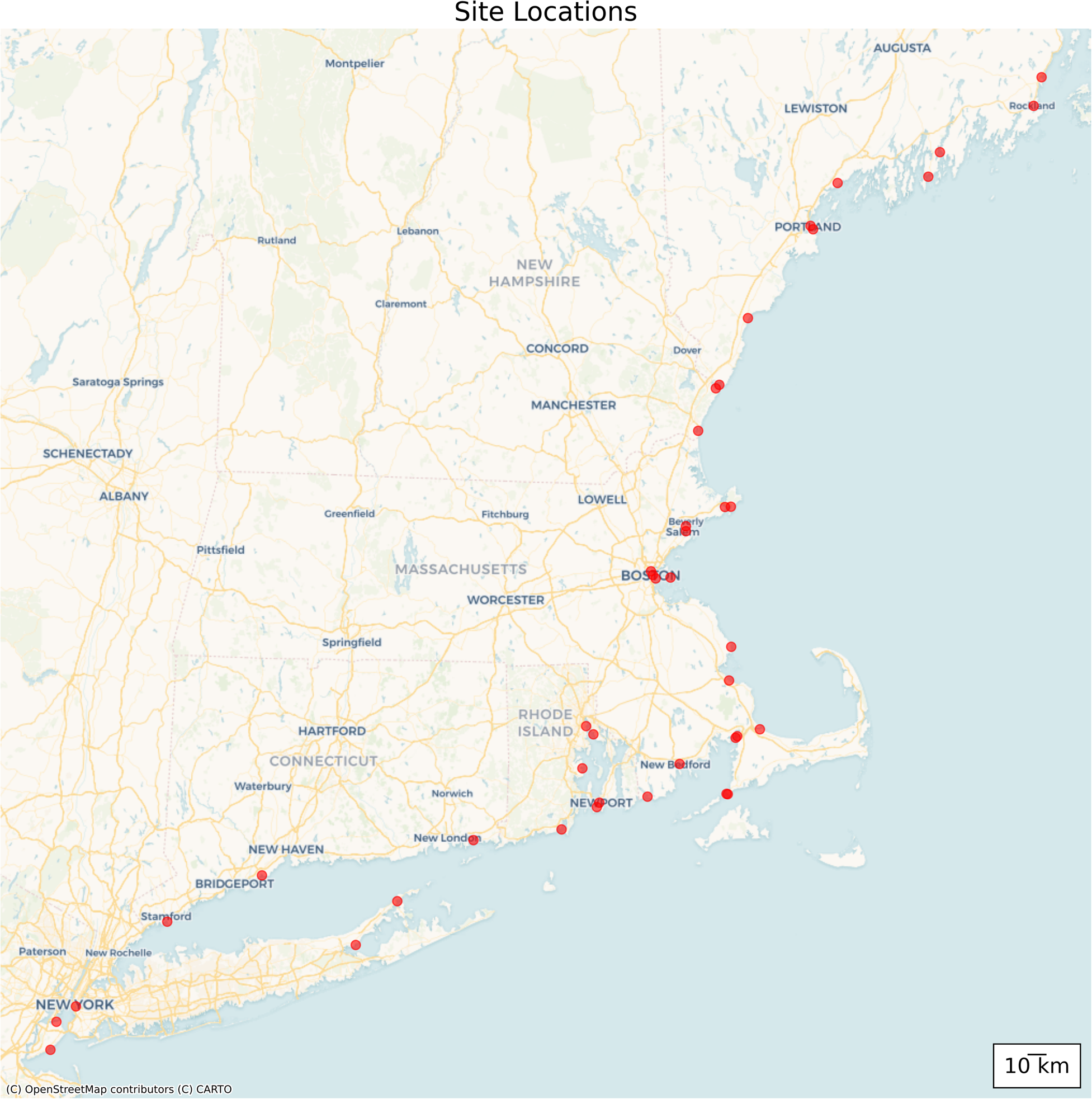
Figure 1. Sampling localities for polychaetes collected during rapid assessment surveys carried out between 2000 and 2019.
Table 1. Floating dock marinas in New England and adjacent New York sites that were successfully sampled for polychaetes at rapid assessment surveys from 2000–2019
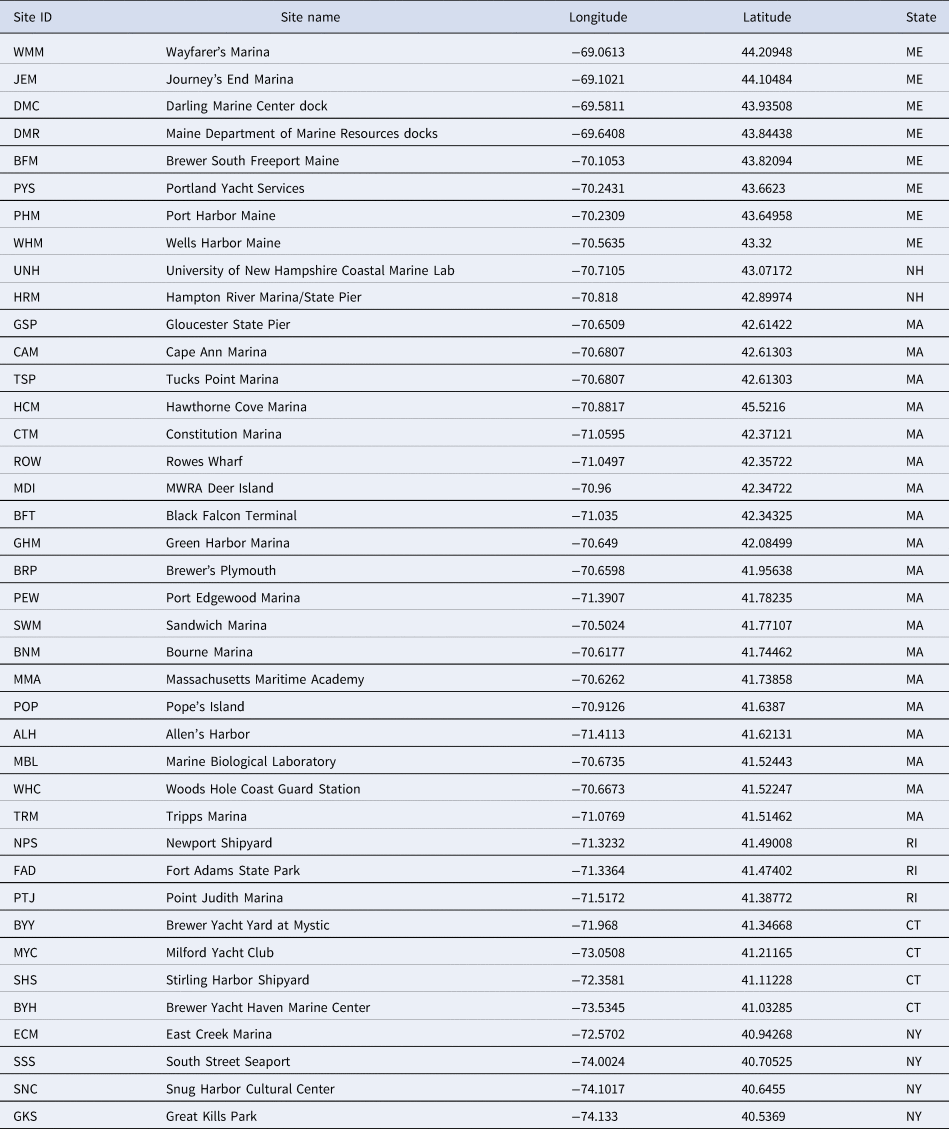
ME, Maine; NH, New Hampshire; MA, Massachusetts; RI, Rhode Island; CT, Connecticut; NY, New York.
Identification of polychaetes was carried out using regional taxonomic keys to identify specimens to the species level. It should be noted that different polychaetologists were consulted on different surveys. For specimens that could not be identified to species level, these were stored in 99% ethanol for genetic barcoding, the results of which are available for the 2018 survey in David and Krick (Reference David and Krick2019). Distributional data collected were binary (presence/absence) and did not include relative abundance. In cases where multiple individuals of the same species were confirmed and recorded, some specimens were stored in 99% ethanol and archived as part of a community voucher at the Museum of Comparative Zoology at Harvard University.
Data mining
We first reviewed the original species inventory spreadsheets that were compiled for each survey since 2000, in addition to reviewing summaries of surveys that have been published in the grey literature by the Massachusetts Office of Coastal Zone Management (McIntyre et al., Reference McIntyre, Pappal, Bryant, Carlton, Cute, Dijkstra, Erickson, Garner, Gittenberger, Grady, Haram, Harris, Hobbs, Lambert, Lambert, Lambert, Marques, Mathieson, McCuller, Mickiewicz, Pederson, Rock-Blake, Smith, Sorte, Stefaniak and Wagstaff2013; Wells et al., Reference Wells, Pappal, Cao, Carlton, Currimjee, Dijkstra, Edquist, Gittenberger, Mathieson, McCuller, Osborne, Pederson, Ros, Smith, Stefaniak and Stevens2014; Kennedy et al., Reference Kennedy, Pappal, Carlton, David, Dijkstra, Duffey, Gibson, Grady, Green-Gavrielidis, Harris, Hobbs, Mauk, McCuller, Neefus, O'Brien, Osborne, Pederson, Robidoux, Tyler and Van Volkom2020). Historical records and species statuses were updated based on recent taxonomic revisions, DNA barcoding studies, updated biogeographical analyses and name/rank changes obtained from the World Register of Marine Species (WoRMS Editorial Board, 2024) database. Furthermore, specimen records from the Smithsonian Institution National Museum of Natural History invertebrate collections online database were examined. Based upon the literature cited for each species and upon our own biogeographic interpretations, we classified species as either Native (N), Introduced (I), Cryptogenic (C) or Unresolved cosmopolitan (U) (the latter referring to species that have been traditionally regarded as cosmopolitan in the literature but for which there are no studies to support this hypothesis). Finally, for each species we investigated whether common DNA barcodes were available for the species in the GenBank database, Barcode of Life Database (BoLD) and the DNA Databank of Japan (DDBJ). We tentatively assume that these deposited sequences were, in general, correctly identified, although these databases do not require indicating the expert taxonomists involved.
Statistical analyses
To assess the spatial and temporal differences in polychaete community composition across sites and across sampling years, we performed two separate Permutational Multivariate Analysis of Variance (PERMANOVA) tests using the scikit-bio library (https://scikit.bio.org) in Python ver. 3.0. The tests compared the Jaccard dissimilarity indices between samples from different years while permutations (n = 999) were used to assess the significance of the observed difference. To assess temporal variation in species composition, we also constructed a NMDS (Non-metric Multidimensional Scaling) plot to identify any distinct clustering of samples by year.
Results
Polychaete diversity analysis
From 2000–2019, 46 polychaete species were found associated with floating dock communities from 40 marinas. Of these, approximately 61% were cryptogenic, 35% were native and less than 4% were either introduced or an unresolved cosmopolitan (one species for each of those two categories). These species represent 16 of the 85 accepted polychaete families, with the most well represented being the Spionidae (14%) followed by Terebellidae (12%) (Figure 2). A PERMANOVA test found that there was no significant difference in polychaete composition across marinas (pseudo-F = 1.11, P = 0.08). While we did detect a significant difference in species composition across years sampled (pseudo-F = 6.91, P = 0.001) this was not reflected in the NMDS plot, which did not exhibit distinct clustering of samples (Figure 3).
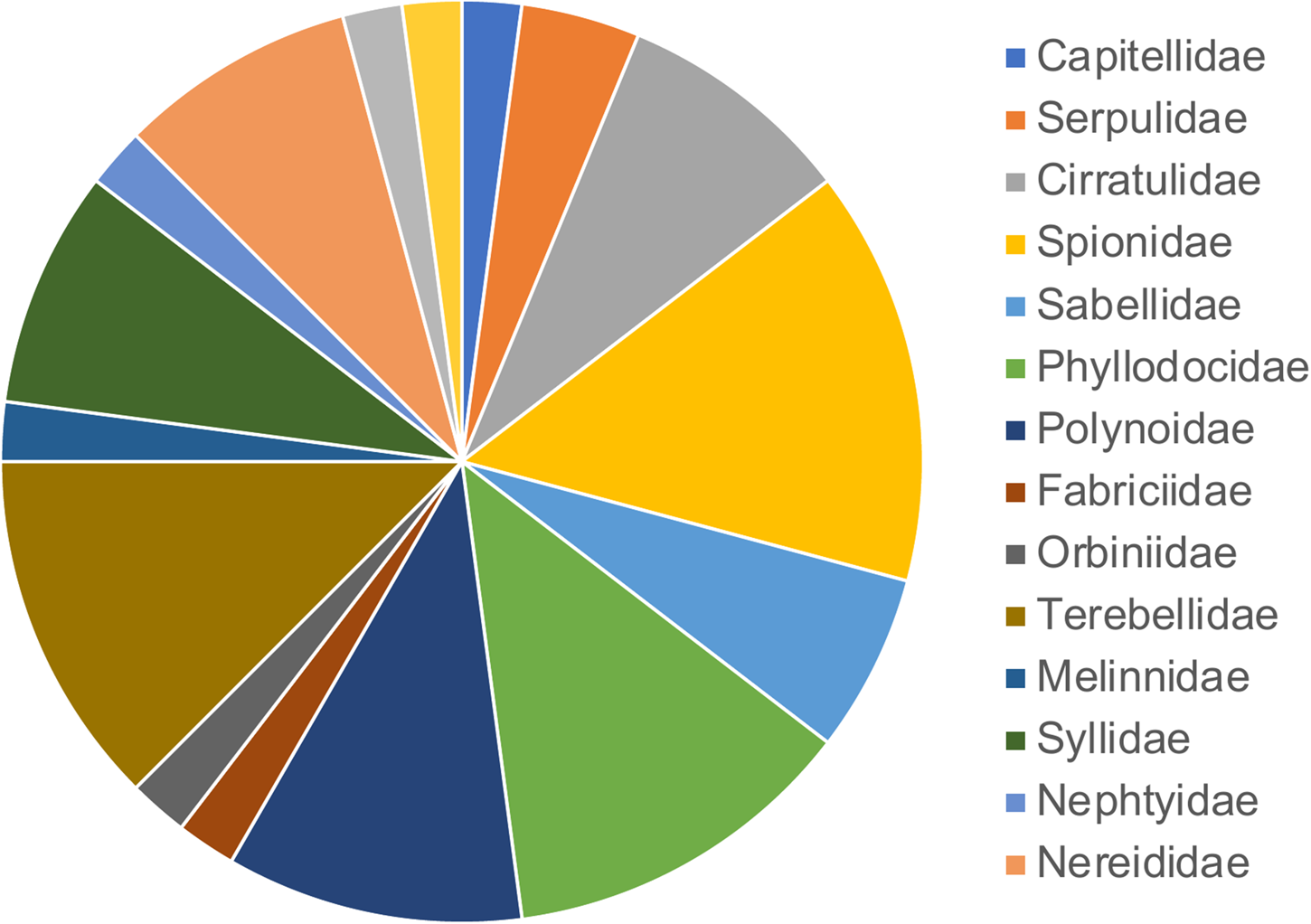
Figure 2. Proportion of polychaete families represented in the New England Rapid Assessment Surveys (2000–2019).
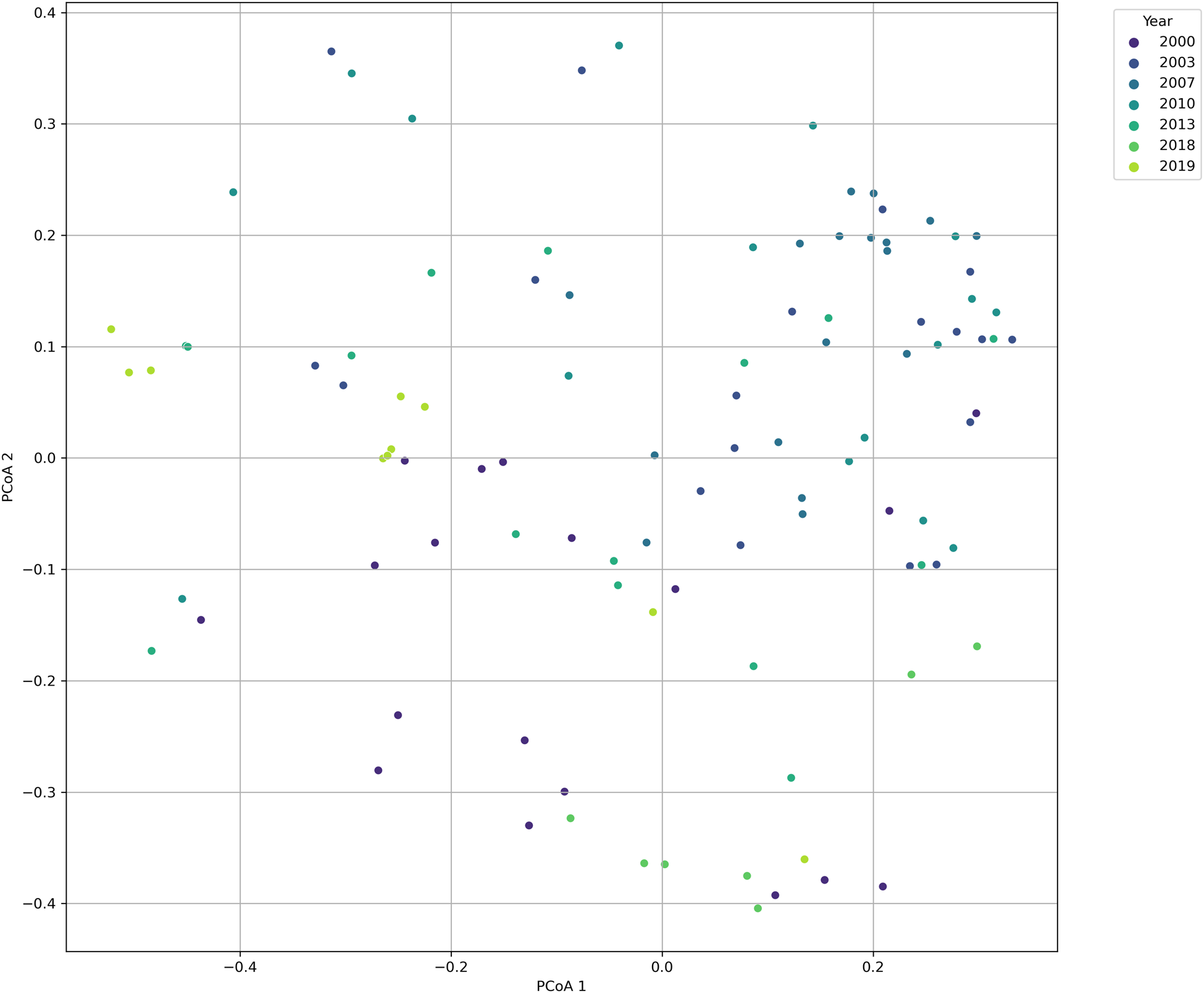
Figure 3. Non-metric multidimensional scaling (NMDS) plot of polychaete species composition based on Jaccard dissimilarity indices. Each point represents the species composition of a sample, with the colour indicating the year of collection. The proximity of points suggests the similarity in species composition, with closer points indicating more similar communities. The axes, PCoA 1 and PCoA 2, represent the two dimensions explaining the most variation in the dataset.
Annotated checklist of polychaeta associated with floating dock communities
FAMILY CAPITELLIDAE
Capitella teleta Blake et al., Reference Blake, Grassle and Eckelbarger2009
Synonymized names: Capitella sp. 1 [informal, 1976]
Status: Cryptogenic
Type Locality: New England region, USA
Barcodes available: Yes
Notes: Capitella teleta is the species name given by Blake et al. (Reference Blake, Grassle and Eckelbarger2009) to replace Capitella sp. 1, an opportunistic capitellid worm that had been cultured in laboratory experiments for decades. The species appears to have a wide distribution, with genetic confirmation of its presence in the Pacific and Atlantic – likely the result of transoceanic introductions (Tomioka et al., Reference Tomioka, Kondoh, Sato-Okoshi, Ito, Kakui and Kajihara2016). Considering the long history of studies on Capitella sp. 1 along with the possibility of other potential cryptic species, pinpointing an exact geographic origin, i.e., the native range for the species is not possible to determine based on the current data.
FAMILY CIRRATULIDAE
Cirriformia grandis (Verrill, Reference Verrill1873)
Synonymized names: Cirratulus grandis Verrill, Reference Verrill1873, Cirratulus tenuis Verrill, Reference Verrill1873
Status: Native
Type Locality: ‘New Haven to Vineyard Sound’, southern New England, USA
Barcodes available: No
Notes: Cirriformia grandis has been reported exclusively from the western Atlantic, with a distribution from the Gulf of Maine to the Gulf of Mexico (Kinner and Maurer, Reference Kinner and Maurer1971).
Ctenodrilus serratus (Schmidt, Reference Schmidt1857)
Synonymized names: Ctenodrilus paradalis Claparède, Reference Claparède1863, Parthenope cirrata [auct. lapsus, cirrata for serrata], Parthenope serrata Schmidt, Reference Schmidt1857
Status: Introduced
Type Locality: Mediterranean Sea
Barcodes available: Yes
Notes: While Ctenodrilus serratus is reported to have a cosmopolitan distribution (Bellan, Reference Bellan2001; Glasby et al., Reference Glasby, Read, Lee, Blakemore, Fraser, Pinder, Erséus, Moser, Burreson, Govedich, Davies, Dawson and Gordon2009), it is likely a global species complex interlaced with introduced populations (for example, Weidhase et al., Reference Weidhase, Bleidorn and Simon2016). Westheide et al. (Reference Westheide, Haß-Cordes, Krabusch and Müller2003) demonstrated that C. serratus on both sides of the North Atlantic were genetically identical. Given the possibility of early dispersal in ships' sand ballast (Westheide et al., Reference Westheide, Haß-Cordes, Krabusch and Müller2003) along with its highly patchy distribution in New England, we regard it as an introduced species. The species was found at only two sites in different years, once in 2007 at the Hawthorne Cove Marina in Salem, Massachusetts, and three years later in 2010 at Port Edgewood Marina in Cranston, Rhode Island. While found in 2010, the record escaped reporting in McIntyre et al. (Reference McIntyre, Pappal, Bryant, Carlton, Cute, Dijkstra, Erickson, Garner, Gittenberger, Grady, Haram, Harris, Hobbs, Lambert, Lambert, Lambert, Marques, Mathieson, McCuller, Mickiewicz, Pederson, Rock-Blake, Smith, Sorte, Stefaniak and Wagstaff2013); there was no published report of the 2007 RAS, and we are thus first recording these records here. While Hansen et al. (Reference Hansen, Mahony, Berry, Benyi, Corbin, Pratt, Di Toro and Abel1996) reported it from Long Island Sound, we find few published records of Ctenodrilus serratus for New England.
Dodecaceria concharum Örsted, Reference Örsted1843
Synonymized names: Dodecaceria caulleryi Dehorne, 1933, Heterocirrus graviera McIntosh, 1911, Nereis sextentaculata Delle Chiaje, 1828, Terebella ostreae Dalyell, Reference Dalyell1853, Zeppelina mediopigmentata Gillandt, 1979
Status: Cryptogenic
Type Locality: Denmark
Barcodes available: Yes
Notes: Similar to the closely related Ctenodrilus serratus, D. concharum has had a relatively problematic taxonomic history compounded by a lack of genetic data (Gibson, Reference Gibson2015).
Tharyx acutus Webster and Benedict, Reference Webster and Benedict1887
Synonymized names: none
Status: Native
Type Locality: Maine
Barcodes available: Yes
Notes: Based on the distributional reports, descriptions, and revisions of the genus Tharyx by Hilbig and Blake (Reference Hilbig and Blake2000), Blake (Reference Blake2015), and Blake and Göransson (Reference Blake and Göransson2015), T. acutus appears to be restricted to the northeastern United States.
FAMILY FABRICIIDAE
Fabricia stellaris (Müller, Reference Müller1774)
Synonymized names: Amphicora fabricia (Müller, Reference Müller1776), Amphicora fabricii [auct. misspelling], Amphicora sabella Ehrenberg, 1836, Fabricia affinis Leuckart, 1849, Fabricia amphicora Quatrefages, 1866, Fabricia leidyi Verrill, Reference Verrill1873, Fabricia sabella (Ehrenberg, 1836), Novafabricia bilobata Martin & Giangrande, 1991, Othonia fabricii (Johnston, 1835), Tubularia fabricia Müller, Reference Müller1776, Tubularia stellaris Müller, Reference Müller1774
Status: Cryptogenic
Type Locality: Denmark
Notes: The majority of reports are from the Arctic and North Atlantic region (Davoult et al., Reference Davoult, Dewarumez, Luczak and Migné1999; Christie et al., Reference Christie, Jørgensen, Norderhaug and Waage-Nielsen2003) with additional reports from the Caribbean Sea and the Mediterranean Sea (Davoult et al., Reference Davoult, Dewarumez, Luczak and Migné1999; Miloslavich et al., Reference Miloslavich, Diaz, Klein, Alvarado, Diaz, Gobin, Escobar-Briones, Cruz-Motta, Weil, Cortes, Bastidas, Robertson, Zapata, Martin, Castillo, Kazandjian and Ortiz2010). There are currently no biogeographic or population genetic studies available for this species.
FAMILY HESIONIDAE
Oxydromus obscurus (Verrill, Reference Verrill1873)
Synonymized names: Ophiodromus obscurus (Verrill, Reference Verrill1873), Podarke obscura Verrill, Reference Verrill1873
Status: Native
Type Locality: Massachusetts, USA
Barcodes available: Yes
Notes: The distribution of Oxydromus obscurus is largely restricted to the northeastern and northwestern Atlantic and Gulf of Mexico (Bellan, Reference Bellan2001; Felder and Camp, Reference Felder and Camp2009). Lana (Reference Lana1984) reported the species from Paraná in Brazil, while Fauchald (Reference Fauchald1977) recorded the species from Panama, although a review by Rizzo and Salazar-Vallejo (Reference Rizzo and Salazar-Vallejo2014) considers these identifications questionable. The species was also recently reported from Egypt (Abdelnaby, Reference Abdelnaby2019), although this report is highly dubious because of an inadequate description.
FAMILY MELINNIDAE
Melinna cristata (Sars, Reference Sars1851)
Synonymized names: Sabellides cristata Sars, Reference Sars1851
Status: Cryptogenic
Type Locality: Norway
Barcodes available: Yes
Notes: Melinna cristata is found throughout the Atlantic Ocean, including north, east, mid, and south Atlantic (Gunton et al., Reference Gunton, Zhang, Kupriyanova and Hutchings2023). While some barcodes do exist for the species, a large-scale phylogeographic study is needed to better understand the status of the species in New England.
FAMILY NEREIDIDAE
Alitta virens (Sars, Reference Sars1835)
Synonymized names: Neanthes virens (Sars, Reference Sars1835), Nereis (Alitta) virens Sars, Reference Sars1835, Nereis (Neanthes) paucidentata Treadwell, 1939, Nereis (Neanthes) varia Treadwell, 1941, Nereis (Neanthes) virens Sars, Reference Sars1835, Nereis virens Sars, Reference Sars1835, Nereis yankiana Quatrefages, 1866
Status: Cryptogenic (Pseudocosmopolitan)
Type Locality: Norway
Barcodes available: Yes
Notes: David and Krick (Reference David and Krick2019) classified A. virens from New England as pseudocosmopolitan, owing to the fact that it is part of a species complex and morphologically indistinguishable from its sibling species, A. brandti and A. grandis (Villalobos-Guerrero and Bakken, Reference Villalobos-Guerrero and Bakken2018).
Hediste diversicolor (Müller, Reference Müller1776)
Synonymized names: Neanthes diversicolor (Müller, Reference Müller1776), Nereis (Hediste) diversicolor Müller, Reference Müller1776, Nereis (Nereis) diversicolor Müller, Reference Müller1776, Nereis brevimanus Johnston, 1840, Nereis depressa Frey & Leuckart, 1847, Nereis diversicolor Müller, Reference Müller1776, Nereis sarsii Rathke, 1843, Nereis versicolor [misspelling for diversicolor], Nereis viridis Johnston, 1840
Status: Cryptogenic
Type Locality: North Sea
Barcodes available: Yes
Notes: Hediste diversicolor is a well-known environmental indicator species that is increasingly used to detect emerging contaminants (Catalano et al., Reference Catalano, Moltedo, Martuccio, Gastaldi, Virno-Lamberti, Lauria and Ausili2012; Moltedo et al., Reference Moltedo, Martuccio, Catalano, Gastaldi, Maggi, Virno-Lamberti and Cicero2019; Silva et al., Reference Silva, Pires, Almeida and Oliveira2020). The worm is one of the more ubiquitous polychaetes found throughout marinas in the New England region, usually buried within mudpacks on fouling communities (David and Krick, Reference David and Krick2019). It has an amphi-Atlantic distribution, with other reports outside of this area being dubious due to the species' problematic taxonomic history (Scaps, Reference Scaps2002). A phylogeographic study by Virgilio et al. (Reference Virgilio, Fauvelot, Costantini, Abbiati and Backeljau2009) recovered three cryptic lineages for H. diversicolor, indicating that multiple species may be present in different geographical regions. Unfortunately, the western Atlantic was not included in that study but is an area ripe for future investigation.
Nereis pelagica Linnaeus, Reference Linnaeus1758
Synonymized names: Heteronereis arctica Örsted, Reference Örsted1843, Heteronereis assimilis Örsted, Reference Örsted1843, Heteronereis grandifolia (Rathke, 1843), Heteronereis migratoria Quatrefages, 1866, Nereilepas fusca Örsted, Reference Örsted1843, Nereis (Lycoris) pelagica Linnaeus, Reference Linnaeus1758, Nereis (Nereis) pelagica Linnaeus, Reference Linnaeus1758, Nereis bowerbanckii Quatrefages, 1866, Nereis delagica [auct. misspelling for pelagica], Nereis deneticulata Stimpson, 1853, Nereis ferruginea Gunnerus, 1770, Nereis fimbriata Müller, Reference Müller1776, Nereis fulgens Dalyell, Reference Dalyell1853, Nereis grandifolia Rathke, 1843, Nereis renalis Johnston, 1840, Nereis reynaudi Quatrefages, 1866, Nereis subulicola Leach in Johnston, 1865, Nereis verrucosa Müller, Reference Müller1776
Status: Cryptogenic
Type Locality: Western Europe
Barcodes available: Yes
Notes: Bakken and Wilson (Reference Bakken and Wilson2005) in their phylogeny of the Nereididae reported Nereis pelagica as a cosmopolitan species. However, in light of the large number of cryptic complexes currently being uncovered in this group, a species complex may be involved.
Platynereis dumerilii (Audouin & Milne Edwards, 1833)
Synonymized names: Eunereis africana Treadwell, 1943, Heteronereis fucicola Örsted, Reference Örsted1843, Heteronereis maculata Bobretzky, 1868, Heteronereis malmgreni Claparède, 1868, Iphinereis fucicola (Örsted, Reference Örsted1843), Leontis dumerili (Audouin & Milne Edwards, 1833), Leptonereis maculata Treadwell, 1928, Mastigonereis quadridentata Schmarda, Reference Schmarda1861, Mastigonereis striata Schmarda, Reference Schmarda1861, Nereilepas variabilis Örsted, Reference Örsted1843, Nereis (Platynereis) dumerilii Audouin & Milne Edwards, 1833, Nereis (Platynereis) dumerilii striata (Schmarda, Reference Schmarda1861), Nereis (Platynereis) striata (Schmarda, Reference Schmarda1861), Nereis alacris Verrill, 1879, Nereis antillensis McIntosh, 1885, Nereis dumerilii Audouin & Milne Edwards, 1833, Nereis glasiovi Hansen, 1882, Nereis peritonealis Claparède, 1868, Nereis taurica Grube, 1850, Nereis zostericola Örsted, Reference Örsted1843, Platynereis dumerili [auct. misspelling], Platynereis jucunda Kinberg, 1865, Platynereis striata (Schmarda, Reference Schmarda1861), Uncinereis lutea Treadwell, 1928, Uncinereis trimaculosa Treadwell, 1940
Status: Cryptogenic (Pseudocosmopolitan)
Type Locality: La Rochelle, France
Barcodes available: Yes
Notes: Platynereis dumerilii is a well-known animal model used in a variety of molecular studies (Zantke et al., Reference Zantke, Bannister, Babu, Rajan, Raible and Tessmar-Raible2014). However, the species itself consists of at least 10 divergent lineages (Teixeira et al., Reference Teixeira, Langeneck, Vieira, Hernandez, Sampieri, Kasapidis, Mucciolo, Bakken, Ravara, Nygren and Costa2022), which challenges its former cosmopolitan designation, but also makes it extremely difficult to determine which areas, if any, it has been introduced to. For example, a study by Kara et al. (Reference Kara, Santos, Macdonald and Simon2020) used molecular data to show that P. dumerilii, despite being reported from South Africa in the past, is actually absent from the region. Instead, a new Platynereis species was discovered and described, and a second potential species remains unresolved.
FAMILY NEPHTYIDAE
Nephtys incisa Malmgren, Reference Malmgren1865
Synonymized names: Nephthys incisa Malmgren, Reference Malmgren1865, Nephthys incisa bilobata Heinen, 1911, Nephthys lawrencii McIntosh, 1900
Status: Cryptogenic
Type Locality: Sweden
Barcodes available: Yes
Notes: Nephtys incisa has a reported amphi-Atlantic distribution (Hayward and Ryland, Reference Hayward and Ryland1990; Dauvin et al., Reference Dauvin, Dewarumez and Gentil2003; Trott, Reference Trott2004). Currently, there are no morphogenetic or biogeographic studies on the species to determine its status in the New England region.
FAMILY ORBINIIDAE
Leitoscoloplos robustus (Verrill, Reference Verrill1873)
Synonymized names: Anthostoma robustum Verrill, Reference Verrill1873, Haploscoloplos robustus (Verrill, Reference Verrill1873), Haploscoloplos tortugaensis Monro, 1933, Haploscoloplos bustorus (Eisig, 1914), Scoloplos bustorus Eisig, 1914, Scoloplos robustus (Verrill, Reference Verrill1873), Scoloplos rufa Treadwell, 1941
Status: Native
Type Locality: New England region, USA
Barcodes available: Yes
Notes: The species is largely restricted to the northeastern United States (Trott, Reference Trott2004), with Miloslavich et al. (Reference Miloslavich, Diaz, Klein, Alvarado, Diaz, Gobin, Escobar-Briones, Cruz-Motta, Weil, Cortes, Bastidas, Robertson, Zapata, Martin, Castillo, Kazandjian and Ortiz2010) reporting additional records from the Caribbean Sea (see Table S5 in Miloslavich et al., Reference Miloslavich, Diaz, Klein, Alvarado, Diaz, Gobin, Escobar-Briones, Cruz-Motta, Weil, Cortes, Bastidas, Robertson, Zapata, Martin, Castillo, Kazandjian and Ortiz2010). Furthermore, Blake (Reference Blake2021) has also reported the distribution of this species from eastern Canada to Florida.
FAMILY PHYLLODOCIDAE
Eulalia viridis (Linnaeus, Reference Linnaeus1767)
Synonymized names: Eracia virens (Ehlers, 1864), Eulalia (Eumida) microceros Claparède, 1868, Eulalia annulate Verrill, Reference Verrill1873, Eulalia brevisetis Saint-Joseph, 1899, Eulalia virens Ehlers, 1864, Eumidia vivida Verrill, Reference Verrill1873, Nereis viridis Linnaeus, Reference Linnaeus1767, Phyllodoce gervillei Audouin & Milne Edwards, 1833, Pterocirrus microceros (Claparède, 1868)
Status: Cryptogenic (Pseudocosmopolitan)
Type Locality: Western Europe
Barcodes available: Yes
Notes: Eulalia viridis was discovered to be part of a cryptic complex by Bonse et al. (Reference Bonse, Schmidt, Eibye-Jacobsen and Westheide1996), who separated the species into E. viridis sensu-stricto, found in Sweden, Denmark, and Germany, and E. clavigera found in France and England. Using DNA barcoding, David and Krick (Reference David and Krick2019) identified the species found on the New England coast as belonging to E. viridis. However, a recent study by Teixeira et al. (Reference Teixeira, Vieira, Fenwick, Langeneck, Pleijel, Sampieri, Hernandez, Ravara, Costa and Nygren2023) using three different molecular markers recovered six distinct lineages within this complex. Because none of the individuals sequenced were from the western Atlantic, the clade identity and status of E. viridis in New England remain unknown at this time.
Phyllodoce arenae Webster, Reference Webster1879
Synonymized names: Anaitides arenae (Webster, Reference Webster1879)
Status: Native
Type Locality: New Jersey, USA
Barcodes available: No
Notes: Phyllodoce arenae has an exclusively western Atlantic distribution from Maine to the Gulf of Mexico (Pettibone, Reference Pettibone1963; Felder and Camp, Reference Felder and Camp2009), with one noted report from the southern Caribbean in Trinidad and Tobago (Gobin, Reference Gobin1990).
Phyllodoce groenlandica Örsted, Reference Örsted1842
Synonymized names: Anaitides groenlandica (Örsted, Reference Örsted1842), Phyllodoce (Anaitides) groenlandica Örsted, Reference Örsted1842, Phyllodoce arctica Hansen, 1882, Phyllodoce luetkeni Malmgren, 1867
Status: Native
Type Locality: Greenland
Barcodes available: Yes
Notes: Phyllodoce groenlandica is noted as an Arctic-boreal species (Nygren et al., Reference Nygren, Eklöf and Pleijel2009) ranging south to the Gulf of Maine (Verrill, Reference Verrill1874) and Long Island Sound (Verrill, Reference Verrill1875) and thence to North Carolina (Pettibone, Reference Pettibone1963). It should be noted that Magalhães et al. (Reference Magalhães, de Oliveira and Bailey-Brock2022) remarked on the morphological similarities of a large number of species of Phyllodoce, indicating that misidentification could be a possibility. Interestingly, David and Krick (Reference David and Krick2019), using DNA barcoding, were only able to recover Phyllodoce maculata and Phyllodoce mucosa from the 2018 rapid assessment survey. While P. groenlandica is reported from piling fouling communities (Pettibone, Reference Pettibone1963), it may be largely sublittoral in the studied region, where it has been reported primarily in dredged samples (Verrill, Reference Verrill1874, Reference Verrill1875). Alternatively, records of this species in earlier RAS samples (for example, in the year 2003) may represent a misidentification of P. maculata, P. mucosa, or both. A targeted revision of Phyllodoce in this region would help clarify the diversity of this genus in the Northwest Atlantic.
Phyllodoce maculata (Linnaeus, Reference Linnaeus1767)
Synonymized names: Anaitides maculata (Linnaeus, Reference Linnaeus1767), Nereis maculata Linnaeus, Reference Linnaeus1767, Phyllodoce (Anaitides) maculata (Linnaeus, Reference Linnaeus1767), Phyllodoce pulchella Malmgren, 1867, Phyllodoce rinki Malmgren, 1867, Phyllodoce teres Malmgren, Reference Malmgren1865
Status: Cryptogenic
Type Locality: Western Europe
Barcodes available: Yes
Notes: This species has been reported from the North Atlantic from Scandinavia to Iceland, Greenland, and Hudson Bay south to Rhode Island (Pettibone, Reference Pettibone1963) south to New Jersey (Kennish, Reference Kennish2001), as well as in the North Pacific from Japan, Alaska, and British Columbia (Pettibone, Reference Pettibone1963). Additional records are from West Africa (Pettibone, Reference Pettibone1963) and the Mediterranean (Bakalem et al., Reference Bakalem, Gillet, Pezy and Dauvin2020). A species complex may be involved. Due to confusion with Phyllodoce mucosa (Pleijel, Reference Pleijel1993), the full extent of the range of P. maculata is unknown (David and Krick, Reference David and Krick2019).
Phyllodoce mucosa Örsted, Reference Örsted1843
Synonymized names: Anaitides mucosa (Örsted, Reference Örsted1843), Phyllodoce (Anaitides) mucosa Örsted, Reference Örsted1843
Status: Cryptogenic
Type Locality: Western Europe
Barcodes available: Yes
Notes: Like many of the phyllodocids reported in the New England rapid assessment surveys, P. mucosa is another species with an amphi-Atlantic distribution. In addition, since this species is often easily confused with P. maculata, the full extent of its range is unknown. Both P. mucosa and P. maculata were found together at one site in Maine.
FAMILY POLYNOIDAE
Eunoe nodosa (Sars, Reference Sars1861)
Synonymized names: Antinoe zetlandica Lankester, 1866, Eunoe islandica Hansen, 1878, Harmothoe (Eunoe) nodosa (Sars, Reference Sars1861), Harmothoe nodosa (Sars, Reference Sars1861), Lepidonotus pharetratus Johnston, 1865, Polynoe (Eunoe) islandica Hansen, 1878, Polynoe foraminifera Hansen, 1882, Polynoe nodosa Sars, Reference Sars1861, Polynoe spinulosa Hansen, 1882
Status: Cryptogenic
Type Locality: Norway
Barcodes available: Yes
Notes: Eunoe nodosa has an amphi-Atlantic distribution (Dauvin et al., Reference Dauvin, Dewarumez and Gentil2003; Trott, Reference Trott2004), with its status on the New England coast pending a detailed taxonomic study on the species.
Harmothoe extenuata (Grube, Reference Grube1840)
Synonymized names: Evarnella triannulata (Moore, 1910), Harmothoe plumosa (Grube, Reference Grube1840), Harmothoe sarniensis Lankester, 1866, Hermadion extenuata (Grube, Reference Grube1840), Lagisca ehlersi Malmgren, 1867, Lagisca ehlersi pontica Czerniavsky, 1882, Lagisca extenuata (Grube, Reference Grube1840), Lagisca propinqua Malmgren, 1867, Lepidonotus dumetosus Quatrefages, 1866, Lepidonotus leachii Quatrefages, 1866, Polynoe extenuata Grube, Reference Grube1840, Polynoe plumosa Grube, Reference Grube1840
Status: Cryptogenic
Type Locality: Mediterranean Sea
Barcodes available: Yes
Notes: Harmothoe extenuata was recorded in four rapid assessment surveys dating as far back as 2007. Barnich and Fiege (Reference Barnich and Fiege2009), in a review of the genus Harmothoe, noted that the species has a primarily northeastern Atlantic distribution, with other reports being ‘doubtful due to confusion with other species.’ This implies that the species may have been inaccurately identified in other regions where it has been reported. Considering that the collections at the Smithsonian Institution National Museum of Natural History house more than 900 lots of H. extenuata, many of which have been sampled from the New England region with dates going back to the 1870s and positively identified by both Addison Verrill and Marian Pettibone, this conclusion seems highly unlikely. Instead, we argue that further studies reconciling these vouchers with the most current descriptions, together with genetic studies, will be required to determine the species' status in New England and whether multiple lineages may be involved. As a consequence, we designated the species as cryptogenic for the U.S. Atlantic coast.
Harmothoe imbricata (Linnaeus, Reference Linnaeus1767)
Synonymized names: Aphrodita cirrata Müller, Reference Müller1776, Aphrodita imbricata Linnaeus, Reference Linnaeus1767, Aphrodita lepidota Pallas, 1766, Aphrodita plana Gmelin in Linnaeus, 1788, Aphrodita varians Dalyell, Reference Dalyell1853, Aphrodita violacea Strøm, 1768, Harmothoe hartmanae Pettibone, 1948, Harmothoe imbricata incerta (Bobretzky, 1881), Harmothoe levis Treadwell, 1937, Harmothoe maxillospinosa de Saint-Joseph, 1888, Harmothoe unicolor Baird, 1865, Lepidonote cirrata Örsted, Reference Örsted1843, Polynoe (Harmothoe) imbricata (Linnaeus, Reference Linnaeus1767), Polynoe complanata Quatrefages, 1866, Polynoe incerta Bobretzky, 1881
Status: Cryptogenic
Type Locality: Western Europe
Barcodes available: Yes
Notes: Harmothoe imbricata is a morphologically diverse species that exhibits remarkable color polymorphisms (Nygren et al., Reference Nygren, Norlinder, Panova and Pleijel2011). The most detailed description of the species from the New England region can be found in Pettibone (Reference Pettibone1963). Harmothoe imbricata is also a frequent resident of floating dock communities in New England, found at almost every marina throughout the rapid assessment surveys, and usually associated with tunicate and mussel aggregations (David and Krick, Reference David and Krick2019). A recent population genetic study by David and Cahill (Reference David and Cahill2020) using the COI genetic marker uncovered high levels of genetic diversity across the species' known range, indicating that it might be part of a potential cryptic complex.
Lepidonotus squamatus (Linnaeus, Reference Linnaeus1758)
Synonymized names: Aphrodita armadillo Bosc, Reference Bosc1802, Aphrodita longirostra Bruguière, 1789, Aphrodita pedunculata Pennant, 1777, Aphrodita punctata Müller, 1771, Aphrodita squamata Linnaeus, Reference Linnaeus1758, Lepidonote armadillo (Bosc, Reference Bosc1802) sensu Leidy, 1855, Lepidonote punctata Örsted, Reference Örsted1843, Lepidonotus granularis Leach in Johnston, 1865, Lepidonotus verrucosus Leach in Johnston, 1865, Polinoe scutellata Risso, 1826, Polynoe dasypus Quatrefages, 1866, Polynoe inflata Castelnau, 1842, Polynoe punctata Castelnau, 1842, Polynoe squamata (Linnaeus, Reference Linnaeus1758)
Status: Cryptogenic (Pseudocosmopolitan)
Type Locality: Western Europe
Barcodes available: Yes
Notes: Lepidonotus squamatus, like H. imbricata, is morphologically diverse but also genetically diverse. A large-scale study by Carr et al. (Reference Carr, Hardy, Brown, Macdonald and Herbert2011) found that the species exhibited an unusually high level of genetic divergence, particularly between specimens from the Atlantic and Pacific basins. This indicates that it is likely a ‘pseudo-cosmopolitan’ species as defined by Darling and Carlton (Reference Darling and Carlton2018) and proposed by David and Krick (Reference David and Krick2019). As a consequence, here we designate the species as being cryptogenic until more detailed morphogenetic studies can properly clarify the specimens from New England.
FAMILY SABELLIDAE
Parasabella microphthalma (Verrill, Reference Verrill1873)
Synonymized names: Demonax microphthalma (Verrill, Reference Verrill1873), Sabella microphthalma Verrill, Reference Verrill1873
Status: Native
Type Locality: Vineyard Sound, Massachusetts
Barcodes available: Yes
Notes: The distribution of P. microphthalma is almost exclusive to the western Atlantic with at least one report of the species as far south as the Gulf of Mexico (Felder and Camp, Reference Felder and Camp2009).
Potamilla neglecta (Sars, Reference Sars1851)
Synonymized names: Aspeira modesta Bush, 1905, Sabella breviberbis Langerhans, 1881, Sabella neglecta Sars, Reference Sars1851
Status: Cryptogenic
Type Locality: Norway
Barcodes available: Yes
Notes: Potamilla neglecta has been reported from both Atlantic and Pacific basins (Imajima, Reference Imajima1961; Trott, Reference Trott2004; Mark et al., Reference Mark, Provencher, Albert and Nozères2010) with little or no genetic information available for the species.
FAMILY: SABELLARIIDAE
Sabellaria vulgaris Verrill, Reference Verrill1873
Synonymized names: Sabellaria falcigera Johansson, 1927, Sabellaria floridensis stephensoni Hartman, 1949, Sabellaria varians Webster, Reference Webster1879, Sabellaria vulgaris beaufortensis Hartman, 1944, Sabellaria vulgaris vulgaris Verrill, Reference Verrill1873
Status: Native
Type Locality: Massachusetts, USA
Barcodes available: Yes
Notes: This species has been reported from the northeastern United States to the Gulf of Mexico (Curtis, Reference Curtis1975; Felder and Camp, Reference Felder and Camp2009).
FAMILY SERPULIDAE
Circeis spirillum (Linnaeus, Reference Linnaeus1758)
Synonymized names: Dexiospira spirillum (Linnaeus, Reference Linnaeus1758), Janua (Dexiospira) spirillum (Linnaeus, Reference Linnaeus1758), Janua (Dexiospira) spirillum lucida (Montagu, Reference Montagu1803), Serpula cornea Adams, 1800, Serpula lucida Montagu, Reference Montagu1803, Serpula sinistorsa Montagu, Reference Montagu1803, Serpula spirillum Linnaeus, Reference Linnaeus1758, Spirillum pellucidum Oken, 1815, Spirorbis comptus Bush, 1905, Spirorbis cornea (Adams, 1800), Spirorbis fewkesii Bush in Sumner, Osburn & Cole, 1913, Spirorbis lucida (Montagu, Reference Montagu1803), Spirorbis reflexa Adams in Morch, 1863, Spirorbis spirillum (Linnaeus, Reference Linnaeus1758)
Status: Cryptogenic
Type Locality: Europe
Barcodes available: Yes
Notes: Very little is known about the biogeography of C. spirillum.
Hydroides dianthus (Verrill, Reference Verrill1873)
Synonymized names: Eupomatus dianthus (Verrill, Reference Verrill1873), Serpula dianthus Verrill, Reference Verrill1873, Serpula dianthus citrina Verrill, Reference Verrill1873
Status: Native
Type Locality: Connecticut, USA
Barcodes available: Yes
Notes: Hydroides dianthus is a well-known fouling polychaete in the New England region and has an almost cosmopolitan distribution (Sun et al., Reference Sun, Wong, Keppel, Williamson and Kupriyanova2017). A previous study by Sun et al. (Reference Sun, Wong, Keppel, Williamson and Kupriyanova2017) found that populations of H. dianthus from the Mediterranean had higher levels of haplotype diversity compared to populations from the eastern United States, arguing that the former may be the native range for this worm. However, several factors elevate genetic diversity in a region (e.g., multiple introductions) or reduce it (e.g., historical bottlenecks, sampling bias). In fact, a large portion of the eastern United States (e.g., Maine, Georgia, and the Carolinas) were not sampled in the Sun et al. (Reference Sun, Wong, Keppel, Williamson and Kupriyanova2017) study. Therefore, until a more comprehensive population genetic study is performed that adequately samples its eastern US range, we retain its native status in this region.
Janua heterostropha (Montagu, Reference Montagu1803)
Synonymized names: Bushiella (Jugaria) granulata (Linnaeus, Reference Linnaeus1767), Janua (Dexiospira) pagenstecheri (Quatrefages, 1866), Janua (Dexiospira) pusilloides (Bush, 1905), Janua pagenstecheri (Quatrefages, 1866), Jugaria granulata (Linnaeus, Reference Linnaeus1767), Laeospira granulata (Linnaeus, Reference Linnaeus1767), Mera pusilla Saint-Joseph, 1894, Serpula granulata Linnaeus, Reference Linnaeus1767, Serpula heterostropha Montagu, Reference Montagu1803, Serpula minuta Montagu, Reference Montagu1803, Serpula sulcuta Adams, 1797, Spirorbis (Dexiospira) pusilloides (Bush, 1905), Spirorbis (Janua) epichysis Bailey, 1969, Spirorbis (Janua) gnomonicus Bailey, 1969, Spirorbis affinis Levinsen, 1883, Spirorbis epichysis Bailey, 1970, Sprirorbis fabricii Malmgren, 1867, Spirorbis granulata (Linnaeus, Reference Linnaeus1767), Spirorbis granulatus (Linnaeus, Reference Linnaeus1767), Spirorbis pagenstecheri Quatrefages, 1866, Spirorbis pagenstecheri incoloris Gee, 1964, Spirorbis pusilloides Bush, 1905, Spirorbis sulcata Adams in Morch, 1863
Status: Cryptogenic
Type Locality: England
Barcodes available: No
Notes: Janua heterostropha, like H. dianthus, is a well-known fouling serpulid reported under this and other names from all oceans of the world (Knight-Jones et al., Reference Knight-Jones, Knight-Jones and Kawahara1975), doubtless in large part as a result of introductions via historical and contemporary shipping. Knight-Jones et al. (Reference Knight-Jones, Knight-Jones and Kawahara1975) suggested a European origin based on this species' breadth of physiological and ecological adaptations of this species to the climate of the Northeast Atlantic Ocean. However, in the absence of genetic data, and given the probability of a cryptic species complex, we conservatively treat it as cryptogenic here.
FAMILY SPIONIDAE
Dipolydora socialis (Schmarda, Reference Schmarda1861)
Synonymized names: Leucodore socialis Schmarda, Reference Schmarda1861, Polydora socialis (Schmarda, Reference Schmarda1861)
Status: Unresolved cosmopolitan species
Type Locality: Chile
Barcodes available: Yes
Notes: Dipolydora socialis is assumed to have a cosmopolitan distribution because of its occurrence in multiple oceanic basins across the globe (see David et al., Reference David, Williams and Simon2021 and references therein); it falls into the ‘unresolved cosmopolitan’ category of Darling and Carlton (Reference Darling and Carlton2018). Dipolydora socialis is frequently found associated with encrusting organisms such as sponges and coralline algae. In addition, the species has also been found associated with boring polychaetes that burrow into the shells of commercially reared shellfish such as oysters and abalone. As a consequence, it is possible that through vectors such as shipping and the aquaculture trade, the species has been able to disperse to multiple biogeographic regions. It should also be noted that many of the reports of D. socialis have not been accompanied by thorough taxonomic investigations, and considering the lack of genetic data available (Radashevsky et al., Reference Radashevsky, Pankova, Malyar and Carlton2023) especially from the type localities of this species, the possibility of the existence of a cryptic complex cannot be ruled out.
Polydora aggregata Blake, Reference Blake1969
Synonymized names: none
Status: Native
Type Locality: Maine, USA
Barcodes available: No
Notes: Adults of Polydora aggregata were first described by Blake (Reference Blake1971), preceded by his description of its larvae (Blake, Reference Blake1969). It ranges from northern New England (its type locality) to the Gulf of Mexico (Blake, Reference Blake1971). Five specimens from Limfjord, Denmark, collected in 1981 and identified by Mary Petersen as Polydora cf. aggregata, on deposit at the Smithsonian Institution's National Museum of Natural History (USNM 71444), require re-examination.
Polydora colonia Moore, Reference Moore1907
Synonymized names: Polydora ancistra Jones, 1962, Polydora hoplura inhaca Day, 1957
Status: Cryptogenic
Type Locality: Massachusetts, USA
Barcodes available: Yes
Notes: Polydora colonia is exclusively a sponge borer and does not appear to exhibit any host-specificity (David and Williams, Reference David and Williams2012; Gouillieux et al., Reference Gouillieux, Masse, Derrien-Courtel, Bachelet and Lavesque2022). The species is morphologically similar to another exclusive sponge borer, Polydora spongicola, but a study by McGrail (Reference McGrail2023) found that both species were genetically distinct, with P. colonia possessing species-specific posterior recurved ‘boat’ hooks that P. spongicola lacked. There is a limited amount of genetic data available for this species on the GenBank database. Historical reports of the species have largely been confined to the east coast of the United States. Day (Reference Day1967) reported the species from South Africa, but a revision of his work currently underway in light of modern integrative taxonomic methods has yet to report this species in the region (see Simon et al., Reference Simon, Kara, Clarke and Sedick2022). The species was described on the New England coast by Blake (Reference Blake1971) and has been frequently reported from various sites along the western Atlantic. However, due to gaps in our understanding of this species' biogeography, David and Williams (Reference David and Williams2012) categorized P. colonia as cryptogenic for the region, and we reiterate that status here.
Polydora cornuta Bosc, Reference Bosc1802
Synonymized names: Polydora amarincola Hartman, 1936, Polydora ligni Webster, Reference Webster1879, Polydora littorea Verrill, 1881, Spio caudatus Lamarck, 1818
Status: Cryptogenic
Type Locality: South Carolina, USA
Barcodes available: Yes
Notes: Polydora cornuta is the type species for the genus Polydora (Blake, Reference Blake1971). The species has a reported cosmopolitan distribution and is arguably the most widely distributed polydorid, being found within mudpacks on fouling communities and within the crevices of molluscs (Radashevsky, Reference Radashevsky2005). The species was reported throughout New England as far back as 1884 (Webster and Benedict, Reference Webster and Benedict1884) and was at one point considered native to the region, prior to Rice et al.'s (Reference Rice, Karl and Rice2008) discovery of reproductively isolated cohorts. Polydora cornuta remains an unresolved species complex, but despite the genetic divergence, Radashevsky (Reference Radashevsky2005), using an extensive morphological dataset, concluded that there were insufficient taxonomically informative differences to warrant an assignment of unique species status to any of the isolated populations. While found as early as 1802 on the U.S. Atlantic coast, shipping from Europe and eventually from around the world, has touched the South Carolina coast since the 1500s. Nevertheless, it is regarded as introduced to the Pacific coast from British Columbia to California, initially with the extensive transport of Atlantic oysters (Crassostrea virginica) to the Eastern Pacific commencing in the 1870s (Cohen and Carlton, Reference Cohen and Carlton1995).
Polydora websteri Hartman in Loosanoff and Engle, Reference Loosanoff and Engle1943
Synonymized names: none
Status: Cryptogenic
Type Locality: Connecticut, USA
Barcodes available: Yes
Notes: Globally, Polydora websteri is an economically important and genetically homogenous species (Rice et al., Reference Rice, Lindsay and Rawson2018; Rodewald et al., Reference Rodewald, Snyman and Simon2021; Davinack et al., Reference Davinack, Strong and Brennessel2024). It is an obligate shell borer where it is responsible for burrowing into the shells of commercially reared shellfish on every continent except Antarctica (Radashevsky et al., Reference Radashevsky, Lana and Nalesso2006; Read, Reference Read2010; Sato-Okoshi et al., Reference Sato-Okoshi, Okoshi, Abe and Li2013; Waser et al., Reference Waser, Lackschewitz, Knol, Reise, Mathias Wegner and Thieltges2020; Rodewald et al., Reference Rodewald, Snyman and Simon2021; Davinack et al., Reference Davinack, Strong and Brennessel2024). The species' global genetic homogeneity has been attributed to repeated introductions as a result of the aquaculture trade (Rodewald et al., Reference Rodewald, Snyman and Simon2021). Polydora websteri was once regarded as native to the New England region, but a study by Rice et al. (Reference Rice, Lindsay and Rawson2018) focusing on the COI marker found that populations from Asia had the highest genetic diversity, indicating that this region could be the actual native range for P. websteri. However, it should be noted that genetic diversity can be elevated in a population if multiple introductions are occurring from different regions (Roman and Darling, Reference Roman and Darling2007), while such diversity can be greatly reduced in another region as a result of historical bottlenecks. Therefore, we classify the species here as cryptogenic until a more comprehensive study incorporating additional markers and population genetic analyses can further clarify the issue.
Spio setosa Verrill, Reference Verrill1873
Synonymized names: Spio robusta Verrill, Reference Verrill1873
Status: Native
Type Locality: Massachusetts (Vineyard Sound), USA
Barcodes available: Yes
Notes: Spio setosa is arguably one of the most well-studied spionids ecologically. Its feeding behavior has been reported by Bock and Miller (Reference Bock and Miller1997) and Dauer (Reference Dauer2000), its reproduction and larval development by Simon (Reference Simon1967), its growth rates by Hentschel and Herrick (Reference Hentschel and Herrick2005), and its bioturbative contributions to its intertidal habitat by Waldbusser et al. (Reference Waldbusser, Marinelli, Whitlatch and Visscher2004). Based on the most recent description by Maciolek (Reference Maciolek1990), the species can easily be distinguished from other members of the genus Spio and is therefore unlikely to be part of a cryptic complex. Based on all of the historic and current occurrence reports, along with location information on every S. setosa voucher from the Smithsonian Institution National Museum of Natural History, the species' range appears to be exclusive to the western Atlantic indicating that it might be endemic to this region.
Streblospio benedicti Webster, Reference Webster1879
Synonymized names: Streblospio lutincola Hartman, 1936
Status: Native
Type Locality: New Jersey, USA
Barcodes available: Yes
Notes: Streblospio benedicti has a relatively widespread distribution, being reported as introduced to the Pacific coasts of the United States and to several regions in western Europe (Pernet and McArthur, Reference Pernet and McArthur2006). A genetic study by Mahon et al. (Reference Mahon, Mahon, Dauer and Halanych2009) confirmed S. benedicti as a distinct species, undoing a previous synonymization with Streblospio shrubsolii (Buchanan, 1890) (Foster, Reference Foster1971) and reiterating the native status of the species in the western Atlantic.
FAMILY SYLLIDAE
Myrianida prolifera (Müller, Reference Müller1788)
Synonymized names: Autolytus agassizii Quatrefages, 1866, Autolytus ehbiensis Saint Joseph, 1887, Autolytus hesperidum Claparède, 1868, Autolytus prolifer (Müller, Reference Müller1788), Autolytus prolifera (Müller, Reference Müller1788), Crithida prolifera (Müller, Reference Müller1788), Nereis prolifera Müller, Reference Müller1788
Status: Cryptogenic
Type Locality: Norway
Barcodes available: Yes
Notes: Myrianida prolifera has been reported throughout the western and eastern Atlantic, Mediterranean Sea, and Arctic Oceans (Holly, Reference Holly1938; Trott, Reference Trott2004; Musco and Giangrande, Reference Musco and Giangrande2005). Aside from a single M. prolifera barcode generated as a result of a redescription of another syllid (Nygren and Pleijel, Reference Nygren and Pleijel2010), there are no genetic data available for the species and very little identified material available in national collections. Further studies are needed to determine the status of this species in the New England region.
Proceraea prismatica (Müller, Reference Müller1776)
Synonymized names: Amytis prismatica (Müller, Reference Müller1776), Autolytus (Proceraea) trilineatus Berkeley & Berkeley, 1945, Autolytus incertus Malmgren, 1867, Autolytus longosetosus (Örsted, Reference Örsted1843), Autolytus prismaticus (Müller, Reference Müller1776), Autolytus trilineatus Berkeley & Berkeley, 1945, Nereis prismatica Müller, Reference Müller1776, Nereisyllis prismatica (Müller, Reference Müller1776), Polybostrichus longosetosus Örsted, Reference Örsted1843, Polybostrichus longosetus [auct.], Proceraea gracilis [auct. misspelling]
Status: Native
Type Locality: Greenland (Arctic Ocean)
Barcodes available: Yes
Notes: Proceraea prismatica appears to have a very restricted range in the Arctic Ocean (Pettibone, Reference Pettibone1963; Jørgensen and Gulliksen, Reference Jørgensen and Gulliksen2001). Early reports from the eastern Atlantic were considered rare, isolated, or dubious by Hamond (Reference Hamond2002). Very little is known about the worm's distribution in other regions where it has been reported, including the western Atlantic and the north Pacific. In New England, P. prismatica was recorded in only one survey (2000) at five marinas – three in the Gulf of Maine, one in New Hampshire, and one in Boston, Massachusetts. Considering the latitudinal consistency, it is likely that these records may represent a part of its natural boreal and sub-boreal distribution.
Salvatoria clavata (Claparède, Reference Claparède1863)
Synonymized names: Brania clavata (Claparède, Reference Claparède1863), Grubea clavata (Claparède, Reference Claparède1863), Grubea dolichopoda Webster, Reference Webster1879, Grubea fusifera Quatrefages, 1866, Grubea websteri Verrill, 1882, Grubeosyllis clavata (Claparède, Reference Claparède1863), Pseudobrania clavata (Claparède, Reference Claparède1863), Salvatoria dolichopoda (Marenzeller, 1874), Syllis clavata Claparède, Reference Claparède1863
Status: Cryptogenic
Type Locality: Normandy, France
Barcodes available: Yes
Notes: Salvatoria clavata was reported as the most dominant polychaete associated with marinas on the Iberian Peninsula and was regarded as an ‘opportunistic’ syllid in that study since it thrived in areas of environmental stress (Fernandez-Romero et al., Reference Fernandez-Romero, Moreira and Guerra-Garcia2019). In contrast, on the New England coast, the species was only reported in one survey in 2000 at two sites (Woods Hole Coast Guard Station and the Marine Biological Laboratory, both in Woods Hole, Massachusetts) and has not appeared in any subsequent survey. The species has been reported worldwide (San Martín, Reference San Martín and Ramos2003; Nascimento et al., Reference Nascimento, Fukuda, Paresque, Nogueira and Paiva2021). However, with only a single 18S rRNA sequence available on GenBank and no population genetic or biogeographic information available, it is impossible to determine its introduced status in any of these regions. To further complicate matters, San Martín (Reference San Martín and Ramos2003) noticed variation within taxonomically informative traits, indicative of a potential cryptic species complex.
Syllis gracilis Grube, Reference Grube1840
Synonymized names: Syllis (Syllis) gracilis Grube, Reference Grube1840, Syllis (Syllis) longissima Gravier, 1900, Syllis brachycirris Grube, 1857, Syllis buchholziana Grube, 1877, Syllis longissima Gravier, 1900, Syllis mixosetosa Bobretzky, 1870, Syllis navicellidens Czerniavksy, 1881, Syllis nigrovittata Czerniavsky, 1881, Syllis nigro-vittata Czerniavsky, 1881, Syllis palifica Ehlers, 1901, Syllis quadridentata Czerniavsky, 1881, Syllis vancaurica Grube, 1867
Status: Cryptogenic (Pseudocosmopolitan)
Type Locality: Gulf of Naples
Barcodes available: Yes
Notes: Syllis gracilis is an unresolved species complex and arguably one of the most difficult ones that has yet to be resolved. A complex of two lineages was first discovered using allozymes by Maltagliati et al. (Reference Maltagliati, Peru, Casu, Rossi, Lardicci, Curini-Galletti and Castelli2000). Since then, it has been shown that the species exhibits a remarkably high level of phenotypic plasticity in various regions (Langeneck et al., Reference Langeneck, Scarpa, Maltagliati, Sanna, Barbieri, Cossu, Mikac, Curini-Galletti, Castelli and Casu2020), which ultimately nullifies general taxonomic keys. Furthermore, even more divergent lineages have been discovered that do not correspond to morphotypes (Alvarez-Campos et al., Reference Alvarez-Campos, Giribet and Riesgo2017). Finally, different species delineation tests have yielded conflicting results (Langeneck et al., Reference Langeneck, Scarpa, Maltagliati, Sanna, Barbieri, Cossu, Mikac, Curini-Galletti, Castelli and Casu2020). This species can best be thought of as pseudocosmopolitan and is regarded as cryptogenic here.
FAMILY TEREBELLIDAE
Amphitrite cirrata Müller, Reference Müller1776
Synonymiz1ed names: Amphiro cirrata (Müller, Reference Müller1776), Amphitrite palmata Moore, 1906, Amphitrite radiata Moore, 1908, Nereis cirrosa Linnaeus, Reference Linnaeus1767, Sabella cirrata (Müller, Reference Müller1776), Spio cirrata König [pre-Linnaean], Terebella cirrata (Müller, Reference Müller1776), Terebella cirrhata [auct. misspelling], Terebella montagui Quatrefages, 1866, Teredo arenaria Forsskål, 1775
Status: Cryptogenic
Type Locality: Scandinavia (exact location unknown)
Barcodes available: Yes
Notes: The location of the type material for A. cirrata is unknown but is believed to have originated from the Scandinavian region (Norway, Denmark, or Iceland). While this species has an extensive distribution with reports from the Mediterranean and the Pacific Ocean, and Africa, there are no molecular data to support these reports. Furthermore, Jirkov (Reference Jirkov2020) did not find the species in any of the Mediterranean or Pacific locations where it was reported. Jirkov (Reference Jirkov2020) also cast doubt on Day's (Reference Day1967) report from the Cape of Good Hope and Senegal along with Hartman's (Reference Hartman1969) description of the species from California. David and Krick (Reference David and Krick2019) used COI sequence data to report the species for the first time in rapid assessment surveys from New England (referred to as Amphitrite cf. cirrata), specifically at marinas located in Maine, Massachusetts, and New Hampshire. However, considering the lack of molecular data from many of the aforementioned regions, along with its challenging taxonomy (Hutchings and Kupriyanova, Reference Hutchings and Kupriyanova2018; Jirkov, Reference Jirkov2020), we categorize this species as cryptogenic to the New England coast.
Loimia sp.
Status: Cryptogenic
Notes: A Loimia species in our RAS samples (for example, from 2000 and 2003) has long been presented in Atlantic coast literature as Loimia medusa (Savigny, Reference Savigny1822), a tropical species with a type locality of the Gulf of Suez. Loimia ‘medusa’ (Savigny, Reference Savigny1822) has been reported from many habitats worldwide in the Atlantic, Pacific, and Indian Oceans, and doubtless represents a species complex, perhaps including introduced tropical or subtropical populations. Our biofouling Loimia from the cold waters of New England represents an as-yet to be resolved species.
Neoamphitrite figulus (Dalyell, Reference Dalyell1853)
Synonymized names: Amphitrite figulus (Dalyell, Reference Dalyell1853), Amphitrite johnstoni Malmgren, 1866, Amphitrite nana Claparède, 1870, Amphitrite stimpsoni Meyer, 1912, Terebella elongata Quatrefages, 1866, Terebella figulus Dalyell, Reference Dalyell1853
Status: Cryptogenic
Type Locality: United Kingdom
Barcodes available: Yes
Notes: Neoamphitrite figulus has been reported in both the eastern and western Atlantic in addition to the North Sea and the Mediterranean (Hartmann-Schröder, Reference Hartmann-Schröder1996; Brunel et al., Reference Brunel, Bosse and Lamarche1998; Bellan, Reference Bellan2001). The species is a well-known epibiont of the European oyster (Ostrea edulis) (Smyth and Roberts, Reference Smyth and Roberts2010), which has had a long history of translocation to various parts of the world for oyster cultivation (Haydar and Wolff, Reference Haydar and Wolff2011). As a consequence, it is difficult to determine the true native range for N. figulus, especially considering that there is a paucity of genetic data available for this species.
Nicolea sp.
Status: Native
Notes: While the European terebellid Nicolea zostericola Ørsted, 1844, has been long reported from the Northwestern Atlantic Ocean, including New England (e.g., Sebens, Reference Sebens1986, from a subtidal rock wall in Nahant, Massachusetts), and while Lopez et al. (Reference Lopez, Carey, Carlton, Cerrato, Dam, DiGiovanni and Elphick2014) considered it a possible cryptogenic species in Long Island Sound, de Matos Nogueira (Reference de Matos Nogueira2008, a paper overlooked by Lopez et al., Reference Lopez, Carey, Carlton, Cerrato, Dam, DiGiovanni and Elphick2014) suggested that records of this species likely represent native taxa, including N. cetrata (now Pista cetrata), N. simplex, or N. lazowasemi (incorrectly originally spelled as lazo-wasemi). Reported in the RAS 2000, 2007, and 2013 surveys, we have not had access to these specimens to re-determine the species.
Pista palmata (Verrill, Reference Verrill1873)
Synonymized names: Scionopsis palmata Verrill, Reference Verrill1873
Status: Native
Type Locality: Vineyard Sound, Massachusetts, USA
Barcodes available: Yes
Notes: Pista palmata has a restricted distribution in the western Atlantic. The majority of occurrences of the species are between the New England region, Gulf of Mexico and the Caribbean (Hartman, Reference Hartman1951, Reference Hartman1959; Felder and Camp, Reference Felder and Camp2009; Miloslavich et al., Reference Miloslavich, Diaz, Klein, Alvarado, Diaz, Gobin, Escobar-Briones, Cruz-Motta, Weil, Cortes, Bastidas, Robertson, Zapata, Martin, Castillo, Kazandjian and Ortiz2010).
Terebella verrilli Holthe, Reference Holthe1986
Synonymized names: Lepraea rubra Verrill, Reference Verrill1873, Terebella rubra (Verrill, Reference Verrill1873)
Status: Native
Type Locality: Vineyard Sound, Massachusetts, USA
Barcodes available: No
Notes: According to de Matos Nogueira (Reference de Matos Nogueira2008), who examined T. verrilli specimens from both the western Atlantic, including the type locality, and the Mexican-Caribbean, there is ample morphological variation in specimens. Coupled with similar findings from previous reports, this suggests that a species complex could be present within what is likely a group native to the Northwest Atlantic Ocean.
Discussion
The current study presents the first annotated checklist of polychaetes from floating dock communities in New England and adjacent New York areas, marking a significant advancement in our understanding of marine biodiversity in marina environments. These Rapid Assessment Surveys allow for the detection of trends and anomalies, the latter being an important feature as they are often associated with the appearance of a non-indigenous species as a result of translocation or range expansion of southern fauna. The predominance of cryptogenic species (61%) in our study highlights a critical challenge in marine biodiversity studies – the difficulty in determining the native status of many species. This is particularly relevant for polychaetes, which have complex life histories and exhibit high levels of cryptic diversity (Nygren, Reference Nygren2014), complicating efforts to categorize them as native or non-native. The high proportion of cryptogenic species also echoes the findings of previous studies, emphasizing the need for comprehensive taxonomic and genetic analyses to unravel the origins of these species (Darling and Carlton, Reference Darling and Carlton2018; David and Krick, Reference David and Krick2019). Equally problematic is that many of the polychaetes on this checklist have been the subject of various ecological and physiological experimental research, with some, such as P. dumerili, serving as important laboratory model systems despite their taxonomy being unresolved. The apparently low number of introduced species in the current data set (we report only one, Ctenodrilus serratus) should not be taken to mean that non-native polychaetes are rare in New England, but, rather, may reflect that a large number of species are currently categorized as cryptogenic, the majority of which likely arrived long before any biosecurity efforts (such as the control of ballast water) were in place.
The vast majority of the polychaete families recovered was tubicolous (e.g., Spionidae, Serpulidae, Terebellidae, Sabellidae) which is not surprising considering that this ecological niche easily lends itself to anthropogenic-mediated transport mechanisms. For example, the adults of spionids, sabellids and terebellids live within mud-tubes of varying strength that can be found lodged among a variety of fouling organisms (such as mussels, barnacles, sponges, and algae) that commonly attach themselves to vessels. Members of the Polydora-complex that parasitize commercially reared shellfish are a prime example of species that can quickly spread across multiple biogeographic regions by hitchhiking on imported oysters, as well as burrowing into calcareous substrates (such as barnacles and oysters) on vessels (Williams et al., Reference Williams, Matthee and Simon2017). Serpulids are unique due to their ability to create and reside within calcium carbonate tubes that can encrust on the hulls of ships, oysters, and hard structures that can be translocated from one region to the next (Bastida-Zavala et al., Reference Bastida-Zavala, McCann, Keppel and Ruiz2017). Pettengill et al. (Reference Pettengill, Wendt, Schug and Hadfield2007) using microsatellite loci to assess the genetic connectivity of the serpulid Hydroides elegans found that its dispersal across multiple global regions was likely the result of biofouling on ships.
The current checklist consists of several species that are part of cryptic complexes making it difficult to determine the species' true range, that in turn also makes it difficult to determine their origin. The most problematic species are those that have historically been referred to as ‘cosmopolitan’. In developing this checklist, we adopted Hutchings and Kupriyanova's (Reference Hutchings and Kupriyanova2018) null hypothesis of assuming that these species have a narrower range than reported in the literature. The only exception was for species whose populations have been assessed genetically and whose biogeography has been resolved.
Our findings also revealed no significant difference in polychaete composition across marinas but did show a significant difference across years sampled. As expert polychaete taxonomists were involved over the years, we believe that these differences are largely not related to different workers being involved over time, although there are possible exceptions (as noted below). This temporal variation underscores the dynamic nature of floating dock communities, which can be influenced by a variety of factors including changes in environmental conditions, marina management practices, and species dispersal events. The lack of distinct clustering in the NMDS plot suggests that while composition varies over time, these changes are not leading to the formation of distinct community assemblages by year. This could indicate a high degree of community resilience or reflect the influence of similar environmental and anthropogenic pressures across the study period.
The application of DNA barcoding in the identification process of the Rapid Assessment Survey, as initiated by David and Krick (Reference David and Krick2019) has proven to be invaluable in increasing the resolution of species diversity. For example, the phyllodocid Phyllodoce groenlandica, which has been reported on the New England coast for more than a century including during earlier surveys, was not recovered in the 2018 survey, but the morphologically similar P. maculata and P. mucosa were. This brings up the question of whether P. groenlandica had been misidentified during past biomonitoring surveys and opens the door for a revision of New England phyllodocids. As taxonomy continues to evolve with the integration of genetic data, future surveys should incorporate these methodologies including eDNA surveys to ensure accurate species identification and status determination.
Limitations and Future Directions
One limitation of the rapid assessment survey is the reliance on presence/absence data, which does not account for the relative abundance of species. This built-in limitation is intentional as it allows for broader explorations, and can quickly detect anomalies and changes in species composition. Additionally, while our study has made significant strides in cataloguing polychaete diversity in the New England region, the high number of cryptogenic species identified points to the need for further taxonomic and biogeographic research to clarify the origins of many species. Unfortunately, due to a dearth of taxonomic specialists (Kholia and Fraser-Jenkins, Reference Kholia and Fraser-Jenkins2011; McClain, Reference McClain2011), the rate at which regional taxonomic revisions are occurring cannot keep pace with the speed of biotic changes. This disconnect can have severe consequences on biomonitoring surveys, which in turn can hamper conservation and management efforts. Finally, while DNA barcoding has invigorated the science of taxonomy, as Hutchings and Kupriyanova (Reference Hutchings and Kupriyanova2018) state, ‘not all sequences are created equal.’ Voucher-linked sequence data are good, voucher linked-sequence data from the type locality are even better, and barcodes obtained from type material are the gold-standard. Therefore, prudent mining of sequence data, regardless of which database they are pulled from, is a critical but often overlooked step in the barcoding workflow.
In conclusion, our findings provide not only a baseline for future research but also a framework for ongoing monitoring efforts aimed at understanding the integrity of marine ecosystems in the New England region. By highlighting the importance of these surveys and the complexities of polychaete taxonomy and biogeography, this study contributes to the broader understanding of marine biodiversity and the scale of non-native biodiversity in a rapidly changing marine environment.
Data
The data that support the findings of this study are available from the corresponding author, [A. D.], upon reasonable request.
Acknowledgements
We are grateful to colleagues who have previously contributed to identifying species on the New England rapid assessments over nearly 20 years. These include Harlan Dean, Leslie Harris, Eric Lazo-Wasem, and Jason Williams. We also thank two anonymous reviewers of this paper for their useful comments and insights.
Author Contributions
A. A. D. formulated the research question and designed the study, A. A. D., J. T. C. and J. P. carried out the study, A. A. D. and J. T. C. analysed the data and interpreted the findings. All authors contributed to writing the article.
Financial Support
This research received no specific grant from any funding agency, commercial or not-for-profit sectors.
Competing interest
None.
Ethical Standards
All ethical standards were adhered to for this study.






