- Akt
protein kinase B/Akt
- BBB
blood–brain barrier
- BBB
blood–brain barrier
- BDNF
brain-derived neurotrophic factor
- CREB
cAMP-response element-binding protein
- ERK
extracellular signal-regulated protein kinase
- LTP
long-term potentiation
- MAPK
mitogen-activated protein kinase
- mTOR
mammalian target of rapamycin
- PI3K
phosphoinositide 3-kinase
Representing one of the most important lifestyle factors, diet can strongly influence the incidence and onset of CVD and neurodegenerative disorders, and thus a healthy diet is an essential factor for healthy ageing. Various phytochemical constituents of foods and beverages, in particular a class of photochemicals termed flavonoids, have been avidly investigated in recent years. A number of dietary intervention studies in human subjects and animals, in particular those using foods and beverages derived from Vitis vinifera (grape), Camellia sinensis (tea), Theobroma cacao (cocoa) and Vaccinium spp. (blueberry), have demonstrated beneficial effects on vascular function and mental performance. While such foods and beverages differ greatly in chemical composition, macro- and micronutrient content and energy load per serving, they have in common that they are amongst the major dietary sources of flavonoids. Dietary intervention studies in several mammalian species, including man, using flavonoid-rich plant or food extracts have indicated that flavonoids are capable of improving both memory and learning(Reference Youdim and Joseph1–Reference Wang, Wang, Wu and Cai7), via their ability to protect vulnerable neurons, enhance existing neuronal function and stimulate neuronal regeneration. In addition, their neuroprotective potential is well reported and they have been shown to protect against neuronal death in both oxidative stress-induced(Reference Inanami, Watanabe, Syuto, Nakano, Tsuji and Kuwabara8) and Aβ-induced neuronal-death models(Reference Luo, Smith, Paramasivam, Burdick, Curry, Buford, Khan, Netzer, Xu and Butko9). Furthermore, evidence supports the beneficial and neuromodulatory effects of flavonoid-rich ginkgo biloba (Ginkgo biloba L.) extracts, particularly in connection with age-related dementias and Alzheimer's disease(Reference Luo, Smith, Paramasivam, Burdick, Curry, Buford, Khan, Netzer, Xu and Butko9–Reference Zimmermann, Colciaghi, Cattabeni and Di Luca11) and the citrus flavanone tangeretin has been observed to help maintain nigrostriatal integrity and functionality following lesioning with 6-hydroxydopamine(Reference Datla, Christidou, Widmer, Rooprai and Dexter12).
Historically, the biological actions of flavonoids have been attributed to their antioxidant properties(Reference Rice-Evans, Miller and Paganga13), through their ability to scavenge reactive species(Reference Pollard, Kuhnle, Vauzour, Vafeiadou, Tzounis, Whiteman, Rice-Evans and Spencer14) or through their influences on the intracellular redox status. However, it has been speculated that their classical H-donating antioxidant activity is not the explanation for the bioactivity of flavonoids in vivo, particularly in the brain where their levels are very low. Indeed, it has become evident that flavonoids are more likely to exert their neuroprotective actions by: the modulation of intracellular signalling cascades that control neuronal survival, death and differentiation; affecting gene expression; interactions with mitochondria(Reference Williams, Spencer and Rice-Evans15–Reference Schroeter, Boyd, Spencer, Williams, Cadenas and Rice-Evans17). The present review will highlight the impact of flavonoids on learning, memory and neuro-cognitive performance. In particular, it will highlight probable mechanisms that underpin such actions in the brain, including their interactions with neuronal intracellular signalling pathways pivotal in controlling long-term potentiation (LTP) and memory in human subjects.
Flavonoid structure, source and metabolism
Flavonoids comprise the most common group of polyphenolic compounds in the human diet and are found ubiquitously in plants. Major dietary sources of flavonoids include fruits, vegetables, cereals, tea, wine and fruit juices (for review, see Manach et al. (Reference Manach, Scalbert, Morand, Remesy and Jimenez18)). Flavonoids consist of two aromatic C rings, benzopyran (rings A and C) and benzene (ring B), and may be divided in six subgroups based on the extent of the oxidation of ring C, the hydroxylation pattern of the ring structure and the substitution of the C-3-position (Fig. 1). The main dietary groups of flavonoids are: flavonols, e.g. kaempferol and quercetin, which are found in onions (Allium cepa L.), leeks (Allium ampeloprasum var. porrum (L.)) and broccoli; flavones, e.g. apigenin and luteolin, which are found in parsley (Petroselinum crispum) and celery (Apium graveolens L.); isoflavones, e.g. daidzein and genistein, which are mainly found in soyabean and soya products; flavanones, e.g. hesperetin and naringenin, which are mainly found in citrus fruit and tomatoes; flavanols, e.g. catechin, epicatechin, epigallocatechin and epigallocatechin gallate, which are abundant in green tea, red wine and cocoa; anthocyanidins, e.g. pelargonidin, cyanidin and malvidin, whose sources include red wine and berry fruits. Further information relating to the structure and classes of flavonoids may be found in the thorough review by Manach et al. (Reference Manach, Scalbert, Morand, Remesy and Jimenez18).

Fig. 1. The structures of the main classes of flavonoids. The major differences between the individual groups reside in the hydroxylation pattern of the ring-structure, the extent of saturation of the C-ring and the substitution of in the 3-position: (A) general structure of flavonoids, (B) structure of flavonols and flavones, (C) structure of flavanols, also referred as flavan-3-ols, (D) structure of anthocyanidins, (E) structure of flavanones and flavanonols and (F) structure of isoflavones. EGC, epigallocatechin; ECG, epicatechin gallate; EGCG, EGC gallate.
Although flavonoids display potent antioxidant capacity in vitro (Reference Rice-Evans, Miller and Paganga13, Reference Rice-Evans19, Reference Rice-Evans20), during absorption they are extensively metabolised, resulting in substantial alteration of their redox potentials. For example, the majority of flavonoid glycosides and aglycones present in plant-derived foods are extensively conjugated and metabolised during absorption (for reviews, see Spencer et al. (Reference Spencer, Schroeter, Rechner and Rice-Evans21, Reference Spencer, Abd El Mohsen, Minihane and Mathers22)). In particular, they are subject to extensive phase I de-glycosylation and phase II metabolism of the resulting aglycones to glucuronides, sulfates and O-methylated forms during transfer across the small intestine(Reference Spencer, Schroeter, Rechner and Rice-Evans21, Reference Spencer, Chowrimootoo, Choudhury, Debnam, Srai and Rice-Evans23, Reference Spencer24) and then again in the liver. Further transformation has been reported in the colon where the enzymes of the gut microflora degrade flavonoids to simple phenolic acids(Reference Scheline25). In addition, flavonoids may undergo at least three types of intracellular metabolism: oxidative metabolism; P450-related metabolism; conjugation with thiols, particularly glutathione(Reference Spencer, Kuhnle, Williams and Rice-Evans26, Reference Spencer, Schroeter, Crossthwaithe, Kuhnle, Williams and Rice-Evans27). Circulating metabolites of flavonoids such as glucuronides, sulfates and conjugated O-methylated forms or intracellular metabolites such as flavonoid–glutathione adducts have greatly reduced antioxidant potential(Reference Spencer, Schroeter, Crossthwaithe, Kuhnle, Williams and Rice-Evans27). Indeed, studies have indicated that although such conjugates and metabolites may participate directly in plasma antioxidant reactions and in scavenging reactive oxygen and nitrogen species in the circulation, their effectiveness is reduced relative to their parent aglycones(Reference Miyake, Shimoi, Kumazawa, Yamamoto, Kinae and Osawa28–Reference da Silva, Piskula, Yamamoto, Moon and Terao32).
Flavonoid-induced improvements in memory, learning and cognitive performance
There is a growing interest in the potential of phytochemicals to improve memory, learning and general cognitive ability. A recent prospective study aimed at examining flavonoid intake in relation to cognitive function and decline, has provided strong evidence that dietary flavonoid intake is associated with better cognitive evolution, i.e. the preservation of cognitive performance with ageing(Reference Letenneur, Proust-Lima, Le Gouge, Dartigues and Barberger-Gateau33). A total of 1640 subjects (aged ≥65 years) free from dementia at baseline and with reliable dietary assessment data were examined for their cognitive performance (mini-mental state examination, Benton's visual retention test, ‘Isaacs’ set test) four times over a 10-year period. After adjustment for age, gender and educational level flavonoid intake was found to be associated with significantly better cognitive performance at baseline and with a better evolution of the performance over time. In particular, subjects in the two highest quartiles of flavonoid intake (mg/d; 13·60–17·69 and 17·70–36·94) were found to have better cognitive evolution than subjects in the lowest quartile (0–10·39 mg/d), and after 10 years of follow-up subjects with the lowest flavonoid intake were found to have lost on average 2·1 points on the mini-mental state examination, whereas subjects with the highest quartile had lost only 1·2 points. Such data provides a strong indication that regular flavonoid consumption may have a positive effect on neuro-cognitive performance with ageing, although it does not provide information relating to the activity of specific flavonoid groups.
There has been much interest in the neuro-cognitive effects of soyabean isoflavones, primarily in post-menopausal women(Reference Ho, Chan, Ho, So, Sham, Zee and Woo34–Reference Kim, Xia, Li and Gewin37). The rationale behind the potential of isoflavones to exert positive effects on cognitive function is believed to lie primarily in their potential to mimic the actions and functions of oestrogens in the brain(Reference Birge38). For example, epidemiological investigations have provided evidence that post-menopausal women who undertake oestrogen-replacement therapy have a significantly lower risk for the onset of Alzheimer's disease than women who do not(Reference Henderson39). Furthermore, animal behavioural studies have shown that ovariectomy results in the development of cognitive dysfunction, which may be prevented by oestrogen replacement, suggesting that normal mammalian cognitive function is impaired by oestrogen reduction(Reference Birge38). Isoflavone supplementation (60 mg/d) has been observed to have a favourable effect on cognitive function(Reference Casini, Marelli, Papaleo, Ferrari, D'Ambrosio and Unfer40), particularly verbal memory, in post-menopausal women(Reference Kritz-Silverstein, Von, Barrett-Connor and Bressel41) and a 6-week and 12-week supplementation has been observed to have a positive effect of frontal lobe function(Reference File, Hartley, Elsabagh, Duffy and Wiseman42, Reference Zippel, Balestrini, Lomazzi and Sturani43). However, other large intervention trials have reported that dietary isoflavone supplementation (50–100 mg/d) does not improve cognitive function(Reference Ho, Chan, Ho, So, Sham, Zee and Woo34,Reference Kreijkamp-Kaspers, Kok, Grobbee, de Haan, Aleman, Lampe and van der Schouw44–Reference Kreijkamp-Kaspers, Kok, Grobbee, de Haan, Aleman and van der Schouw46). If isoflavones do possess the potential to influence human memory and cognitive performance it is likely that their mechanism of action would include their role as weak oestrogens, their ability to inhibit tyrosine kinase-dependent signal transduction and their ability to act as weak antioxidants(Reference Barnes, Boersma, Patel, Kirk, Darley-Usmar, Kim and Xu47, Reference Kim, Peterson and Barnes48).
Other flavonoid-rich foods, in particular those containing flavanols, have been observed to improve peripheral blood flow and surrogate markers of cardiovascular function in human subjects(Reference Schroeter, Heiss, Balzer, Kleinbongard, Keen, Hollenberg, Sies, Kwik-Uribe, Schmitz and Kelm49). In the context of the central nervous system brain-imaging studies in human subjects have demonstrated that the consumption of flavanol-rich cocoa may enhance cortical blood flow(Reference Francis, Head, Morris and Macdonald50–Reference Dinges52). This finding is important as increased cerebrovascular function, especially in the hippocampus (a brain region important for memory), may facilitate adult neurogenesis(Reference Gage53). Indeed, new hippocampal cells are clustered near blood vessels, proliferate in response to vascular growth factors and may influence memory(Reference Palmer, Willhoite and Gage54). As well as new neuronal growth, increases in neuronal spine density and morphology are considered vital for learning and memory(Reference Harris and Kater55). Changes in spine density, morphology and motility have been shown to occur with paradigms that induce synaptic as well as altered sensory experience and lead to alterations in synaptic connectivity and strength between neuronal partners, affecting the efficacy of synaptic communication. These events are controlled at the cellular and molecular level and are strongly correlated with memory and learning. The flavanol (–)-epicatechin, especially in combination with exercise, has been observed to enhance the retention of rat spatial memory in a water maze test(Reference van Praag, Lucero and Yeo56). This improvement in spatial memory was shown to be associated with increased angiogenesis and neuronal spine density in the dentate gyrus of the hippocampus and with the up-regulation of genes associated with learning in the hippocampus.
There is also extensive evidence that berries, in particular blueberries, are effective at reversing age-related deficits in motor function and spatial working memory(Reference Galli, Shukitt-Hale, Youdim and Joseph3,Reference Joseph, Shukitt-Hale, Denisova, Prior, Cao, Martin, Taglialatela and Bickford57–Reference Joseph, Denisova, Arendash, Gordon, Diamond, Shukitt-Hale and Morgan63). For example, the latency period to find a platform and the distance swum to a platform in a Morris water maze task are significantly reduced following blueberry supplementation(Reference Joseph, Shukitt-Hale, Denisova, Prior, Cao, Martin, Taglialatela and Bickford57, Reference Joseph, Shukitt-Hale, Denisova, Bielinski, Martin, McEwen and Bickford58). Such results may suggest favourable effects of the blueberry diet on locomotor activity in old animals(64,65). However, reductions in the time taken to make a choice may also reflect an improved memory component, where rats ‘remember’ more rapidly and thus respond quicker. Animal studies with tea(Reference Unno, Takabayashi, Kishido and Oku4, Reference Chan, Hosoda, Tsai, Yamamoto and Wang66), grape juice(Reference Shukitt-Hale, Carey, Simon, Mark and Joseph67) or flavonols such as quercetin(Reference Singh, Naidu and Kulkarni68, Reference Patil, Singh, Satyanarayan, Jain, Singh and Kulkarni69) have provided further evidence that dietary flavonoids are beneficial in reversing the course of neuronal and behavioural ageing. Although such effects have been linked with antioxidant actions, it is more likely that these effects are mediated by a modulation of neurotransmitter release(Reference Joseph, Shukitt-Hale, Denisova, Prior, Cao, Martin, Taglialatela and Bickford57, Reference Joseph, Shukitt-Hale, Denisova, Bielinski, Martin, McEwen and Bickford58), a stimulation of hippocampal neurogenesis(Reference Casadesus, Shukitt-Hale, Stellwagen, Zhu, Lee, Smith and Joseph59) and changes in neuronal signalling(Reference Joseph, Shukitt-Hale and Casadesus61, Reference Goyarzu, Malin and Lau62).
Cellular and molecular control of memory and learning
The laying down of long-term memory is usually divided into four distinct stages: learning (or acquisition of new information); consolidation; storage; retrieval (Fig. 2)(Reference Wang, Hu and Tsien70, Reference Squire and Zola-Morgan71). Studies in patients with amnaesia and experimental animals have demonstrated an important role for the hippocampus in consolidating labile short-term memory into a more stable long-term memory(Reference Squire72–Reference Alvarez and Squire74). For example, studies have indicated that the disruption of the hippocampal structure affects recent memories preferentially(Reference Anagnostaras, Maren and Fanselow75, Reference Zola-Morgan and Squire76), whereas damage in neocortex affects more remote (long-term) memories(Reference Squire and Alvarez77). Thus, the general consensus is that the hippocampus plays a time-limited role in consolidating labile new memory into more stable long-term memory, and on the completion of hippocampal-dependent consolidation, these memories are eventually stored in the cortex without major hippocampal contribution(Reference Squire72, Reference Zola-Morgan and Squire76). Within brain regions LTP is widely considered to be one of the major mechanisms by which the brain learns and maintains memories. LTP refers to a persistent increase in the chemical strength of a synapse that can last from minutes to several days, and this process is thought to contribute to synaptic plasticity and increases in synaptic strength that are thought to underlie memory formation.

Fig. 2. The processes involved in memory acquisition, consolidation and storage in the brain. Acquisition and consolidation occur primarily in the hippocampus whilst storage is in the cortex or neocortex. BDNF, brain-derived neurotrophic factor; TrkB, tropomyosin receptor kinase B; mTOR, mammalian target of rapamycin; Arc, Arc/Arg3.1.
Studies into human mental retardation syndromes have led to new insights into the molecular underpinnings of human cognitive processing, in particular into mechanisms likely to contribute to learning and memory (for review, see Weeber & Sweatt(Reference Weeber and Sweatt78), Dash et al. (Reference Dash, Moore, Kobori and Runyan79) and Tully(Reference Tully80)). Such studies have highlighted the essential role of a number of neuronal signalling pathways in bringing about changes in LTP and therefore human memory and learning. It is known that the enhancement of both short-term and long-term memory is controlled at the molecular level in neurons(Reference Carew81). Whereas short-term memory involves covalent modifications of pre-existing proteins, long-term memory requires the synthesis of new mRNA and proteins(Reference Bramham and Wells82–Reference Kelleher, Govindarajan and Tonegawa84) (Fig. 2). The rapid enhancement of the synthesis of a diverse array of neuronal proteins through such mechanisms provides the components necessary for persistent forms of LTP. Various signalling pathways have been linked with the control of de novo protein synthesis in the context of LTP and memory (Fig. 3): cAMP-dependent protein kinase (protein kinase A)(Reference Arnsten, Ramos, Birnbaum and Taylor85); protein kinase B/Akt (Akt)(Reference van der Heide, Ramakers and Smidt86); protein kinase C(Reference Alkon, Sun and Nelson87); Ca–calmodulin kinase(Reference Wei, Qiu, Liauw, Robinson, Ho, Chatila and Zhuo88); mitogen-activated protein kinase (MAPK)(Reference Sweatt89, Reference Sweatt90). All five pathways converge to signal to the cAMP-response element-binding protein (CREB), a transcription factor that binds to the promoter regions of many genes associated with memory and synaptic plasticity(Reference Impey, McCorkle, Cha-Molstad, Dwyer, Yochum, Boss, McWeeney, Dunn, Mandel and Goodman91, Reference Barco, Bailey and Kandel92) (Fig. 3).

Fig. 3. Neuronal signalling pathways involved in long-term potentiation (LTP). Five distinct signalling pathways lead to the activation of the cAMP response element-binding protein (CREB), a transcription factor important in controlling LTP at neuronal synapses. Varying stimuli activate the mitogen-activated protein kinase (MAPK) pathway (I), the calcium–calmodulin kinase (CaMK) pathway (II), the protein kinase A (PKA) pathway (III), the protein kinase B (PKB)/Akt pathway (IV) and the protein kinase C (PKC) pathway (V) in response to synapse firing. Activation of these pathways results in the activation of CREB and a variety of downstream responses, including neurotrophin expression, enhanced de novo protein synthesis, dendritic spine remodelling and ultimately stable long-term LTP. MEK, MAPK kinase; MEKK, MEK kinase; ERK, extracellular signal-regulated protein kinase; Ca chan, calcium channel; GluR, glutamate receptor; AC, adenyl cyclase; PI3 kinase, phosphoinositide 3-kinase; PDK, 3-phosphoinositide-dependent protein kinase.
The importance of CREB activation in the induction of long-lasting changes in LTP and memory are highlighted by studies that show that disruption of CREB activity specifically blocks the formation of long-term memory(Reference Bourtchuladze, Frenguelli, Blendy, Cioffi, Schutz and Silva93), whereas agents that increase the amount or activity of CREB accelerate the process(Reference Tully, Bourtchouladze, Scott and Tallman94). Furthermore, robust CREB phosphorylation and cAMP-response element-reporter gene expression are detected in cortical neurons during developmental plasticity(Reference Pham, Impey, Storm and Stryker95) and in hippocampal neurons in response to both LTP-inducing stimuli and memory-training tasks(Reference Impey, Smith, Obrietan, Donahue, Wade and Storm96, Reference Impey, Mark, Villacres, Poser, Chavkin and Storm97). Furthermore, CREB is known to be a critical transcription factor linking the actions of neurotrophins such as brain-derived neurotrophic factor (BDNF) to neuronal survival, differentiation and synaptic function(Reference Finkbeiner98, Reference Finkbeiner, Tavazoie, Maloratsky, Jacobs, Harris and Greenberg99). Consequently, the central role of CREB in these processes has led to considerable interest in identifying safe effective agents that may enhance the activity of CREB in specific regions of the brain, as they may lead to an improvement in memory(Reference Tully, Bourtchouladze, Scott and Tallman94).
Do flavonoids access the brain?
In order to understand whether flavonoids and their metabolic derivatives are capable of acting as neuromodulators it is crucial to ascertain whether they are able to enter the central nervous system. In order for flavonoids to access the brain they must first cross the blood–brain barrier (BBB). The functions of the BBB include controlling the entry of xenobiotics into the brain and maintenance of the brain's microenvironment(Reference Abbott100). In vitro and in vivo studies have indicated that the flavanones hesperetin, naringenin and their relevant in vivo metabolites, as well as some dietary anthocyanins (cyanidin-3-rutinoside and pelargonidin-3-glucoside), are able to traverse the BBB(Reference Youdim, Dobbie, Kuhnle, Proteggente, Abbott and Rice-Evans101–Reference Youdim, Qaiser, Begley, Rice-Evans and Abbott103). Furthermore, it appears that the potential for flavonoid penetration is dependent on compound lipophilicity(Reference Youdim, Dobbie, Kuhnle, Proteggente, Abbott and Rice-Evans101). Accordingly, it is plausible that the uptake of the less-polar O-methylated metabolites, such as the O-methylated epicatechin metabolites (formed in the small intestine and liver), may be greater than the parent aglycone. For the same reason, the more-polar flavonoid glucuronidated metabolites, which seem to have low BBB permeability values(Reference Youdim, Dobbie, Kuhnle, Proteggente, Abbott and Rice-Evans101), may not be able to access the brain. However, evidence exists to suggest that certain drug glucuronides may cross the BBB(Reference Aasmundstad, Morland and Paulsen104) and exert pharmacological effects(Reference Sperker, Backman and Kroemer105, Reference Kroemer and Klotz106), suggesting that there may be a specific uptake mechanism for glucuronides in vivo. Apart from the flavonoids lipophilicity, their ability to enter the brain may also be influenced by their interactions with specific efflux transporters expressed in the BBB. One such transporter is P-glycoprotein, which plays an important role in drug absorption and brain uptake(Reference Lin and Yamazaki107) and appears to be responsible for the differences between naringenin and quercetin flux into the brain in situ (Reference Youdim, Qaiser, Begley, Rice-Evans and Abbott103).
Animal feeding studies also provide evidence that flavonoids may access the brain, with the tea flavanol epigallocatechin gallate being reported to access the brain after oral administration to mice(Reference Suganuma, Okabe, Oniyama, Tada, Ito and Fujiki108). Furthermore, oral ingestion of pure epicatechin results in the detection of epicatechin glucuronide and 3′-O-methyl-epicatechin glucuronide in rat brain tissue(Reference Abd El Mohsen, Kuhnle, Rechner, Schroeter, Rose, Jenner and Rice-Evans109). Anthocyanidins have also been detected in the brain after oral administration(Reference Talavera, Felgines, Texier, Besson, Gil-Izquierdo, Lamaison and Remesy110, Reference Abd El Mohsen, Marks, Kuhnle, Moore, Debnam, Kaila, Rice-Evans and Spencer111), with several anthocyanidins being identified in different regions of the rat brain after animals were fed with blueberry(Reference Andres-Lacueva, Shukitt-Hale, Galli, Jauregui, Lamuela-Raventos and Joseph64). Such flavonoid localisation has been correlated with increased cognitive performance, suggesting a central neuroprotective role of these components. Despite their ability to access the brain, the concentrations of flavonoids and their metabolite forms accumulated in vivo (Reference Abd El Mohsen, Kuhnle, Rechner, Schroeter, Rose, Jenner and Rice-Evans109) are lower (high nm–low μm) than those recorded for small-molecule antioxidant nutrients such as ascorbic acid and α-tocopherol(Reference Halliwell, Zhao and Whiteman112). Consequently, the beneficial effects of flavonoid metabolites in the brain are unlikely to result from their ability to out-compete antioxidants such as ascorbate, which are present at higher concentrations (high μm–mm). Instead, it appears that the cellular effects of flavonoids are likely to be mediated by their interactions with specific proteins central to neuronal intracellular signalling cascades(Reference Schroeter, Boyd, Spencer, Williams, Cadenas and Rice-Evans17), such as the MAPK signalling pathway and the phosphoinositide 3-kinase (PI3K)/Akt signalling cascade.
How might flavonoids act to induce neuro-cognitive changes?
There are many ways in which dietary flavonoids may exert beneficial effects in the central nervous system. For example, they may protect neurons against oxidative stress-induced injury(Reference Vauzour, Vafeiadou, Corona, Pollard, Tzounis and Spencer113), alleviate neuroinflammation(Reference Vafeiadou, Vauzour and Spencer114) and promote synaptic plasticity. As evidence supports the localisation of flavonoids within the brain, these phytochemicals may be regarded as potential neuroprotective agents or neuromodulators. It appears highly likely that such properties are mediated by their abilities to interact with both protein and lipid kinase signalling cascades(Reference Spencer, Rice-Evans and Williams16,Reference Matter, Brown and Vlahos115–Reference Schroeter, Spencer, Rice-Evans and Williams120) rather than via their potential to act as classical antioxidants, and the concentrations of flavonoids in the brain are thought to be sufficiently high to exert pharmacological activity at receptors, kinases and transcription factors. Presently, the precise sites of action are unknown, although it is likely that their activity depends on their ability to: bind to ATP sites on enzymes and receptors; modulate the activity of kinases directly, i.e. MAPK kinase kinase, MAPK kinase or MAPK; affect the function of important phosphatases, which act in opposition to kinases; preserve Ca2+ homeostasis, thereby preventing Ca2+-dependent activation of kinases in neurons; modulate signalling cascades lying downstream of kinases, i.e. transcription factor activation and binding to promoter sequences(Reference Spencer121).
Flavonoids have the potential to bind to the ATP-binding sites of a large number of proteins(Reference Conseil, Baubichon-Cortay, Dayan, Jault, Barron and Di Pietro122) including mitochondrial ATPase(Reference Di Pietro, Godinot, Bouillant and Gautheron123), Ca plasma-membrane ATPase(Reference Barzilai and Rahamimoff124), protein kinase A(Reference Revuelta, Cantabrana and Hidalgo125), protein kinase C(Reference Gamet-Payrastre, Manenti, Gratacap, Tulliez, Chap and Payrastre118,Reference Lee and Lin126–Reference Rosenblat, Belinky, Vaya, Levy, Hayek, Coleman, Merchav and Aviram129) and topoisomerase(Reference Boege, Straub, Kehr, Boesenberg, Christiansen, Andersen, Jakob and Kohrle130). In addition, interactions with the benzodiazepine-binding sites of GABAA receptors and with adenosine receptors(Reference Medina, Viola, Wolfman, Marder, Wasowski, Calvo and Paladini131, Reference Dekermendjian, Kahnberg, Witt, Sterner, Nielsen and Liljefors132) have been reported. For example, the stilbene resveratrol and the citrus flavanones hesperetin and naringenin have been reported to have inhibitory activity at a number of protein kinases(Reference Fischer and Lane133–Reference So, Guthrie, Chambers, Moussa and Carroll135). This inhibition is mediated via the binding of the polyphenols to the ATP-binding site, presumably causing three-dimensional structural changes in the kinase leading to its inactivity. They may also interact directly with mitochondria, for example by modulating the mitochondrial transition pore, which controls cytochrome c release during apoptosis(Reference Green and Reed136, Reference Tatton and Olanow137), or by modulating other mitochondrial-associated pro-apoptotic factors such as DIABLO/smac(Reference Goyal138, Reference Srinivasula, Hegde and Saleh139). Potential interactions with the mitochondrial transition pore are especially interesting, as the transition pore possesses a benzodiazepine-binding site where flavonoids may bind(Reference Medina, Viola, Wolfman, Marder, Wasowski, Calvo and Paladini131, Reference Dekermendjian, Kahnberg, Witt, Sterner, Nielsen and Liljefors132) and influence pore opening and cytochrome c release during apoptosis.
Interactions of flavonoids within the extracellular signal-regulated protein kinase/cAMP-response element-binding protein signalling pathway
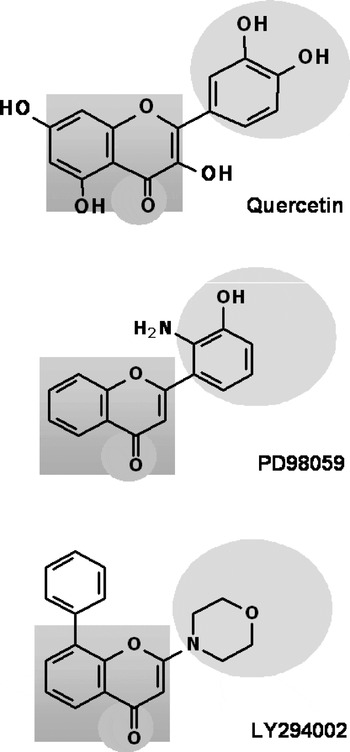
Previous studies have suggested that phytochemicals, especially flavonoids, may exert cellular effects via direct modulation of protein and lipid kinase signalling pathways(Reference Williams, Spencer and Rice-Evans15). Interactions within the MAPK pathway are thought to be central to mediating the cellular effects of flavonoids such as those found in berries, tea and cocoa(Reference Spencer, Rice-Evans and Williams16, Reference Schroeter, Spencer, Rice-Evans and Williams120). For example, the flavanol (–)-epicatechin induces both extracellular signal-regulated protein kinase (ERK) 1/2 and CREB activation in cortical neurons and subsequently increases CREB-regulated gene expression(Reference Schroeter, Bahia, Spencer, Sheppard, Rattray, Rice-Evans and Williams140). Furthermore, another flavonoid, fisetin, has been shown to improve LTP and memory through a CREB/ERK mechanism(Reference Maher, Akaishi and Abe141) and nanomolar concentrations of quercetin have also been observed to enhance CREB activation in neurons(Reference Spencer, Rice-Evans and Williams16). Thus, one potential mechanism of action of flavonoids in modulating neuronal function, LTP and synaptic plasticity may proceed via signalling through CREB. In support of this possibility, other flavonoids have also been shown to influence the ERK pathway, with the citrus flavanone hesperetin being capable of activating ERK1/2 signalling in cortical neurons(Reference Vauzour, Vafeiadou, Rice-Evans, Williams and Spencer142) and flavanols such as epigallocatechin gallate restoring both protein kinase C and ERK1/2 activities in 6-hydroxydopamine-treated and serum-deprived neurons(Reference Levites, Amit, Youdim and Mandel143, Reference Reznichenko, Amit, Youdim and Mandel144). Furthermore, this ability to activate the ERK pathway is not restricted to neurons and has also been observed in fibroblasts exposed to low concentrations of epicatechin(Reference Pollard, Whiteman and Spencer145). However, although the majority of investigations have centred on the potential of flavonoids to modulate the phosphorylation state of ERK1/2(Reference Spencer, Rice-Evans and Williams16, Reference Schroeter, Boyd, Spencer, Williams, Cadenas and Rice-Evans17, Reference Schroeter, Spencer, Rice-Evans and Williams120), it is more likely that their actions on this MAPK isoform result from effects on upstream kinases, such as MAPK kinases 1 and 2, and potentially membrane receptors(Reference Schroeter, Boyd, Spencer, Williams, Cadenas and Rice-Evans17) (Fig. 3). This possibility appears likely as flavonoids have close structural homology to specific inhibitors of MAPK kinase 1, such as PD98059 (2′-amino-3′-methoxyflavone; Fig. 4). Overall, their effects on the ERK pathway are likely to be related to their ability to exert high-affinity receptor-agonist-like actions at low concentrations and direct enzyme inhibition at higher concentrations(Reference Agullo, Gamet-Payrastre, Manenti, Viala, Remesy, Chap and Payrastre117, Reference Walker, Pacold, Perisic, Stephens, Hawkins, Wymann and Williams146). Receptors reported to act as flavonoid-binding sites that are present in cortical neurons are adenosine(Reference Jacobson, Moro, Manthey, West and Ji147) and GABAA receptors(Reference Johnston148, Reference Adachi, Tomonaga, Tachibana, Denbow and Furuse149). However, a specific plasma-membrane binding site for polyphenols has recently been described in rat brain(Reference Han, Bastianetto, Dumont and Quirion150). In addition, monomeric and dimeric flavanols show nanomolar affinity and efficacy at testosterone receptors(Reference Nifli, Bosson-Kouame, Papadopoulou, Kogia, Kampa, Castagnino, Stournaras, Vercauteren and Castanas151) and resveratrol rapidly activates ERK signalling through α and β oestrogen receptors(Reference Klinge, Blankenship, Risinger, Bhatnagar, Noisin, Sumanasekera, Zhao, Brey and Keynton152). Collectively, these findings raise the possibility that flavonoids may act on the ERK pathway by acting through steroid-like receptors in neurons to modulate ERK and CREB-mediated gene expression.

Fig. 4. The structure of mitogen-activated protein kinase kinase inhibitor PD98059 and the phosphoinositide 3-kinase inhibitor LY294002 have close structural homology to that of flavonoids. LY294002 and quercetin both fit into the ATP-binding pocket of the phosphoinositide 3-kinase, inhibiting its activity. It appears that the number and substitution of hydroxyl groups on the flavonoid ring B and the extent of unsaturation of the C-2–C-3 bond are important determinants of their activity. Such inhibitory actions have been proposed as potential mechanisms by which flavonoids act to modulate neuronal function.
If flavonoids are able to promote the activation of neuronal CREB, they may be capable of enhancing the expression of a number of genes that contain cAMP-response element sequences in their promoter regions, including a number of neurotrophins(Reference Conkright, Guzman, Flechner, Su, Hogenesch and Montminy153). Particular emphasis has been given to the regulation of BDNF(Reference Tao, Finkbeiner, Arnold, Shaywitz and Greenberg154, Reference Shieh, Hu, Bobb, Timmusk and Ghosh155), which has been implicated in synaptic plasticity and long-term memory(Reference Bramham and Messaoudi156) and is robustly induced in hippocampal neurons on synaptic stimulation(Reference Patterson, Grover, Schwartzkroin and Bothwell157). BDNF belongs to the neurotrophin family of growth factors and affects the survival and function of neurons in the central nervous system. Its secretion from neurons is under activity-dependent control and is crucial for the formation of appropriate synaptic connections during development and for learning and memory in adults(Reference Thomas and Davies158). Decreases in BDNF and pro-BDNF have been reported in Alzheimer's disease(Reference Peng, Wuu, Mufson and Fahnestock159, Reference Michalski and Fahnestock160) and the importance of pro-BDNF has been emphasised by the finding that a polymorphism that replaces valine for methionine at position 66 of the pro-domain is associated with memory defects and abnormal hippocampal function in human subjects(Reference Egan, Kojima and Callicott161). In addition, genetic(Reference Linnarsson, Bjorklund and Ernfors162) as well as pharmacological inhibition(Reference Mu, Li, Yao and Zhou163) of BDNF or its receptor tropomyosin receptor kinase B(Reference Minichiello, Korte, Wolfer, Kuhn, Unsicker, Cestari, Rossi-Arnaud, Lipp, Bonhoeffer and Klein164) impairs learning and memory. On the other hand, agents that increase BDNF levels lead to improvements in spatial working memory, in part through the regulation of protein translation via the mammalian target of rapamycin (mTOR) signalling pathway(Reference Wullschleger, Loewith and Hall165) (Fig. 5). It has recently been shown that a 3–12-week supplementation of old rats with a 20 g blueberry/kg diet leads to improvement in spatial working memory, which is correlated with an activation of CREB and increases in both pro- and mature levels of BDNF in the hippocampus (CM Williams, MM Abd El Mohsen and JPE Spencer, unpublished results).

Fig. 5. Formation of stable long-term potentiation (LTP) at synapses. Increased expression and release of brain-derived neurotrophic factor (BDNF) from the synapse through enhanced cAMP-response element-binding protein (CREB) activation (1). BDNF binds to pre-and post-synaptic tropomyosin receptor kinase B (TrkB) receptors (2), triggering glutamate release and phosphoinositide 3-kinase (PI3K)/mammalian target of rapamycin (mTOR) signalling and Arc/Arg3.1 (Arc) synthesis (3). Sustained activation of mTOR leads to enhanced translational efficiency whilst Arc, in association with Cofilin, triggers F-actin expansion and synapse growth (mushroom synapse; 4). Akt, protein kinase B/Akt; PKC, PKA, protein kinase A and C respectively; CaMK, calcium–calmodulin kinase; ERK, extracellular signal-regulated protein kinase.
Interactions of flavonoids within the phosphoinositide 3-kinase/Akt signalling pathway
Flavonoids have long been known to inhibit PI3K and Akt via direct interactions with its ATP-binding site. Indeed, a number of studies have demonstrated that the structure of flavonoids determines whether or not they act as potent inhibitors of PI3K(Reference Agullo, Gamet-Payrastre, Manenti, Viala, Remesy, Chap and Payrastre117, Reference Ferriola, Cody and Middleton166). One of the most selective PI3K inhibitors available, LY294002 (Fig. 4), was modelled on the structure of quercetin(Reference Matter, Brown and Vlahos115, Reference Vlahos, Matter, Hui and Brown116). LY294002 and quercetin fit into the ATP-binding pocket of the enzyme although with surprisingly different orientations(Reference Walker, Pacold, Perisic, Stephens, Hawkins, Wymann and Williams146). It appears that the number and substitution of hydroxyl groups on the B-ring and the extent of unsaturation of the C-2–C-3 bond are important determinants of this particular bioactivity. Interestingly, in this context quercetin and some of its in vivo metabolites inhibit pro-survival Akt signalling pathways(Reference Spencer, Rice-Evans and Williams16) by a mechanism of action consistent with quercetin and its metabolites acting at and inhibiting PI3K activity(Reference Matter, Brown and Vlahos115). However, other flavonoids such as the citrus flavanone hesperetin cause the activation of Akt and the inhibition of pro-apoptotic proteins such as apoptosis signal-regulating kinase 1, Bad, caspase-9 and caspase-3 in cortical neurons(Reference Vauzour, Vafeiadou, Rice-Evans, Williams and Spencer142). Thus, flavanones, and other flavonoids, may be capable of exerting beneficial effects in neurons via signalling through Akt and may also have the potential to activate CREB through activation of this pathway (Fig. 3).
At neuronal synapses flavonoid-induced activation of CREB and enhancement of BDNF expression in neurons would be expected to initiate the activation of the PI3K/Akt signalling pathway via the binding of BDNF to pre- or post-synaptic tropomyosin receptor kinase B receptors (Fig. 5). These events trigger the activation of the mTOR pathway and the increased translation of specific mRNA subpopulations(Reference Schratt, Nigh, Chen, Hu and Greenberg167), including the activity-regulated cytoskeletal-associated protein termed Arc/Arg3.1. Arc/Arg3.1 is known to be important in LTP and has been proposed to be under regulatory control of both BDNF(Reference Yin, Edelman and Vanderklish168) and the ERK signalling pathway(Reference Waltereit, Dammermann, Wulff, Scafidi, Staubli, Kauselmann, Bundman and Kuhl169) (Fig. 5). In addition to ERK and CREB activation, blueberry supplementation for 12 weeks has also been observed to lead to an activation of mTOR and the increased expression of hippocampal Arc/Arg3.1 (CM Williams, MM Abd El Mohsen and JPE Spencer, unpublished results). The sustained synthesis of Arc/Arg3.1 during a protracted time-window is necessary to consolidate LTP, whilst translation of pre-existing Arc/Arg3.1 mRNA contributes to early LTP expression and translation of new Arc/Arg3.1 mRNA mediates consolidation(Reference Soule, Messaoudi and Bramham170). Increased Arc/Arg3.1 expression may facilitate changes in synaptic strength and the induction of morphological changes such as that observed when small dendritic spines are converted into large mushroom-shaped spines through a mechanism dependent on actin polymerisation(Reference Lyford, Yamagata, Kaufmann, Barnes, Sanders, Copeland, Gilbert, Jenkins, Lanahan and Worley171) (Fig. 5). Whether flavonoids are capable of promoting changes in neuronal morphology in vivo is currently unclear, although studies have indicated that effects of flavonoid effects on neuronal morphology are possible(Reference van Praag, Lucero and Yeo56) and that certain flavonoids can influence neuronal dentrite outgrowth in vitro (Reference Reznichenko, Amit, Youdim and Mandel172). In addition, the known ability of flavonoids to activate signalling cascades upstream of mTOR and Arc/Arg3.1, notably ERK, CREB and BDNF, strengthens the concept that they are also capable of inducing changes in neuronal morphology that underlie improvements in memory, learning and cognitive performance in mammalian species, including man.
Emerging evidence suggests that dietary phytochemicals, in particular flavonoids, may exert beneficial effects in the central nervous system by protecting neurons against stress-induced injury, by suppressing neuroinflammation and by promoting LTP and synaptic plasticity. Such effects, in particular the latter two, are likely to underpin their observed beneficial effects on human memory and neuro-cognitive performance. There is strong evidence that such beneficial properties are mediated by their ability to interact with a number of neuronal protein and lipid kinase signalling cascades known to be crucial in determining LTP and hence the acquisition, consolidation and storage of human memory. Such pathways include the MAPK signalling cascade, in particular the ERK1/2 pathway, the protein kinase A pathway and the Ca–calmodulin kinase cascade. The activation of these pathways along with the activation of the transcription factor CREB is known to be required during memory acquisition and consolidation, and agents capable of inducing pathways leading to CREB activation have the potential to enhance both short-term and long-term memory.
In contrast to short-term memory, the storage of long-term memory requires the formation of stable LTP at synapses. This process is known to require an enhancement of synaptic mRNA translation and de novo synthesis of proteins such as F-actin and synaptic membrane receptors, which lead to an increase in dendritic spine density and membrane receptor density respectively. Flavonoids may trigger all these events via their ability to activate CREB and CREB-induced gene expression. In doing so they may increase the expression of neurotrophins, such as BDNF, which initiate the de novo synthesis of many proteins associated with LTP via the activation of mTOR signalling. Ultimately, these events lead to synapse growth, an increase in dendritic spine density and increased membrane receptor density, all factors known to be essential for efficient LTP, synaptic plasticity and ultimately the storage of long-term memory.
Acknowledgements
J.P.E.S. is funded by the Biotechnology and Biological Sciences Research Council (BB/C518222/1) and the Medical Research Council (G0400278/NI02).