Phenolic acids are secondary plant metabolites widely spread throughout the plant kingdom(Reference Pérez-Jiménez, Neveu and Vos1). Alongside the better-known flavonoids, they form one of the two major classes of dietary polyphenols. Chemically, phenolic acids are hydroxylated derivatives of benzoic, cinnamic, phenylacetic and phenylpropanoic acids. Hydroxycinnamic acids occur most frequently as simple esters with hydroxy carboxylic acids or sugars, whereas hydroxybenzoic acids are present mainly in glycosylated forms(Reference Manach, Scalbert and Morand2). Most often, only low amounts of phenolic acids in the free form (aglycones) are found in foods. In nature, hundreds of phenolic acids have been identified, although only ninety (thirty-six aglycones) were found to be consumed by a French cohort(Reference Pérez-Jiménez, Fezeu and Touvier3). They are most abundant in coffee, tea, red wine and some vegetables and fruits, especially berries(Reference Pérez-Jiménez, Neveu and Vos1).
Recent interest in phenolic acids stems from their potential protective role against oxidative stress, inflammation, diabetes and cancer in experimental studies(Reference Nagasaka, Chotimarkorn and Shafiqul4–Reference Thom7). However, very few epidemiological studies have evaluated the effects of dietary phenolic acids upon chronic diseases due to a lack of food composition data on these compounds(Reference Arts and Hollman8). Phenol-Explorer (www.phenol-explorer.eu) is a comprehensive food composition database (FCDB) on polyphenols, including phenolic acids, that has been available to the scientific community since 2009(Reference Neveu, Pérez-Jiménez and Vos9). This FCDB enables researchers to more accurately assess the intake of phenols, their food sources and their potential effects on health, taking into account both the chemical structure of phenolic acids and their combination in glycosidic or ester forms.
To our knowledge, only three groups have assessed the dietary intake of phenolic acids. Using their own FCDB, German(Reference Radtke, Linseisen and Wolfram10) and Finnish(Reference Ovaskainen, Torronen and Koponen11) studies estimated mean daily intakes of 222 and 641 mg of dietary phenolic acids (expressed as aglycones), respectively. Using Phenol-Explorer, the French cohort SUpplémentation en VItamines et Minéraux AntioXydants (SU.VI.MAX) estimated mean intakes of 599 mg/d (316 mg as aglycones/d) and 41 mg/d (40 mg as aglycones/d) for hydroxycinnamic acids and hydroxybenzoic acids, respectively(Reference Pérez-Jiménez, Fezeu and Touvier3). It is difficult to determine whether these differences are due to different dietary and lifestyle patterns or to differences in the FCDB used. Therefore, the aim of the present study was to estimate the intake of phenolic acids and their main food sources across the ten European countries participating in the European Prospective Investigation into Cancer and Nutrition (EPIC) study and across population subgroups using the same FCDB and dietary assessment tools.
Materials and methods
Study population
The EPIC is an ongoing multi-centre prospective cohort study involving twenty-three centres in ten European countries: Greece; Spain; Italy; France; Germany; The Netherlands; UK; Denmark; Sweden; Norway(Reference Riboli, Hunt and Slimani12). Over half a million subjects (29·5 % men) aged 20–85 years were enrolled between 1992 and 2000. Participants were mostly recruited from the general population within defined geographical areas, with some exceptions: women who were members of a health insurance scheme for state school employees (France); women attending breast cancer screening (Utrecht, The Netherlands and Naples, Italy); mostly blood donors (some centres in Italy and Spain); mainly vegetarians (the ‘health conscious’ cohort in Oxford, UK). For the purpose of dietary analyses, the twenty-three administrative EPIC centres were redefined into twenty-seven geographical areas(Reference Slimani, Kaaks and Ferrari13), of which nineteen included both male and female participants and eight recruited women only.
Data used in the present study were derived from the EPIC calibration study, in which a 24 h recall was administered. The cohort comprises an 8 % (n 36 994) age- and sex-stratified random sample, weighted according to the number of cancer cases expected over 10 years of follow-up(Reference Slimani, Kaaks and Ferrari13). After 957 exclusions due to an age restriction to 35–74 years or incomplete dietary information, 36 037 subjects were included in the present analysis. Approval for the study was obtained from ethical review boards of the International Agency for Research on Cancer (IARC) and from all local participating institutions. All participants provided written informed consent.
Dietary and lifestyle information
Dietary information was collected through a single 24 h recall using specialised software (EPIC-Soft)(Reference Slimani, Deharveng and Charrondiere14, Reference Slimani, Ferrari and Ocke15), which was developed to ensure standardisation of the interviews. All centres administered the interviews face to face, except in Norway where telephone interviews were conducted(Reference Brustad, Skeie and Braaten16).
Data on other lifestyle factors, including educational level, anthropometry, physical activity (combining both occupation and leisure-time activities) and smoking history, were collected at baseline through standardised questionnaires and clinical examinations(Reference Riboli, Hunt and Slimani12, Reference Slimani, Kaaks and Ferrari13, Reference Wareham, Jakes and Rennie17). Data on age, as well as on body weight and height, were mostly self-reported by the participants during the 24 h recall interview. The mean time interval between these baseline measures and the 24 h recall interview varied by country, from 1 d to 3 years later(Reference Slimani, Kaaks and Ferrari13).
Food composition database on phenolic acids
A food composition table was generated based on the mean content of individual phenolic acids in the Phenol-Explorer database and on the 24 h recall occurrences. Phenol-Explorer provides data on 502 polyphenol compounds in 452 plant-based foods(Reference Neveu, Pérez-Jiménez and Vos9), including data on 109 phenolic acids, of which forty-five are aglycones. They are distributed across four different subclasses: hydroxybenzoic acids (n 16); hydroxycinnamic acids (n 21); hydroxyphenylacetic acids (n 5); hydroxyphenylpropanoic acids (n 3). Total phenolic acid intake was calculated as the sum of the total intake of individual phenolic compounds using Phenol-Explorer food content values determined by chromatography without hydrolysis(Reference Neveu, Pérez-Jiménez and Vos9). Intakes were expressed as aglycone equivalents (n 45) after the conversion of phenolic acid glycoside and ester contents into aglycone contents on the basis of their respective molecular weights. For phenolic acids linked to the food matrix, food content values obtained by chromatography after hydrolysis(Reference Neveu, Pérez-Jiménez and Vos9) were used; this applied to hydroxycinnamic acids in cereals, legumes and olives. In addition, when data obtained by chromatography without hydrolysis were unavailable, phenolic acid contents obtained by chromatography after hydrolysis were used.
Phenolic acid composition data for a small number of foods reported in the 24 h recall were absent from the Phenol-Explorer database and therefore obtained from scientific literature. These food items included honey, roasted chicory powder, orange and lemon peels, carob flour, mushrooms, okra seed pod, sweet potato, jujube, vanilla pod, seaweed and rose hip. A few more food items reported in the 24 h recall and expected to contain phenolic compounds (coconut, some liquors and spirits, hop, grapevine leaves, orgeat, pumpkin flower and some minor vegetable oils) were not available in the Phenol-Explorer database. However, the frequency of consumption or the polyphenol content of these foods was low and their contribution to phenolic acid intake was therefore considered minimal. Caffeic, ferulic, sinapic and p-coumaric acids in orange fruit were obtained by extrapolation from orange juice data. For some concentrated foods (e.g. tomato purée, tomato paste), dried foods (e.g. dried fruit, sun-dried tomato) and dehydrated foods (e.g. garlic and onion in powder) reported in the 24 h recall but not included in Phenol-Explorer, yield factors were applied to adjust for the gain or loss of moisture during food processing or cooking. The effect of cooking on phenolic acid content was not considered due to the paucity of data available. To estimate the phenolic acid contents of generic food items reported in the 24 h recall (e.g. fruit not specified), the profile of consumption of individual foods in that group and in each country was applied. Complex foods (e.g. cakes, sauces) were calculated as recipes taking into account the individual ingredients and their corresponding proportions as estimated from data available on the Internet.
Statistical analyses
Dietary phenolic acid intakes were calculated using generalised linear models and presented as means with their standard errors stratified by sex and the redefined centres, which are ordered geographically from south to north. These models were adjusted for age (continuous variable) and weighted by season and weekday of the 24 h recall to control for different distributions of participants across seasons and days of the recall. The contribution of each individual and subgroup of phenolic acids to the total intake was calculated as a percentage for each of the three European regions (south: all centres in Greece, Spain, Italy and the south of France; central: all centres in the north-east and north-west of France, Germany, The Netherlands and the UK; north: all centres in Denmark, Sweden and Norway). The contribution of each food group to the total phenolic acid intake by European region was also computed as a percentage.
Differences in phenolic acid intakes were compared using general linear models according to the categories of sex, age (35–44, 45–54, 55–64 and 65–74 years), BMI ( < 25, 25–30 and >30 kg/m2), European regions (south, central and north), educational level (none, primary completed, technical/professional, secondary school and university degree), smoking status (never smoker, current smoker and former smoker) and level of physical activity (inactive, moderately inactive, moderately active and active). All these models were adjusted for sex (categorical), age (years, continuous), centre (categorical), BMI (kg/m2, continuous) and energy intake (kJ/d, continuous), and weighted by season and weekday. P values < 0·05 (two-tailed) were considered as significant. All analyses were conducted using SPSS Statistics software (version 19.0; SPSS, Inc.).
Results
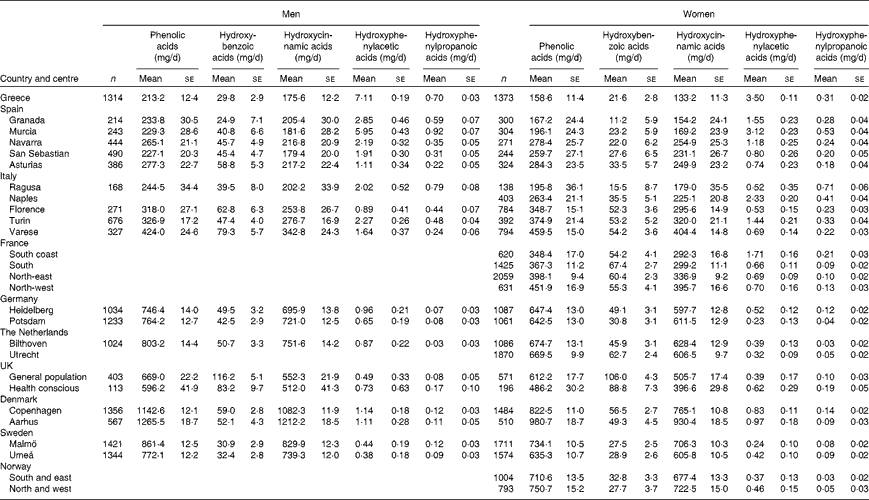
Table 1 shows the centre-specific daily mean intakes of total and classes of phenolic acids, stratified by sex, adjusted for age and weighted for season and weekday of the 24 h recall. A south-to-north gradient in the mean intake of total phenolic acids and hydroxycinnamic acids was observed among the EPIC centres in both men and women. The highest total phenolic acid intake was observed in Aarhus, Denmark (1265·5 and 980·7 mg/d in men and women, respectively), whereas the lowest intake was in Greece (213·2 and 158·6 mg/d in men and women, respectively).
Table 1 Adjusted* mean daily intakes of total and classes of phenolic acids by sex and centre ordered from south to north (Mean values with their standard errors)

* Adjusted for age and weighted by season and day of recall.
The main phenolic acid contributors were hydroxycinnamic acids (ranging from 84·6 % in the south region to 95·3 % in the north region), followed by hydroxybenzoic acids (ranging from 4·6 % in the north region to 14·4 % in the south region), hydroxyphenylacetic acids (ranging from 0·1 % in both central and north regions to 0·8 % in the south region) and, finally, hydroxyphenylpropanoic acids ( ≤ 0·1 % in all regions). The main hydroxybenzoic acid contributors were gallic acid, ellagic acid and 4-hydroxybenzoic acid. For hydroxycinnamic acids, the main contributors were caffeic acid and ferulic acid. Homovanillic and 4-hydroxyphenylacetic acids were the most abundant hydroxyphenylacetic acids, while the three hydroxyphenylpropanoic acid aglycones contributed in similar proportions.
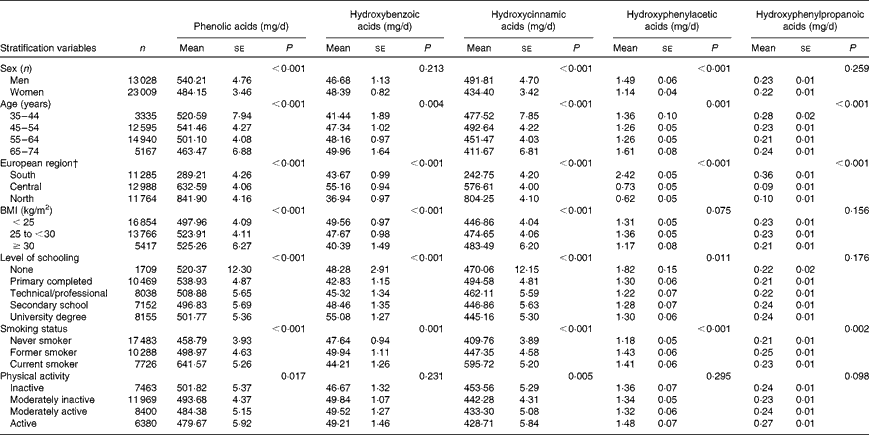
Table 2 shows the evaluation of the relationship between sociodemographic, lifestyle and anthropometric characteristics and phenolic acid intakes adjusted for age, sex, centre, energy and BMI and weighted for season and weekday. Men had a statistically significant higher intake of total phenolic acids (540·2 v. 484·2 mg/d), hydroxycinnamic acids (491·8 v. 434·4 mg/d) and hydroxyphenylacetic acids (1·49 v. 1·14 mg/d) than women. There were no statistically significant differences in total hydroxybenzoic acid and hydroxyphenylpropanoic acid intakes between sexes. Participants who were between 45 and 54 years had the highest intakes of total phenolic acids. A geographical gradient of increasing total phenolic acids, and hydroxycinnamic acids from south to north, was observed. The opposite geographical gradient was found for hydroxyphenylacetic acids and hydroxyphenylpropanoic acids, whereas hydroxybenzoic acid intake was highest in the central region. Physically inactive individuals, current smokers, obese participants and those with a low level of education had higher intakes of phenolic acids and hydroxycinnamic acids. However, for hydroxybenzoic acids, the opposite association was observed with BMI and level of education.
Table 2 Adjusted* mean daily intakes of total and classes of phenolic acids by sex and selected characteristics (Mean values with their standard errors)

* Adjusted for sex, age, energy intake and BMI and weighted by season and day of recall. P values are for overall differences in mean consumption by the general linear model among the sociodemographic, lifestyle and anthropometric subgroups.
† European region: south (all centres in Greece, Spain, Italy and the south of France); central (all centres in the north-east and north-west of France, Germany, The Netherlands and the UK); north (all centres in Denmark, Sweden and Norway).
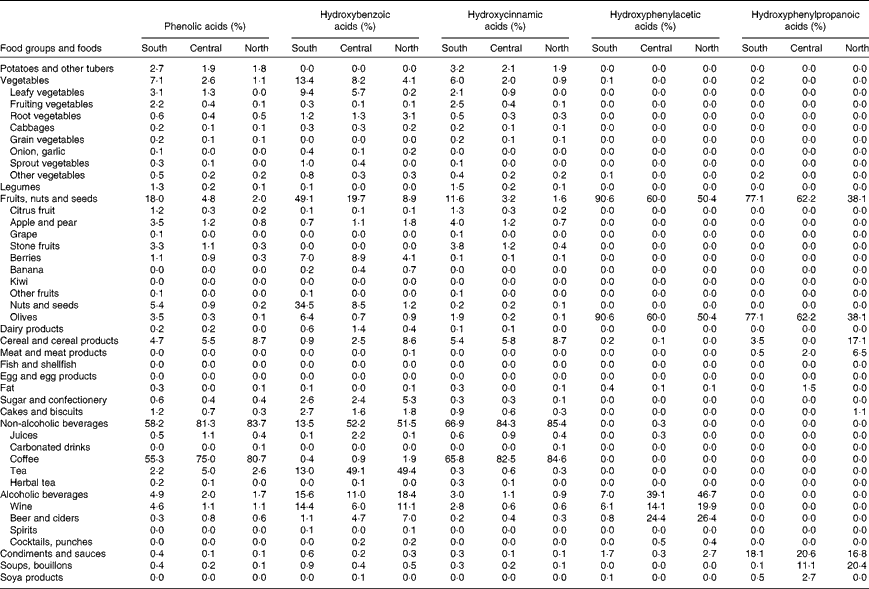
The main food sources of phenolic acid intake by European region were also studied (Table 3). Coffee was the most abundant food source of phenolic acids in all regions, with contributions ranging from 55 to 81 %. Other major food sources were fruits, nuts and seeds, some vegetables, and cereal and cereal products. For hydroxycinnamic acids, the main contributor was coffee, accounting for 67, 84 and 85 % in the south, central and north European regions, respectively. For hydroxybenzoic acids, the main food sources were tea in the central and north European regions, while in the south region, they were nuts and seeds. Other important contributors were wine, leafy vegetables and berries. For hydroxyphenylacetic acids, the major food sources were olives, particularly in the south European region, beer and ciders, and wines. The most abundant sources of hydroxyphenylpropanoic acids were olives and some condiments and sauces.
Table 3 Major food sources of total and classes of dietary phenolic acids in the south, central and north European regions*†

* Values are percentages derived from models adjusted for centre, age and sex, and weighted by season and day of recall.
† European region: south (all centres in Greece, Spain, Italy and the south of France); central (all centres in the north-east and north-west of France, Germany, The Netherlands and the UK); north (all centres in Denmark, Sweden and Norway).
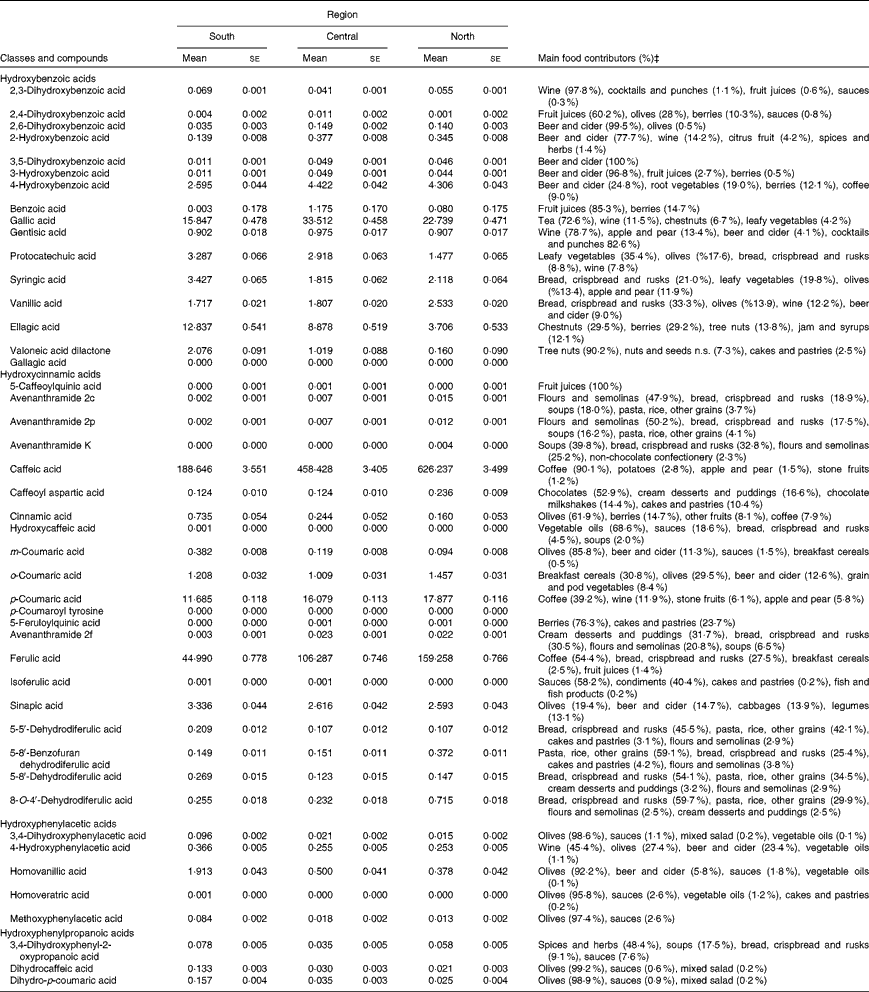
Table 4 shows the mean intakes of individual phenolic acids and their main food sources by European region, adjusted for sex and age and weighted by season and weekday. The most frequently consumed phenolic acids were caffeic acid (188·6–626·2 mg/d), ferulic acid (45·0–159·3 mg/d) and p-coumaric (11·7–17·9 mg/d) from coffee, gallic acid (15·8–33·5 mg/d) from tea and ellagic acid (3·7–12·8 mg/d) from nuts and berries.
Table 4 Adjusted* intakes of individual phenolic acids (mg/d) in the European Prospective Investigation into Cancer and Nutrition cohort and their main food sources by European region† (Mean values with their standard errors)

n.s., Not specified.
* Adjusted for sex and age, and weighted by season and day of recall.
† European region: south (all centres in Greece, Spain, Italy and the south of France); central (all centres in the north-east and north-west of France, Germany, The Netherlands and the UK); north (all centres in Denmark, Sweden and Norway).
‡ Four main food sources for each phenolic acid; a lower number indicates the absence of other food sources.
Discussion
Estimating phenolic acid intake is the first step towards documenting the protective effects of these compounds against the risk of chronic diseases in epidemiological studies. This is the first study to assess the intake of phenolic acids across European countries. Moreover, the present study also shows the main food sources and associated factors of phenolic acid intakes using the same FCDB (Phenol-Explorer) and a common dietary assessment method (24 h recall).
Intake of total phenolic acids was the greatest in northern European countries, particularly Denmark, Sweden and Norway. In these countries, mean intake after adjustment was as much as five times higher than that of the lowest intake (Greece). Most of the phenolic acid intake in all regions was from hydroxycinnamic acids, in agreement with previous local studies(Reference Pérez-Jiménez, Fezeu and Touvier3, Reference Radtke, Linseisen and Wolfram10, Reference Ovaskainen, Torronen and Koponen11). The principal source of these in all regions was coffee, which contributes high levels of caffeic acid derived from as much as 150 mg chlorogenic acids/100 ml(Reference Pérez-Jiménez, Neveu and Vos18). The high phenolic acid intake in Scandinavian and northern European countries is largely a consequence of high coffee consumption(Reference Michaud, Gallo and Schlehofer19, Reference Slimani, Fahey and Welch20). Southern European countries consume considerably less coffee, despite the apparent importance of coffee in Mediterranean culture. Ferulic acid, to a lesser extent, also contributes clearly to this difference in phenolic acid intake, with high intakes in the northern countries. As well as being present in coffee, this hydroxycinnamate is found complexed with hemicelluloses in high-fibre foods, and consequently, other important dietary sources are wheat bran, whole-grain flour and cereals(Reference Kroon, Faulds and Ryden21). Therefore, another contributory factor could be the greater consumption of whole-grain bread, crisp bread and breakfast cereals in northern European countries than in southern Europe(Reference Wirfalt, McTaggart and Pala22), which often contain a relatively high proportion of wheat bran. The third most consumed hydroxycinnamate was p-coumaric acid, primarily provided by fruits, nuts and spices, although its intake was minor in relation to that of caffeic and ferulic acids.
Even in southern European countries, hydroxycinnamic acids were by far the greatest contributor to the total phenolic acid intake, although the percentage contribution tended to be lower than that in northern Europe, due to lower coffee consumption(Reference Michaud, Gallo and Schlehofer19). Hydroxybenzoic acids, whose principal dietary sources are tea, wine and rosaceous fruits, constituted 14·4 % of total phenolic acids in the southern region, yet the contribution of nuts, seeds and fruit vegetables to total hydroxybenzoic acids was the greatest here. Intake of hydroxyphenylacetic and hydroxyphenylpropanoic acids was higher in the southern region than in the northern and central regions. Hydrophenylacetic acids in the southern region were represented mainly by homovanillic acid (77·8 %). This is found primarily in olives and olive oils, whose consumption is more frequent in southern European diets. Hydroxyphenylpropanoic acids were the least consumed phenolic acids in all regions, although significantly more were consumed in the southern region than in the northern and central regions. Again, this difference may be attributed to a greater consumption of olives and olive oils in the southern region.
Men from the EPIC cohort had higher phenolic acid intakes overall than women after adjustment for age, centre, energy and BMI, in agreement with the Finnish(Reference Ovaskainen, Torronen and Koponen11), but inconsistent with the findings of the German study(Reference Radtke, Linseisen and Wolfram10). A greater intake of hydroxycinnamic acids was largely responsible for the higher phenolic acid intake among men, which reflects a greater coffee consumption overall in European men than in women. Phenolic acid and hydroxycinnamic acid intakes were also related to health-detrimental lifestyle characteristics. Lack of exercise, smoking and obesity, as well as a low level of education, were linked to higher intakes among the study subjects. This is consistent with previous findings where coffee consumption was strongly associated with smoking and a less health-conscious lifestyle(Reference van Dam and Feskens23). In sharp contrast, subjects who had consumed greater amounts of hydroxybenzoic acids (through main dietary sources such as tea, wine and fruit) tended to exercise more, smoke less and avoid obesity.
Over a decade ago, it was proposed that phenolic acids contributed approximately to one-third of total polyphenol intake(Reference Scalbert and Williamson24), with flavonoids accounting for the remaining two-thirds. This was expected to vary considerably as a function of coffee consumption, with heavy coffee consumers ingesting a greater proportion of polyphenols as phenolic acids. As detailed databases on polyphenol composition were not available at this time, this hypothesis was generated by roughly estimating the flavonoid and phenolic acid contents of typical diets, based on the polyphenol compositions of commonly consumed food and beverage items. In the present study, the mean adjusted phenolic acid intake across all European countries was 512 mg/d. A recent study on the EPIC cohort, using Phenol-Explorer food composition data, indicated a mean flavonoid intake of 372 mg/d in Europe(Reference Zamora-Ros, Knaze and Luján-Barroso25). Therefore, we can estimate that 58 % of polyphenols are ingested as phenolic acids on average across Europe. Again, this proportion varies along the north–south axis from 44 % in countries bordering the Mediterranean Sea to 66 % in the northern and central European countries.
To date, this is the largest study describing phenolic acid intake across several countries of diverse dietary habits. The use of a single FCDB and a standardised 24 h recall allowed reliable comparisons among countries to be drawn. However, as not all the EPIC cohorts are representative of their respective countries, the observed level of intake cannot be extrapolated to the general population of each region. In addition, for some demographic groups, profiles of phenolic acid intake could be very different from those observed in the EPIC. For example, phenolic acid intake would be quite different for children, as well as for adults who consume little or no tea and coffee. Another limitation of the present study could be possible inaccuracies in estimations due to the variation in the methods of preparation of some foods between countries. In the case of coffee, larger volumes are undoubtedly consumed in northern Europe, but the strength of a typical brew is thought to vary considerably from country to country and larger volumes might be offset by a weaker average brew(Reference Sánchez-González, Jiménez-Escrig and Saura-Calixto26, Reference Parras, Martínez-Tome and Jiménez27). Moreover, Phenol-Explorer contains data on raw foods only, although many processes, such as cooking and peeling, can cause variable losses in the concentrations of polyphenols(Reference Manach, Scalbert and Morand2, Reference Amarowicz, Carle and Dongowski28). Therefore, vegetables and cereals might overestimate their contribution to phenolic acid intake, although these were not the most important food sources in the present study. Further underestimation may arise due to the omission of dietary supplements in this analysis. However, there are few consumers of herb or plant supplements in the present study (up to 5 % in Denmark, the highest consumer country) and supplements containing phenolic acid are not commonly consumed(Reference Skeie, Braaten and Hjartaker29). Despite these limitations, Phenol-Explorer is currently the most extensive FCDB on phenolic acids. A compilation of new data on the effects of food processing and cooking on polyphenol content in foods is in progress, and these data should further enrich the Phenol-Explorer database and the accuracy of polyphenol intake estimations. Therefore, the present estimations of phenolic acid intake might be revised when more data become available.
In conclusion, the present study presents the most extensive description so far of both total and individual phenolic acid intakes and their food sources across the ten European countries. Phenolic acids account for the major fraction of the total polyphenols ingested through Western diets in all European countries examined. Coffee is the main food source of phenolic acids. Differences in the nature and amount of the individual phenolic acids consumed are observed between countries along a north–south gradient, mainly due to the differences found in the consumption of phenolic acid-rich foods, particularly coffee. These differences were shown to be related to sociodemographic, anthropometric and lifestyle factors. More epidemiological studies on phenolic acids should be undertaken to explore links with the risk of chronic diseases.
Acknowledgements
The present study was supported by the European Commission: Public Health and Consumer Protection Directorate 1993–2004; Research Directorate-General 2005; Ligue contre le Cancer, Institut Gustave Roussy, Mutuelle Générale de l'Education Nationale, Institut National de la Santé et de la Recherche Médicale (INSERM) (France); Institut National du Cancer (France), German Cancer Aid; German Cancer Research Center; German Federal Ministry of Education and Research; Danish Cancer Society: Health Research Fund (FIS) of the Spanish Ministry of Health (RTICC (DR06/0020); the participating regional governments from Asturias, Andalucía, Murcia, Navarra and Vasco Country and the Catalan Institute of Oncology of Spain; Cancer Research UK; Medical Research Council, UK; the Stroke Association, UK; British Heart Foundation; Department of Health, UK; Food Standards Agency, UK; the Wellcome Trust, UK; Hellenic Ministry of Health, the Stavros Niarchos Foundation and the Hellenic Health Foundation; Italian Association for Research on Cancer; Compagnia San Paolo, Italy; Dutch Ministry of Public Health, Welfare and Sports; Dutch Ministry of Health; Dutch Prevention Funds; LK Research Funds; Dutch ZON (Zorg Onderzoek Nederland); World Cancer Research Fund (WCRF); Statistics Netherlands (The Netherlands); Swedish Cancer Society; Swedish Scientific Council; Regional Government of Skane, Sweden; Nordforsk – Centre of Excellence programme. Some authors are partners of ECNIS, a network of excellence of the 6FP of the EC. R. Z.-R. is thankful for a postdoctoral programme Fondo de Investigación Sanitaria (FIS; no. CD09/00133) from the Spanish Ministry of Science and Innovation. We thank Raul M. García for developing an application to link the FCDB on phenolics and the 24 h dietary recall occurrences. We also thank Bertrand Hémon for computing the intake of phenolic acids in the EPIC study. The authors' contributions are as follows: R. Z.-R., A. S. and C. A. G. designed the research; R. Z.-R. and J. A. R. conducted the research; R. Z.-R. performed the statistical analysis; R. Z.-R. and J. A. R. wrote the manuscript. All authors contributed to the acquisition and interpretation of the data, to the critical revision of the manuscript and to the approval of the final manuscript. The authors have no conflict of interest.