Consumption of a meal that is high in readily available carbohydrates and fat results in a postprandial increase in markers of oxidative stress(Reference Burton-Freeman, Linares and Hyson1, Reference Burton-Freeman2) and insulin resistance(Reference Shoelson, Lee and Goldfine3). Recently, we have reported that a meal-induced increase in oxidised LDL, an indicator of oxidative stress, was attenuated by concomitant intake of strawberry(Reference Burton-Freeman, Linares and Hyson1). Strawberries have a high antioxidant capacity per serving(Reference Halvorsen, Holte and Myhrstad4) and have been shown to increase the antioxidant capacity of the plasma when consumed(Reference Prior, Gu and Wu5). Strawberries also contain a variety of polyphenolic compounds that include anthocyanins (responsible for the red colour in strawberry flesh), flavonols, flavanols, ellagitannins (including small amounts of ellagic acid) and derivatives of hydroxycinnamic acid(Reference Aaby, Skrede and Wrolstad6). These compounds have a high antioxidant capacity(Reference Appel, Moore and Obarzanek7). While bioavailability of these compounds in plasma appears relatively low (e.g. anthocyanins 1–3 %), most agree that this represents an underestimate due to measurement of free v. bound compounds (in plasma) and unaccounted microbial-derived metabolites of parent compounds. Nonetheless, acute increases in plasma antioxidant capacity measured as oxygen radical absorbance capacity (ORAC) have been documented with the consumption of strawberries, grapes, blueberries and kiwi fruit(Reference Prior, Gu and Wu5). In addition, strawberries also contain significant quantities of vitamin C and glutathione, which are also antioxidants(Reference Guo, Cao and Sofic8). Thus, strawberries could potentially attenuate oxidative stress-related inflammatory changes resulting from the consumption of high-fat/carbohydrate meals.
The insulin signalling cascade is sensitive to the reduction–oxidation (redox) balance of the cell(Reference Vinayagamoorthi, Bobby and Sridhar9), and insulin resistance coexists in pro-oxidant and pro-inflammatory states, such as obesity and diabetes(Reference Heilbronn and Campbell10). The present study is concerned with the acute inflammatory responses associated with meals typifying Westernised eating patterns and their relationship with impaired insulin signalling and the ability of strawberry to attenuate these effects and to reduce insulin resistance. Therefore, the study was undertaken to test the hypothesis that a single serving of strawberry provided in a beverage along with a standard high-carbohydrate, moderate-fat meal (HCFM) would reduce the acute postprandial inflammatory and insulin responses to the meal in men and women. Additional interests included characterising the polyphenolic profile of the absorbed strawberry phenolics in relation to measured biological responses.
Methods
Human study
The study involved human subjects and was conducted according to the guidelines laid down in the Declaration of Helsinki. All procedures involving human subjects were approved by the Institutional Review Board, which protects the rights and welfare of human subjects in research studies conducted under the auspices of the University of California (Davis, CA, USA). Written informed consent was obtained from all subjects before beginning the study procedures.
Subjects
A total of twenty-six men (n 10) and women (n 16) with BMI between 25 and 33·5 kg/m2 were recruited from the Sacramento, CA, USA community after completing an Informed Consent Form approved by the Institutional Review Board. The exclusion criteria were (1) tobacco use, (2) use of medications that would interfere with study endpoints (i.e. lipid-lowering medications, anti-inflammatory drugs and dietary supplements), (3) allergy or intolerance to strawberries, (4) diabetes mellitus or fasting glucose >6·11 mmol/l, (5) uncontrolled hypertension (>140/90 mmHg), (6) documented atherosclerotic and (7) chronic inflammatory or other systemic diseases. Of the sixteen women, two dropped out of the study because of work commitments. The final study sample size was ten men and fourteen women.
Study design, treatments and procedures
This was a single-centre, randomised, single-blind, placebo-controlled, cross-over trial conducted at the National Institutes of Health-sponsored UC Davis Clinical and Translational Science Center, Clinical Research Center at the UC Davis Medical Center-affiliated Northern CA Veteran's Affairs Medical Center in Mather, CA, USA. The basic design involved the administration of a test meal followed by periodic blood sampling.
Test meals
Eligible subjects had a 7 d run-in before the actual experiment during which they were required to avoid consuming berries, including strawberries, while maintaining all other aspects of their diet and physical activity. During the experiment, the subjects consumed two test meals in random order, with each subject serving as his/her own control. Test meals consisted of breakfast food items (Table 1), which were accompanied by either an active strawberry beverage or a strawberry-flavoured beverage that served as a placebo. Both beverages were milk-based and the compositions were matched in terms of total energy (4026·6 v. 4020·4 kJ, respectively). Energy from total fat, saturated fat and carbohydrates and the fibre content were also matched. The details of the meals are given in Table 2.
Table 1 Test meal for postprandial study days

* All foods were purchased from local Safeway stores in Davis and Sacramento, CA, USA, and prepared fresh on each study day. Safeway brands (Oakland, CA, USA) were purchased for all food items except for cantaloupe, which was purchased from the produce section; brand not identified.
† These foods were combined to enhance palatability and maintain a blend.
Table 2 Nutrient breakdown of the test meal, including strawberry and placebo beverages

RAE, retinol activity equivalents, DFE, dietary folate equivalents.
* Nutrients were analysed using the ESHA Food Processor program (Salem, OR, USA).
The active strawberry beverage contained a mixture of cultivated strawberry fruits grown in California and was provided by the California Strawberry Commission (Watsonville, CA, USA) in freeze-dried form. All the ingredients (Table 3) were scale weighed to 0·1 g. All ingredients except for the freeze-dried strawberry powder were then mixed in a blender. The freeze-dried strawberry powder (Watsonville, CA, USA) was sprinkled into the blended mixture while continuing to blend. Prepared beverages were immediately frozen ( − 20°C) to be used the next day.
Table 3 Ingredients of the strawberry and placebo beverages

* Nesquik strawberry artificially flavoured powder (Nestlé USA, Inc., Glendale, CA, USA).
† Premium Sanalac Nonfat Dry Milk (ConAgra Foods, Inc., Omaha, NE, USA).
‡ Domino sugar (Domino Foods, Yonkers, NY, USA).
§ Freeze-dried strawberry powder, California Strawberry Commission (Watsonville, CA, USA).
The postprandial test was conducted according to a standardised protocol(Reference Burton-Freeman, Davis and Schneeman11). Briefly, the subjects reported to the laboratory in the morning in a fasting state on two occasions 3–5 d apart. Indwelling catheters were placed in the antecubital vein of the non-dominant arm of the subjects, and blood samples were collected before (time 0 min) and at multiple time points after the meal, 30, 60, 90, 120 min, and then hourly thereafter to 6 h. Blood samples were prepared for analysis of insulin, glucose, high-sensitivity C-reactive protein (hs-CRP), IL-1β, IL-6, TNF-α, plasminogen activator inhibitor-1 (PAI-1) and anthocyanins. The meal and the beverages were consumed entirely under supervision in 20 min.
Blood analysis
Blood samples were collected and plasma was stored at − 80°C until analysis for hs-CRP, IL-1β, IL-6, PAI-1 and TNF-α. High-sensitivity assay systems were used for the measurement of hs-CRP, IL-1β, IL-6, PAI-1 and TNF-α (R&D Systems, Inc., Minneapolis, MN, USA). Plasma glucose was analysed using the 2300 STAT Plus (YSI, Inc., the Yellow Springs, OH, USA), and insulin was measured by RIA method according to the manufacturer's instructions (Millipore, Billerica, MA, USA).
Chemistry
Oxygen radical absorbance capacity assay
The ORAC assay was performed according to the method described by Prior et al. (Reference Prior, Hoang and Gu12), with minor modifications. A sample of 0·5 g freeze-dried strawberry powder or beverages was prepared in 20 ml acetone–water (50:50) and agitated for 60 min. A Perkin Elmer Envision plate reader (Perkin Elmer, Waltham, MA, USA) was used to measure the fluorescent decay at a 60 s interval for 60 min at an excitation/emission wavelength of 485 nm/528 nm. A standard curve was obtained by plotting Trolox concentrations against the corresponding area under the curve for each standard. ORAC values for the unknown samples were calculated using the standard curve.
Analysis of polyphenolic compounds in strawberry samples using liquid chromatography–MS/MS
(+)-Catechin, ellagic acid, pellargonidin-3-O-glucoside, pellargonidin chloride, isoquercetin, kaempferol, quercetin-3-O-glucoside, kaempferol-3-O-rutinoside, cyanidin-3-O-glucoside and cyanidin-3-O-rutinoside were purchased from Chromadex, Inc. (Santa Ana, CA, USA). Quercetin dehydrate (98 %) and p-coumaric acid were obtained from Sigma Aldrich (St Louis, MO, USA). All solvents were of liquid chromatography–MS grade, purchased from Fisher Scientific (Pittsburg, PA, USA).
Strawberry powder (100 mg), strawberry beverage (1 ml) and placebo (1 ml) were extracted with acetone–water (70:30)(Reference Seeram, Lee and Scheuller13), and filtered through a 0·2 μm glass filter. The supernatant was evaporated to dryness under N2, reconstituted in water–acetonitrile (ACN) (90:10, v/v) with 0·1 % formic acid and injected for HPLC analysis. Sample analysis was performed on an Agilent 1200 Rapid Resolution HPLC system with a 6460 Triple Quadrupole Mass Spectrometer (Agilent Technologies, Santa Clara, CA, USA)(Reference Aaby, Ekeberg and Skrede14). A Luna RP C-18 column (100 mm × 2·00 mm, inner diameter 3 mm; Phenomenex, Inc., Torrance, CA, USA) was used, and the mobile phase was a gradient of water with 0·1 % formic acid and ACN with 0·1 % formic acid at a flow rate of 0·4 ml/min. The gradient consisted of 5 % ACN in water for 5 min followed by a gradient from 5 to 20 % ACN in 10 min, followed by 50 % ACN in 15 min. The column was maintained at 30°C. Peaks were monitored with a diode array detector scanning at 370 and 280 nm and by MS. Using electrospray ionisation, two separate HPLC analyses were utilised to detect anthocyanins (positive mode (M+H)+) and all other compounds (negative mode (M+H)− ). The initial analysis was carried out using a full MS2 scan followed by MS2 product ion scan and multiple reaction monitoring fragmentation. N2 gas flow was 10 ml/min at 325°C, and sheath gas flow was 11 ml/min at 350°C with a nebuliser pressure at 35 psi. Capillary voltage was 3500 V with a charging voltage of 1000 V. Fragmentation energy and collision energy in the second quadruple were 135 and 30 V, respectively, with the dwell time of 15 ms. Phenolic compounds were identified by retention times relative to external standards and MS2 fragmentation patterns. Where available, a reference standard was used to quantify the identified compounds. Identity of phenolic compounds for which no reference standard was available was confirmed by theoretical molecular weights and fragmentation patterns from the literature(Reference Aaby, Skrede and Wrolstad6). Those compounds were quantified as equivalents of other compounds with a similar structure. Concentrations of polyphenolic compounds are reported as mg/10 g for freeze-dried strawberry powder and mg/305 ml for beverages.
Analysis of plasma samples using liquid chromatography–MS/MS
Plasma samples from twenty-four subjects were prepared by acidifying 1 ml of plasma with formic acid, and then deproteinising with ACN. A portion of each plasma sample was evaporated under N2, dissolved in a mobile phase, and then analysed by liquid chromatography–MS/MS(Reference Liang and Morris15).
The samples were analysed using methods developed for freeze-dried strawberry powder as described earlier. Chromatographic separation was achieved on an Agilent C18 Poroshell column (Agilent Technologies, Santa Clara, CA, USA). Due to a lack of authentic standards, quantification was limited to compounds for which standards were available.
Statistical analysis
Data were analysed by repeated-measures ANOVA using PC-SAS (version 9.1.3; SAS Institute, Inc., Cary, NC, USA) general linear model and MIXED procedures, with treatment, time and sex as main factors and subject as the blocking variable. Absolute values were analysed, unless otherwise noted. Clinical laboratory endpoints not conforming to expected distributional assumptions as tested by univariate procedures and Shapiro–Wilk's tests were log transformed and noted accordingly. Laboratory values with significant variability in fasting/baseline concentrations were normalised before statistical analysis by subtracting fasting values out of postprandial responses. Least-square means as estimates of the 0–6 h responses were compared as were time-by-concentration curves, noting differences at specific time points. Least-square means have previously been referred to as ‘adjusted means’ and represent means that have been corrected for imbalances in other variables.
Significant differences among treatment means were analysed by a pairwise t test and Tukey's honestly significant test for appropriate comparisons. The level used to determine the statistical significance was P < 0·05.
Results
Study subjects
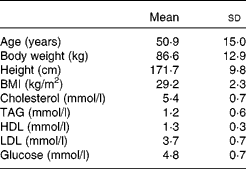
A total of fourteen women and ten men completed the study. The mean age and BMI for the study group were 50·9 (sem 15) years and 29·2 (sem 2·3) kg/m2, respectively. Mean baseline body weight, height, cholesterol, TAG, HDL-cholesterol, LDL-cholesterol and glucose are listed in Table 4.
Table 4 Age, body weight, height, BMI, blood lipids and glucose for twenty-four subjects (ten males and fourteen females) at screening visit
(Mean values and standard deviations)
Anthocyanin and antioxidant capacity of beverages
Preliminary extraction experiments of phenolic compounds from the strawberry powder using methanol–water (80:20, v/v) and acetone–water (70:30, v/v) showed that acetone extracts contained more polyphenols than methanol extracts (data not shown). Therefore, aqueous acetone was chosen for the extraction of phenolic compounds from the strawberry powder in the present study. Table 5 shows identification of major polyphenolic compounds in the strawberry powder through multiple reaction monitoring analysis in both positive- and negative-ion modes labelled as peaks 1–27 in Fig. 1. We detected seven unidentified compounds, and their fragmentation patterns were similar to unknown compounds found in previous studies(Reference Seeram, Lee and Scheuller13, Reference Aaby, Ekeberg and Skrede14). The most abundant anthocyanin in the strawberry powder was pelargonidin-3-O-glucoside, contributing 54 % of the total anthocyanin content followed by pelargonidin-3-O-malonylglucoside (7·4 %), pelargonidin-3-O-rutinoside (1·9 %) and cyanidin-3-O-glucoside (0·63 %). The total amount of anthocyanin in the strawberry powder was 81·6 (sem 5·6) mg/10 g of freeze-dried powder, which is slightly higher than that determined previously with UV–vis and pH differential methods(Reference Aaby, Skrede and Wrolstad6). The total amount of polyphenolic compounds (126·4 (sem 9·2) mg/10 g) of freeze-dried powder was comparable with that reported previously(Reference Aaby, Skrede and Wrolstad6). The corresponding ORAC value was 7700 (sem 20) μm Trolox equivalents.
Table 5 Composition of strawberry powder (mg/10 g), strawberry beverage and placebo beverage (mg/305 ml) as determined by liquid chromatography–MS/MS
(Mean values and standard deviations)

ND, not detected; HHDP, hexahydroxydiphenoyl; ORAC, oxygen radical absorbance capacity.
a,b,c Mean values within a row with unlike superscript letters were significantly different (P < 0·05, n 3).

Fig. 1 Analysis of polyphenolic compounds in the freeze-dried strawberry powder/strawberry beverage using liquid chromatography–MS/MS. Identification of major polyphenolic compounds in the strawberry powder/beverage through multiple reaction monitoring fragmentation analysis in both positive- and negative-ion modes. Peaks are labelled as peaks 1–27; the results of the quantitative analysis are given in Table 5.
The strawberry beverage contained 10 g of freeze-dried strawberry powder, which was equivalent to approximately 100 g of fresh strawberries and delivered 94·7 (sem 2·2) mg total phenols with an ORAC value of 5163 (sem 176) μm Trolox equivalents. The placebo beverage was prepared from non-strawberry ingredients to match flavour, energy and the nutrient profile without the polyphenolic composition of the strawberry beverage (2·4 (sem 0·5) mg total phenols). Its ORAC value was 1006 (sem 79) μm Trolox equivalents). Analysis and comparison of the polyphenolic profile in the strawberry powder v. beverage formulation suggest a potential interaction between polyphenolic compounds and beverage ingredients. The details of the individual polyphenolic compounds are given in Table 5.
Plasma anthocyanin concentrations following the consumption of strawberry and placebo beverages with the high-carbohydrate, moderate-fat meal
Anthocyanins and their metabolites were identified in plasma using the liquid chromatography–MS/MS method as described in the previous sections (Fig. 2). Numerous compounds were identified, of which pelargonidin sulfate, pelargonidin-3-O-glucoside and pelargonidin-3-O-glucuronide were the most abundant. It is interesting to note that pelargonidin sulfate and pelargonidin-3-O-glucoside were present even in fasting plasma samples at concentrations of 64 (sem 5) and 3 (sem 0) nmol/l, respectively (see Fig. 3(a) and (b) and the discussion section).

Fig. 2 Analysis of polyphenolic compounds in plasma samples in response to the strawberry/placebo beverages using liquid chromatography–MS/MS. Chromatogram represents a sample taken at 120 min after a subject given the strawberry beverage with a high-carbohydrate/fat meal. The peak identification method is given in the Materials and methods section.

Fig. 3 Plasma postprandial (a) pelargonidin sulfate and (b) pelargonidin-3-O-glucoside concentrations followed by the strawberry (![]() ) and placebo (
) and placebo (![]() ) beverages with a high-carbohydrate, moderate-fat meal. Values are means, with their standard errors represented by vertical bars (plasma pelargonidin sulfate and pelargonidin-3-O-glucoside concentrations at respective time points before and after the high-carbohydrate, moderate-fat meal challenge with the strawberry beverage or the placebo beverage). Least-square mean values, were significantly different after the meal with the strawberry beverage compared with the placebo beverage for the postprandial pelargonidin sulfate and pelargonidin-3-O-glucoside responses (*** P < 0·001, n 24).
) beverages with a high-carbohydrate, moderate-fat meal. Values are means, with their standard errors represented by vertical bars (plasma pelargonidin sulfate and pelargonidin-3-O-glucoside concentrations at respective time points before and after the high-carbohydrate, moderate-fat meal challenge with the strawberry beverage or the placebo beverage). Least-square mean values, were significantly different after the meal with the strawberry beverage compared with the placebo beverage for the postprandial pelargonidin sulfate and pelargonidin-3-O-glucoside responses (*** P < 0·001, n 24).
The concentration and time maxima (C max, T max) of pelargonidin sulfate and pelargonidin-3-O-glucoside were 139 (sem 7) and 14 (sem 1) nmol/l at 120 min, respectively, after ingestion of strawberry beverage with the HCFM. No significant changes were evident after the placebo beverage with the HCFM compared with the baseline value (P>0·05). Postprandial pelargonidin sulfate and pelargonidin-3-O-glucoside as measured by least-square means over 6 h were significantly increased when the strawberry beverage was consumed concurrently with the HCFM compared with the placebo beverage with the same meal (P < 0·001; Fig. 3(a) and (b)).
Effect of acute exposure to strawberry and placebo beverages with the high-carbohydrate, moderate-fat meal on plasma glucose, insulin and inflammatory markers
Postprandial changes in glucose, insulin and inflammatory markers were measured over the 360 min (6 h) experimental period at defined intervals after meal consumption. Data are presented as least-square means with their standard errors, which was taken to be indicative of the overall postprandial response (Table 6). Postprandial hs-CRP was significantly lower when the strawberry beverage was consumed concurrently with the HCFM compared with the placebo beverage (P = 0·02; Table 6).
Table 6 Summary of 6 h postprandial responses to a high-carbohydrate, moderate-fat meal (HCFM) with the strawberry or placebo beverage
(Least-square means with their standard errors as estimate of the 6 h postprandial response)
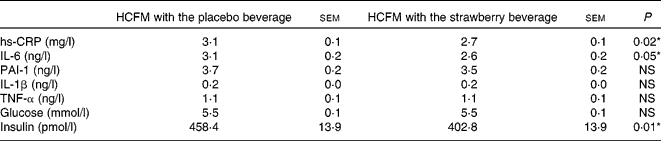
hs-CRP, high-sensitivity C-reactive protein; PAI, plasminogen activator inhibitor.
The postprandial IL-6 response to the meal with the strawberry beverage was significantly lower than the response observed after the meal accompanied by the placebo beverage (P < 0·05). The differences were not apparent until 6 h (3·4 (sem 0·5) v. 4·5 (sem 0·5) pg/ml, P < 0·05; Fig. 4(a)). IL-1β tended to be lower at 6 h after the meal with the strawberry beverage compared with the placebo beverage. However, this difference was not significant (0·15 (sem 0·05) v. 0·27 (sem 0·05) pg/ml P = 0·11; Fig. 4(b)). Postprandial PAI-1, TNF-α and IL-1β concentrations were not significantly different between the treatments (Table 6).
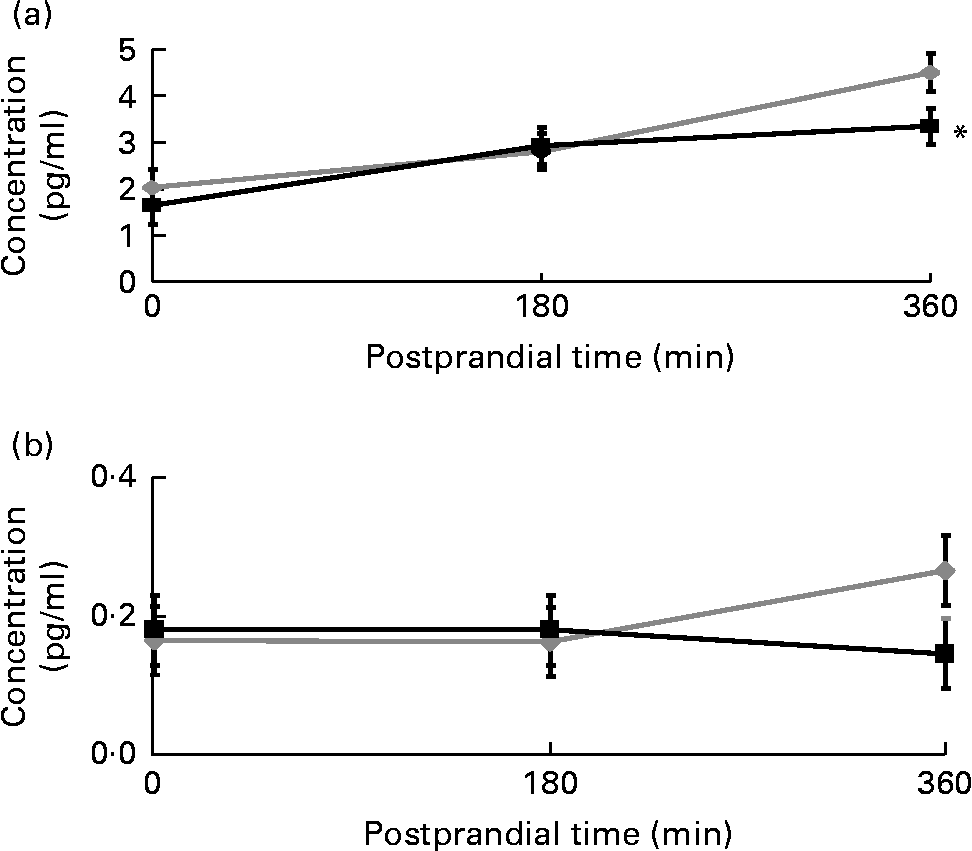
Fig. 4 Postprandial (a) IL-6 and (b) IL-1β concentrations. Values are means, with their standard errors represented by vertical bars (plasma IL-6 and IL-1β concentrations at respective time points before and after a high-carbohydrate, moderate-fat meal challenge with the strawberry (![]() ) or the placebo (
) or the placebo (![]() ) beverage). Least-square mean values were significantly different after the meal with the strawberry beverage compared with the placebo beverage for the postprandial IL-6 response (* P < 0·05, n 24). Least-square mean values were not significantly different between the different treatments for postprandial IL-1β concentrations.
) beverage). Least-square mean values were significantly different after the meal with the strawberry beverage compared with the placebo beverage for the postprandial IL-6 response (* P < 0·05, n 24). Least-square mean values were not significantly different between the different treatments for postprandial IL-1β concentrations.
Postprandial plasma insulin concentrations were significantly lower when the strawberry beverage accompanied the HCFM compared with the placebo beverage (P = 0·01; Fig. 5(a)). However, plasma glucose concentrations were not different between the treatments (Fig. 5(b)).
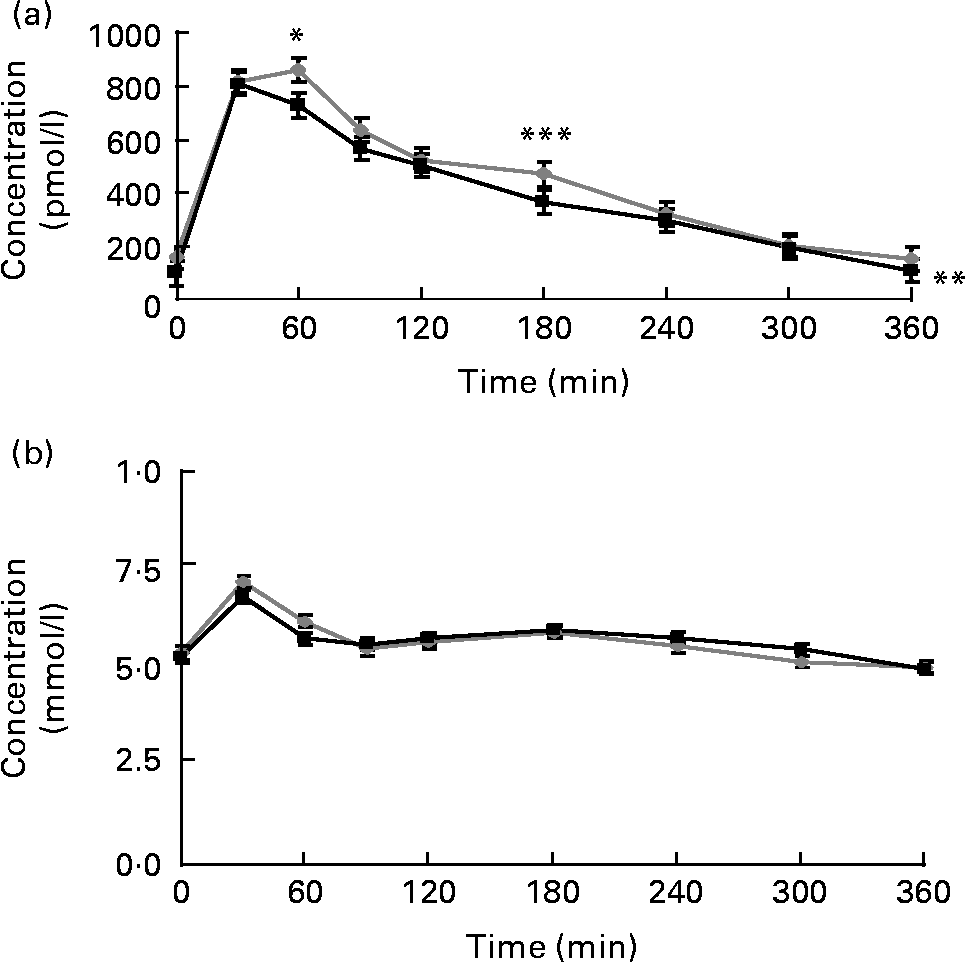
Fig. 5 (a) Postprandial insulin and (b) glucose concentrations. Values are means, with their standard errors represented by vertical bars (plasma insulin or glucose concentrations at respective time points before and after a high-carbohydrate, moderate-fat meal challenge with the strawberry (![]() ) or the placebo (
) or the placebo (![]() ) beverage). Least-square mean values were significantly lower after the meal with the strawberry beverage compared with the placebo beverage for the postprandial insulin concentrations (** P < 0·01 v. placebo, n 24). Additionally, mean insulin concentrations at 60 and 180 min after the meal with the strawberry beverage were significantly lower compared with the meal without the strawberry beverage. Least-square mean values were significantly lower after the meal with the strawberry beverage compared with the placebo beverage for the postprandial insulin concentrations: * P < 0·05, *** P < 0·05.
) beverage). Least-square mean values were significantly lower after the meal with the strawberry beverage compared with the placebo beverage for the postprandial insulin concentrations (** P < 0·01 v. placebo, n 24). Additionally, mean insulin concentrations at 60 and 180 min after the meal with the strawberry beverage were significantly lower compared with the meal without the strawberry beverage. Least-square mean values were significantly lower after the meal with the strawberry beverage compared with the placebo beverage for the postprandial insulin concentrations: * P < 0·05, *** P < 0·05.
Discussion
Obesity, the metabolic syndrome and type 2 diabetes are major risk factors for CVD. Plant foods, particularly fruits and vegetables, have been identified as dietary components that could reduce the risk of developing certain types of CVD such as hypertension(Reference Appel, Moore and Obarzanek16) and myocardial infarction(Reference Yusuf, Hawken and Ounpuu17). Fruit and vegetable intake has also been identified in providing benefit to improving major risk factors of CVD(Reference Liu18). The mechanisms underlying this apparent protective influence remain undefined; however, oxidative stress, inflammation and insulin resistance are common pathologies of CVD. The purpose of the present study was to examine the effects of a common fruit with antioxidant properties, strawberry, on postprandial inflammation and insulin sensitivity in at-risk overweight dyslipidaemic individuals. The study was designed to define the acute effects of consuming a strawberry product on the postprandial changes of inflammation and insulin sensitivity in human subjects.
Strawberry is a polyphenolic-rich fruit and has a unique profile of complex and simple phenolic compounds. The antioxidant activity of strawberry is derived not only from polyphenolic compounds but also from vitamin C and glutathione(Reference Guo, Cao and Sofic8). Analysis of an aqueous extract of freeze-dried strawberry powder indicated that the most abundant polyphenolic compounds present in strawberry were significantly reduced when formulated in a milk-based beverage (Table 5). Although antioxidant activity, as determined by the ORAC value, was significantly lower in the milk-based strawberry beverage compared with the freeze-dried strawberry powder, we were able to demonstrate significant changes in oxidative stress-related inflammatory markers and insulin concentrations in response to the strawberry milk-based formula. The biological significance of the losses observed during preparation of the beverage is unclear at present.
The decreased concentrations of some polyphenolic compounds in the milk-based formula could be due to the formation of secondary bonds between polyphenols and milk proteins(Reference Charlton, Baxter and Khan19) resulting in protein–polyphenol complexes. These interactions can be both reversible and irreversible depending on pH, temperature, and protein and polyphenol concentrations(Reference Luck20). The fate of these complexes in the gastrointestinal tract is also not known. Serafini et al. (Reference Serafini, Ghiselli and Ferro-Luzzi21) found that the addition of milk to black tea abolished the increased antioxidant potential of plasma that was observed when tea was consumed without milk. However, it has also been reported that proteins have little effect on the bioavailability or on the antioxidant capacity of fruit polyphenols(Reference Hollman, Van het Hof and Tijburg22).
Fruits and vegetables contain significant quantities of polyphenolic compounds, which have demonstrable antioxidant activity in vitro (Reference Handique and Baruah23). Although their antioxidant activity in vivo is less well documented(Reference Williams, Spencer and Rice-Evans24), it has been suggested that they may also influence receptor sensitivity and inflammatory enzyme activity(Reference Virgili and Marino25), and thereby potentially ameliorate the metabolic effects of HCFM. Anthocyanins are among the few polyphenols that can be detected in plasma in the same native structure found in plant foods (glycosides). However, they can also undergo changes upon absorption such as glucuronidation, methylation and sulfatation. Several clinical studies have revealed that polyphenolic compounds are poorly absorbed after consuming fruits containing anthocyanins(Reference Lapidot, Harel and Grani26, Reference Felgines, Talavera and Gonthier27). The available data suggested that the concentration of anthocyanins in plasma ranged from 10 to 50 nmol/l (after a 50 mg of aglycone equivalent supply) and T max was 1·5 h. At intakes of 188–3570 mg total cyanidin glucosides, C max was in the range of 2·3–96 nmol/l(Reference Franke, Waterhouse and Teissedre28, Reference Galvano, La Fauci and Vitaglione29). In the present study, we have shown that pelargonidin-3-O-glucoside and pelargonidin sulfate concentrations significantly increased after the consumption of the strawberry beverage containing 28·11 (sem 0·72) mg pelargonidin-3-O-glucoside compared with the placebo beverage (P < 0·001). Pelargonidin sulfate and pelargonidin-3-O-glucoside concentrations in the fasting samples were 64 (sem 5) and 3 (sem 0) nmol/l, respectively. Furthermore, we observed that these compounds did not return to baseline concentrations by 6 h after consuming the meal containing strawberry. The subjects in the present study fasted overnight (10–12 h) and were instructed to avoid berry fruit consumption for at least 2 weeks before the test meal. It is likely that these compounds were derived from other vegetables and fruits that they consumed in the period leading up to the study.
The bioavailability of strawberry anthocyanins has previously been reported(Reference Azzini, Vitaglione and Intorre30–Reference Del Rio, Borges and Crozier32). However, none of the studies has described fasting baseline data with respect to pelargonidin sulfate or pelargonidin-3-O-glucosides after strawberry interventions(Reference Azzini, Vitaglione and Intorre30–Reference Del Rio, Borges and Crozier32). The availability of polyphenolic compounds and their metabolites in fasting samples is largely dependent on the chemical structures of the polyphenolic compounds/their metabolites and their fate in the metabolic pathway(Reference Del Rio, Borges and Crozier32). A study carried out by Koli et al. (Reference Koli, Erlund and Jula33) has clearly shown that certain polyphenolic compounds in fasting plasma are detectable (quercetin, p-coumaric acid, 3-hydroxyphenylacetic acid, caffeic acid, protocatechuic acid, vanillic acid, homovanillic acid and 3-(3-hydroxyphenyl) propionic acid). In this study, the aforementioned compounds were detected in fasting plasma samples in both control and active groups consuming a moderate amount of berries(Reference Koli, Erlund and Jula33). Therefore, the present study provides new data on the bioavailability of pelargonidin sulfate and pelargonidin-3-O-glucoside.
Several studies have demonstrated the antioxidant activity of strawberries(Reference Aaby, Skrede and Wrolstad6, Reference Aaby, Ekeberg and Skrede14, Reference Edirisinghe, Burton-Freeman and Varelis34). We have previously reported that strawberry blocked the postprandial increase in oxidised LDL-cholesterol after a HCFM compared with the placebo group(Reference Burton-Freeman, Linares and Hyson1). The data reported in the present study demonstrate that consuming a strawberry beverage with a meal known to induce postprandial oxidative stress/inflammation also attenuated the meal-induced inflammatory response as indicated by the changes in IL-6 and hs-CRP.
In addition, the results of this investigation also showed that consumption of strawberry beverage significantly reduced the postprandial insulin response to the HCFM, while the changes in glucose concentrations were unaffected. These findings suggest, but do not confirm, that the presence of strawberry may reduce the insulin requirement (i.e. improved insulin efficiency) to achieve glucose homeostasis.
Oxidative stress/inflammation and insulin signalling
In recent years, much has been elucidated about the intracellular signalling pathways activated by oxidative stress and inflammatory responses and how these pathways intersect with and inhibit insulin signalling(Reference Burton-Freeman2). It has been found that treatment of insulin receptor-expressing cells with NEFA(Reference Dresner, Laurent and Marcucci35) and cytokines associated with oxidative and inflammatory stress(Reference Bloch-Damti and Bashan36) stimulates inhibitory phosphorylation of serine (ser-307) residues and reduces tyrosine phosphorylation of insulin receptor substrate (IRS)-1 leading to impaired glucose uptake. ( − )-Epigallacatechin-3-gallate derived from tea has been shown to attenuate this effect in hepatoma cells (HepG2 cells)(Reference Lin and Lin37). Recent in vitro data from our laboratory have indicated that oxidative stress increased inhibitory serine phosphorylation site of IRS-1 (ser-307-IRS-1) and decreased the activation of tyrosine residues (p-tyr-IRS-1) (both critical intermediates in the insulin signalling pathway). An extract of strawberry restored tyrosine phosphorylation in IRS-1. These data also suggest, but do not confirm, that strawberries via their bioactive constituents contribute to improved insulin signalling and, hence, may have a role in reducing insulin resistance(Reference Sandhya, Tadapaneni and Banaszewski38). However, further studies are required to understand the effect of strawberry antioxidants on oxidative stress/inflammatory-induced insulin signalling.
IL-1β and IL-6 are widely known cytokines released by macrophages and are involved in inflammation, cell proliferation, differentiation and apoptosis. A combined elevation of IL-1β and IL-6, rather than the isolated elevation of either IL-1β or IL-6 alone, independently increases the risk of type 2 diabetes(Reference Spranger, Kroke and Möhlig39). Long-term treatment with IL-1β induces insulin resistance in murine and human adipocytes, and IL-1β can increase IL-6 secretion, suggesting that part of the IL-1β effect may be mediated by the up-regulation of IL-6 release(Reference Lagathu, Yvan-Charvet and Bastard40). In the present study, IL-6 was significantly reduced in the postprandial period (6 h). IL1-β concentration at 6 h tended to be reduced, although the overall postprandial response was not significantly decreased, suggesting a delayed effect upon this cytokine. These data suggest an anti-inflammatory role of strawberry with a time course of action that is delayed relative to the antioxidant effects of strawberry. The antioxidant effect of strawberry as measured by LDL oxidation was apparent within 3 h after consumption of strawberry products with a HCFM(Reference Burton-Freeman, Linares and Hyson1). Significantly increased plasma anthocyanin concentrations in response to the strawberry-containing meal were evident as early as 30 min compared with the placebo meal. However, the effect on insulin reduction was observed as early as 1 h. Therefore, the antioxidant/anti-inflammatory effects of strawberry-derived compounds need to be studied further to reconcile the difference in the time course of the changes in glucose, insulin and oxidative stress/inflammatory markers observed in plasma.
Summary
We observed that strawberry significantly attenuated the postprandial inflammatory response following the consumption of a HCFM in an overweight population. We also observed improvement in insulin action. These changes have been observed against a background of increased plasma concentration of anthocyanins derived from strawberry. Overall, these findings suggest beneficial effects that accrue from consumption of fruits that have a high phenolic content.
Acknowledgements
The present study was funded by a grant from the California Strawberry Commission. Chemical analysis of strawberry was conducted at the National Center for Food Safety and Technology (Summit-Argo, IL, USA) under the corporative agreement grant with Food and Drug Administration, Agilent Technologies, USA. The authors would like to acknowledge the contributions of Amanda Linares and Mandeep Cheema who coordinated the trial and performed the hs-CRP and cytokine analyses. Eunyoung Park helped with the statistical analysis. The authors do not have any conflicts of interest with any commercial sponsor. B. M. B.-F. and C. T. K. designed the study; I. E., K. S., J. C., K. B., C. L. E., R. T. conducted the study; B. M. B.-F., I. E. and K. B. analysed the data and compiled the manuscript. All authors read and approved the final manuscript.













