The ability of fish to use dietary carbohydrates is widely variable among and within species and closely associated with their feeding habits. Fish species that show high dietary protein requirements and, therefore, usually presented as ‘carnivorous’ fish are generally considered to be poor utilisers of dietary carbohydrates( Reference Moon 1 – Reference Polakof, Panserat and Soengas 4 ). In spite of the significant research efforts devoted to this theme, the physiological basis for such apparent glucose intolerance in fish is not fully understood. Despite having the whole enzymatic machinery required for carbohydrate utilisation( Reference Enes, Panserat and Kaushik 3 ), fish react with a prolonged postprandial hyperglycaemia after the ingestion of carbohydrate-rich diets( Reference Moon 1 ). Various hypotheses have been proposed to explain this poor utilisation of dietary carbohydrates by fish. Insulin deficiency, lack of insulin-dependent GLUT and lack of an inducible hepatic glucokinase have been proven to be false hypotheses( Reference Mommsen and Plisetskaya 5 – Reference Caruso and Sheridan 8 ). In contrast, the lower potency of glucose over amino acids as insulin secretagogues; a relatively low number of insulin receptors; the lack of an adequate balance between hepatic glucose uptake (glycolysis) and production (gluconeogenesis); and poor hepatic lipogenesis from glucose have been proven to be valid hypotheses( Reference Polakof, Panserat and Soengas 4 , Reference Mommsen and Plisetskaya 5 , Reference Andoh 9 – Reference Ekmann, Dalsgaard and Holm 13 ). Furthermore, the ubiquitous presence of most key enzymes involved in carbohydrate metabolic pathways among fish species indicate that the poor utilisation may be due to an anomalous hormonal and nutritional regulation caused by evolutionary adaptation( Reference Polakof, Panserat and Soengas 4 , Reference Polakof, Mommsen and Soengas 14 ).
The concept of early nutritional programming is being largely studied in mammals to understand how nutritional events during critical periods of development can result in persistent physiological changes in adulthood( Reference Lucas 15 ). In fish nutrition, this raises the possibility of tailoring specific metabolic pathways or functions in juvenile fish, such as the improvement in the use of dietary carbohydrates as energy substrates( Reference Fang, Liang and Zhou 16 , Reference Rocha, Dias and Engrola 17 ). Glucose, as a simple monosaccharide, is a primary source of energy for cells. Immediately after egg fertilisation, glucose derived from maternal glycogen reserves is the first nutrient to be catabolised for cellular division. However, glycogen stores in fertilised fish eggs are extremely low and rapidly depleted( Reference Hoar and Randall 18 , Reference Kamler 19 ). Under these circumstances, glucose is not expected to play a pivotal role in nutrient-sensing pathways during embryogenesis. However, recent studies have suggested that glucose levels, used as a cofactor, can induce modifications on certain epigenetic mechanisms such as histone acetylation and contribute to genomic imprinting, which, in some cases, can be transgenerational( Reference Badeaux and Shi 20 , Reference Patel, Srinivasan and Laychock 21 ). Together, these factors support glucose as a suitable nutrient for studying the effectiveness of nutritional programming in fish.
Recently, early zebrafish embryos at 0·2 d post-fertilisation (dpf) were successfully enriched with glucose by means of direct microinjection into the yolk reserves( Reference Rocha, Dias and Engrola 17 ). In this previous study, we reported that glucose conditioning had no marked beneficial effects on the ability of juvenile fish to cope with high dietary glucose levels, at least at a molecular level; however, a short-term effect (although transient) related to the early stimulus was observed with the down regulation of several metabolic-related genes. Despite being rather weak effects and based only on molecular data, zebrafish juveniles conditioned by an early glucose stimulus showed some indications of enhanced capacity for glucose storage in the muscle, lower glucose production in the liver and lower glucose transport in the intestinal lumen. However, doubts subsisted whether such effects would be more pronounced if we had exerted the glucose stimulus at a later development stage with fully functional metabolic pathways and with a lower supplemental dose of glucose to avoid a potential overload status and, consequently, cellular damage. Meanwhile, Fang et al. ( Reference Fang, Liang and Zhou 16 ) recently showed that a high dietary carbohydrate stimulus exerted at first feeding stages (3–5 dpf) significantly altered the molecular regulation of carbohydrate utilisation, production, digestion and transport in adult zebrafish.
In this context, the objective of the present study was to explore the effects of glucose injection (nutritional stimulus) in the egg at the late embryo stage of 1 dpf on gene expression of target metabolic pathways and [14C]glucose metabolism in juvenile zebrafish challenged with a high-carbohydrate diet. Additionally, the effect of early glucose stimulus on the gene regulation of yolk-sac feeding larvae was also assessed in order to identify possible short-term effects.
Materials and methods
Microinjection procedure
Fertilised zebrafish eggs were obtained from natural spawning of wild-type breeding fish (Centre of Marine Sciences). Embryos were injected into the yolk with 4·6 nl of either a saline solution (Danieau) or 2 m-glucose solution, at the late embryo stage of 1 dpf (during the pharyngula period, according to the method described by Kimmel et al. ( Reference Kimmel, Ballard and Kimmel 22 )). Solutions were prepared according to the procedures described by Rocha et al. ( Reference Rocha, Dias and Engrola 17 ). Microinjection was performed using a 0·5 mm-diameter glass capillary inserted on a nanolitre injector (World Precision Instruments) following the same procedures as described by Rocha et al. ( Reference Rocha, Dias and Engrola 17 ).
Fish rearing and experimental feeds
The present experiment was carried out in compliance with the Guidelines of the European Union Council (2010/63/EU) legislation for the use of vertebrate animals( 23 ). After glucose and saline injections (described above), embryos and larvae were raised in triplicate tanks (n 200) at an initial density of 100 larvae/l, under standardised conditions (28°C) as described previously by Westerfield( Reference Westerfield 24 ). An additional group of non-injected embryos was reared simultaneously to monitor egg quality and embryonic development. From 5 dpf, larvae were fed Artemia nauplii, which were gradually replaced by an inert diet from 10 to 15 dpf. After day 15, larvae were fed exclusively on a low-carbohydrate high-protein (LC) diet used as the control (Fig. 1). From 25 to 35 dpf, all fish from both injection treatments were subjected to a dietary challenge with a high-carbohydrate low-protein (HC) diet (Fig. 1). Throughout the trial, larvae and juvenile zebrafish were fed by hand (four meals/d) until visual satiation. Both diets were well accepted by the fish. Fish mortality was daily monitored, and survival rate was determined at the end of the trial.

Fig. 1 Experimental set-up for zebrafish rearing and feeding regimen up to 35 d post-fertilisation (dpf). Embryos were injected into the yolk at 1 dpf either with a saline or 2 m-glucose solution. At the beginning of exogenous feeding, larvae were fed with Artemia nauplii, which were gradually replaced by a low-carbohydrate high-protein (LC) control diet. Juveniles were subjected to a 10 d dietary challenge, being fed exclusively on a high-carbohydrate low-protein (HC) diet. Sampling points for the metabolic trial and gene expression were marked. Age is expressed as dpf at 28°C.
Formulation of the experimental diets was based on the use of purified ingredients in order to guarantee a high control of nutritional changes among the diets (Table 1). The LC control diet had a high incorporation level of concentrated protein sources (casein, soya isolate, wheat gluten and fish gelatin), guaranteeing a high level of crude protein (70 %) and a low level of carbohydrates (6 %). In the HC (or challenge) diet, the crude protein level was drastically reduced (25 %), whereas carbohydrate level (51 %) was increased through the incorporation of maize dextrin, a highly digestible carbohydrate. Both diets were isolipidic (12 %) and dully supplemented with selected crystalline indispensable amino acids and monocalcium phosphate to avoid essential amino acid or phosphorus imbalance. The experimental diets were manufactured by SPAROS Lda. Powder ingredients were grinded (below 100 μm) in a micropulveriser hammer mill (Hosokawa Micron). Powder ingredients and oil sources were then mixed accordingly to the target formulation in a mixer (Sammic BM5E), and the mixture was humidified with 25 % water. The diets were manufactured by temperature-controlled extrusion (pellet size 2·0 mm) by means of a low-shear extruder (Italplast P55). Upon extrusion, all feed batches were dried in a convection oven (OP 750-UF; LTE Scientifics) for 3 h at 40°C. Dry feed pellets were then grinded in a coffee mill and sieved manually to retrieve the desired particle size (100–200 and 200–400 μm). The diets were analysed for proximate composition according to the following procedures: DM after drying at 105°C for 24 h; ash content by incineration in a muffle furnace at 500°C for 12 h; crude protein (N × 6·25) by a flash combustion technique followed by a gas chromatographic separation and thermal conductivity detection (LECO FP428); fat by dichloromethane extraction (Soxhlet); gross energy in an adiabatic bomb calorimeter (IKA C2000); total phosphorus according to the ISO/DIS 6491 method using the vanado-molybdate reagent.
Table 1 Formulation and composition of the low-carbohydrate high-protein (LC) control and the high-carbohydrate low-protein (HC) challenge diets

* Pharma Grade bloom 240: 92 % CP, LAPI Gelatine SPA.
† Edible acid casein 90 mesh: 85 % CP, EPI Ingredients.
‡ SEAH Soy Instant: 87 CP%, Seah International.
§ VITEN: 86 % CP, 1·3 %, Roquette.
∥ TACKIDEX C070: Roquette.
¶ Marine oil omega-3: Henry Lamotte Oils GmbH.
** PREMIX Lda. Vitamins (mg/kg diet): dl-α-tocopherol acetate, 100 mg; sodium menadione bisulphate, 25 mg; retinyl acetate, 6·9 mg; dl-cholecalciferol, 0·05 mg; thiamin, 30 mg; riboflavin, 30 mg; pyridoxine, 20 mg; cyanocobalamin, 0·1 mg; nicotinic acid, 200 mg; folic acid, 15 mg; ascorbic acid, 1000 mg; inositol, 500 mg; biotin, 3 mg; calcium pantothenate, 100 mg; choline chloride, 1000 mg; betaine, 500 mg. Minerals (g or mg/kg diet): cobalt carbonate, 0·65 mg; copper sulphate, 9 mg; ferric sulphate, 6 mg; potassium iodide, 0·5 mg; manganese oxide, 9·6 mg; sodium selenite, 0·01 mg; zinc sulphate,7·5 mg; NaCl, 400 mg; calcium carbonate, 1·86 g; excipient wheat middlings.
†† Monocalcium phosphate: 22 % P, 16 % Ca, Fosfitalia.
‡‡ Sigma-Aldrich Quimica SA.
§§ Carbohydrate content calculated as: 100–(moisture+protein+fat+ash).
Biological and analytical sampling
Immediately after injection, samples (n 30) of glucose- and saline-injected eggs were collected for the analysis of glucose levels by fluorescent spectroscopy using a commercial kit (Amplit Glucose Quantitation Kit; AAT Bioquest). Fluorescence readings were performed in triplicate using a Synergy™ 4 Multi-Mode Microplate Reader controlled by Gen5™ software (BioTek Instruments). At the end of the experiment (35 dpf), juveniles (n 20 per treatment) were individually sampled for growth determination based on dry weight and total length parameters. Total length was determined using the AxioVision 4.8.2 (Carl Zeiss Limited) program for image analysis, and dry weight measurements were obtained from freeze-dried samples using a precision scale. For gene expression analysis, samples of whole-body larvae (n 20) from each replicate of glucose- and saline-injected treatments were collected during the endogenous feeding period (4 dpf). At the end of the trial, liver and muscle from individual fish (n 6 per treatment) were sampled 6 h after feeding for the same purpose. All samples were randomly collected, snap-frozen in liquid N2 and kept at (80°C until analysis.
Metabolic trial
Tube-feeding method
At 1 d before the final sampling (34 dpf), juveniles from each treatment (n 10) were randomly harvested from the tanks and transferred to the flux laboratory for overnight acclimatisation at room temperature (28°C). Fish were deprived from feed for 16 h before the metabolic trail. The in vivo method of controlled tube-feeding, as described by Rust et al. ( Reference Rust, Hardy and Stickney 25 ) and later modified by Rønnestad et al. ( Reference Rønnestad, Rojas-Garcıa and Tonheim 26 ), for marine fish was adapted to supply nutrients to freshwater species. This approach was used to assess the effects of both nutritional conditioning (early glucose injection) and increase in dietary carbohydrates on the metabolic handling of glucose by zebrafish juveniles. Following a 16 h fasting period, zebrafish juveniles were allowed to feed on the HC diet for a period of 40 min. This feeding period was suitable for the uptake of a full meal, as confirmed by observation of the gastrointestinal tract, and within the beginning of the zebrafish gastrointestinal transit( Reference Field, Kelley and Martell 27 ). Following this single meal, fish were anaesthetised with 33 μm-tricaine methanesulfonate (Sigma-Aldrich). Subsequently, the radioactive label d-[14C(U)]glucose (9·25 MBq; American Radiolabeled Chemicals, Inc.) was added to Ringer salt solution and tube-fed to all fish using a 0·19 mm-diameter plastic capillary (Sigma-Aldrich) inserted on a nanolitre injector (World Precision Instruments). Thereafter, three consecutive injections of 4·6 nl were administered into the fish gut. This injection volume (13·8 nl) was in the range of that previously used for marine species in late larval stages (>25 d after hatching): Senegalese sole( Reference Aragão, Conceição and Martins 28 , Reference Morais, Koven and Rønnestad 29 ); gilthead seabream( Reference Pinto, Rodrigues and Dinis 30 ); white seabream( Reference Saavedra, Conceição and Helland 31 ). After capillary withdrawal, fish were gently rinsed for spillage through two successive wells filled with clean freshwater and transferred into sealed incubation chambers containing 6·5 ml of freshwater. The incubation water was considered to contain all labelled 14C resulting from fish evacuation (evacuated fraction). In addition, an airflow connection was provided between each incubation chamber and a KOH trap (5 ml, 0·5 m), in order to collect all 14CO2 released by the fish through glucose metabolism (catabolised fraction). After an incubation period of 24 h, juveniles from each injection treatment were sampled individually for muscle and viscera, in order to determine the amount of 14C retained in tissues (retained fraction). Tissue samples were immediately solubilised with Solvable (500 μl; PerkinElmer) and kept at 50°C for 24 h. Following larval sampling, the incubation chambers were resealed and 1 ml HCl (0·1 m) was added in a series of gradual steps, resulting in a progressive decrease of pH that causes the rapid diffusion of any remaining 14CO2 from the water into the metabolic trap (catabolised fraction)( Reference Rønnestad, Rojas-Garcıa and Tonheim 26 ). For radioactive counting, dpm were determined in all samples by adding the Ultima Gold XR scintillation cocktail (PerkinElmer) and counting in a TriCarb 2910TR Low Activity Liquid Scintillation analyser (PerkinElmer). Metabolic budgets were calculated after subtracting blanks of each fraction (evacuated, catabolised and retained). Results for each fraction are expressed as a percentage of total label tube-fed, i.e. the sum of dpm in all the compartments of the metabolic chamber and fish.
Gene expression analysis
Real-time PCR
Analyses of mRNA levels were performed at two distinctive periods and sample types: at 4 dpf in whole-body larvae, for assessing the short-term effect of early glucose stimulus (injection), and at 35 dpf in the visceral and muscle tissues of juveniles, for assessing the effects of both early glucose stimulus and dietary challenge. Juvenile fish were sampled 6 h after the last meal, based on previous data identifying this period as relevant for examining the postprandial response of genes in zebrafish( Reference Amaral and Johnston 32 , Reference Seiliez, Medale and Aguirre 33 ). Total RNA was extracted from all samples using 1 ml TRIzol® reagent (Invitrogen). From the resulting total RNA, 1 μg was reverse transcribed into complementary DNA using the SuperScript III RNase H Reverse Transcriptase Kit (Invitrogen) with random primers (Promega). Molecular analysis was focused on the expression of target genes related to glycolysis (GK, glucokinase; HK1, hexokinase 1; 6PFK, phosphofructokinase-6; PK-L, PK-M, pyruvate kinase, both liver and muscle isoforms), gluconeogenesis (PEPCK, phosphoenolpyruvate carboxykinase, both cytosolic and mitochondrial isoforms; FBP, fructose-1,6-bisphosphatase; G6Pase, glucose-6-phosphatase), lipogenesis (FAS, fatty acid synthase; G6PDH, glucose-6-phosphate dehydrogenase; MEc, cytosolic malic enzyme) and glycogen metabolism (GS, glycogen synthase; GP, glycogen phosphorylase). These primers were considered as good molecular markers for nutritional studies in zebrafish( Reference Rocha, Dias and Engrola 17 , Reference Seiliez, Medale and Aguirre 33 ). Gene expression levels were determined by quantitative real-time RT-PCR performed by means of the iCycler iQ (Bio-Rad). Analyses were performed on 5 μl of diluted complementary DNA using the iQ SYBR® Green supermix in a total PCR volume of 15 μl containing, 200 nm of each primer. Thermal cycling was initiated with the incubation at 95°C for 90 s for Taq DNA polymerase activation, then thirty-five steps of PCR were performed, each one consisting of heating at 95°C for 20 s for denaturing and at 55°C or 62°C for 30 s for annealing and extension, depending on the primers. After the final cycle of the PCR, melting curves were systematically monitored (55°C temperature gradient at 0·5°C/s from 55 to 94°C). Each quantitative PCR run included duplicates of samples (reverse transcription) and negative controls (samples without RT or mRNA or complementary DNA). Target gene expression analysis of whole-body larvae and visceral tissue from juveniles was performed using elongation factor-1 (EF1α) as the reference gene, while 18S rRNA was used as the reference gene for muscle samples, once EF1α was not being stably expressed in this tissue. Both EF1α and 18S were employed as non-regulated reference genes and their gene expression values did not significantly change over the respective time frame or tissue type( Reference McCurley and Callard 34 ) (data not shown). Relative quantification of gene expression was performed using the mathematical model described by Pfaffl( Reference Pfaffl 35 ).
Statistical analysis
Data are presented as means with their standard errors of the mean. Criteria expressed as a percentage were arcsine transformed previously to the statistical analysis. The effects of glucose injection on the several analysed parameters in larvae and juvenile fish were tested using SPSS® statistics software 16.0 for Windows by means of an unpaired two-tailed Student's t test. Differences were considered significant at P< 0·05. For relative quantification of gene expression in juvenile fish, the control group was set as the saline-injected HC diet-fed group.
Results
Glucose supplementation, growth performance and survival rate
In comparison with eggs of the saline treatment, those microinjected with a 2 m-glucose solution (9·2 nmol/egg) showed a 6-fold increase in glucose levels. No permanent damage on egg chorion or signs of leakage through the puncture hole was recorded after the injection. Zebrafish growth and survival was affected neither by early glucose stimulus nor by the dietary challenge with high carbohydrates (P>0·05). At the end of the experiment, juveniles showed similar mean values for dry weight (4·7–4·8 mg) and total length (10·0–11·1 mm) (Table 2). The survival rate varied between 61 and 68 % and values were within the expected range for zebrafish fed with live feed and purified diets. Carvalho et al. ( Reference Carvalho, Araujo and Santos 36 ) showed that 27-d-old zebrafish fed from mouth-opening with a semi-purified diet reached a good growth (7·0 (sd 0·2) mm) and survival (55 %) rate.
Table 2 Survival (n 3) and growth (n 20) of zebrafish juveniles initially injected with a saline or glucose solution and challenged with a high-carbohydrate low-protein diet* (Mean values with their standard errors)
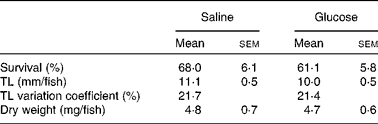
TL, total length.
* Student's t test.
Effects of early glucose stimulus on [14C]glucose metabolism in fish fed with carbohydrates
The survival rate of zebrafish after 24 h of incubation in metabolic chambers was 89 and 100 % for the glucose- and saline-injected treatments, respectively. The results showed that in juvenile fish, glucose evacuation and absorption was not significantly affected by early glucose conditioning (P>0·05; Fig. 2(a)). Zebrafish presented high levels of glucose absorption (over 87 %) under the same intake amount of carbohydrates. Juveniles subjected to the early glucose stimulus showed significantly lower (P <0·05) retention of glucose in visceral tissue (but not in muscle tissue) and, consequently, higher glucose catabolism (P= 0·072) in comparison with the saline-injected group. Retention efficiency in visceral tissue was significantly reduced in juveniles injected with glucose compared with the saline-injected group (6·0 and 11·5 %, respectively; Fig. 2(b)).

Fig. 2 Study of glucose metabolism in zebrafish juveniles. (a) Percentage of the absorbed (black bar) and evacuated (white bar) [14C]glucose in tube-fed zebrafish juveniles initially subjected to either a saline or glucose injection (stimulus) and fed a high-carbohydrate low-protein (HC) diet. (b) Percentage of [14C]glucose retained in muscle tissue (dark-grey bar) and visceral tissue (medium-grey bar), and percentage of [14C]glucose catabolised (light-grey bar) in tube-fed zebrafish juveniles initially subjected to either a saline or glucose injection (stimulus) and fed a HC diet. Retention and catabolism fractions are associated with the absorbed label in the fish. Values (absorption, evacuation, retention and catabolism) are means, with their standard errors represented by vertical bars (n 10). *Mean value was significantly different from that of the saline-injected group (P< 0·05; Student's t test).
Although no significant differences were found, glucose catabolism showed a trend towards an increase in the glucose-injected fish rather than in the saline-injected fish (75·8 and 68 %, respectively) (Fig. 2(b)).
Effects of early glucose stimulus on metabolic gene expression in yolk-sac larvae
The RNA levels of enzymes involved in the intermediary metabolism of yolk-sac larvae (4 dpf) were compared between the saline- and glucose-injected groups (Table 3). For the majority of the analysed genes, mRNA levels remained unaltered between the saline- and glucose-injected treatments. Only the PK gene (muscle isoform) was significantly up-regulated in glucose-injected larvae (P< 0·05; Table 3), while the MEc gene showed a slight but not significant decrease in its expression (P= 0·056). These results suggest that the glucose stimulus had no marked effects on gene regulation in 4 dpf larvae.
Table 3 Relative expression of metabolic genes involved in glycolysis (A), gluconeogenesis (B), lipogenesis (C) and glycogen metabolism (D) in whole-body larvae (4 d post-fertilisation (dpf)) and visceral and muscle tissues from zebrafish juveniles (35 dpf), initially injected with a saline (control) or glucose (stimulus) solution and challenged with a high-carbohydrate low-protein diet* (Mean values with their standard errors; n 6)

* Expression values of larvae and visceral tissue were normalised with α-elongation factor 1 transcripts (EF1α), while that of muscle tissue was normalised with 18S transcripts. Relative fold differences between the treatments were analysed by Student's t test (P< 0·05). Age is expressed as dpf at 28°C. For a description of the gene symbols, refer to the ‘Gene expression analysis’ section.
Effects of early glucose stimulus on metabolic gene expression in fish fed with carbohydrates
The same set of target genes was analysed in two tissue types of juvenile fish (35 dpf): viscera and muscle. The long-term effects on gene expression in fish injected with glucose at an early embryo stage and later subjected to a feeding challenge with high carbohydrates are given in Table 3.
In viscera, the gene expression of GK was unaltered regardless of the injection treatment (glucose stimulus). In contrast, the expression of HK1 was significantly up-regulated (P= 0·04), while that of 6PFK was down-regulated (P= 0·009) in glucose-injected fish, in comparison with the control group (saline-injected). Also, the transcript levels of PK-L were not affected by the early glucose stimulus in the long term. On the gluconeogenic pathway, the expression levels of PEPCKc and PEPCKm were inhibited in glucose-injected fish (P= 0·000 and P= 0·002, respectively), while that of the G6Pase gene was not affected in the long term. Surprisingly, the gene expression level of FBP was reverse-regulated as expected for this pathway: juveniles that were exposed to the glucose stimulus showed higher levels of expression than the saline group (P= 0·034). For genes involved in lipogenesis and energy production (FAS, G6PDH and MEc), no long-term effects related to the glucose stimulus were found (Table 3). Similar observations were found for GS and GP genes responsible for glycogen metabolism (Table 3).
In the muscle tissue of juvenile fish, only the gene expression level of 6PFK was found to be significantly up-regulated (P= 0·038) by early glucose injection (Table 3). Therefore, glucose injection increased the expression of 6PFK in juvenile fish, whereas genes encoding for glycolysis (HK1 and PK-M) and glycogen metabolism (GS and GP) were not affected, in the long term, by early glucose conditioning (Table 3).
Discussion
Fish nutritionists struggle to establish balanced formulations where large amounts of plant ingredients could be included without compromising the nutritional requirements and a good acceptance of the diet by the fish( Reference Gatlin, Barrows and Brown 37 ). The inclusion of plant ingredients, be it dietary protein or starch sources, enhances the overall intake of carbohydrates. However, the ability of fish to use dietary carbohydrates as an energy-yielding substrate is widely variable among species with different feeding habits. New trends in the field of fish nutrition begin to emerge, such as the concept of early nutritional programming, as a promising strategy to enhance the use of alternative feedstuffs( Reference Geurden, Aramendi and Zambonino-Infante 38 , Reference Vagner, Robin and Zambonino-Infante 39 ). We believe that such an approach has the potential to improve specific metabolic pathways, through actions during embryonic development that could permanently alter the capacity of adult fish to cope with dietary sources rich in carbohydrates. However, studies in mammals have shown that the period in which the programming stimulus occurs is usually confined to early stages (pre- or postnatal) that influence the ‘imprinting’ effect as well as the long-lasting outcomes in adulthood( Reference Srinivasan and Patel 40 , Reference Hanley, Dijane and Fewtrell 41 ). The goal of the present study was to gain further knowledge on the effects of a hyperglucidic stimulus during the final stage of fish embryogenesis on growth, nutrient metabolism and gene expression regulation in zebrafish juveniles subjected to a drastic increase in the ratio of dietary carbohydrates:proteins.
Effects of early glucose stimulus on the growth and survival of zebrafish
In our previous study, the glucose injection used as a trigger for nutritional programming in zebrafish eggs (0·2 dpf) was found to suppress in the embryos (4 dpf) the transcription level of several genes involved in glycolysis, glucose transport and lipogenesis( Reference Rocha, Dias and Engrola 17 ). The inhibition of these metabolic pathways was somehow unexpected since the reverse (stimulation) was observed in several fish species and at later life stages, after the intake of high dietary carbohydrates( Reference Polakof, Panserat and Soengas 4 ). We, therefore, considered the possibility that glucose overload at this incipient developmental stage induced cellular damages or compromised key metabolic regulators. Here we explore a new ‘metabolic window’ for glucose delivery (stimulus) at a later period of embryonic development (1 dpf). This period was selected based on two important features: the level of endogenous glucose in the yolk and the stage of embryo development. Zebrafish dynamically regulates glucose during embryogenesis, reaching the highest endogenous levels at 1 dpf, before hatching( Reference Rocha, Dias and Engrola 17 , Reference Soanes, Achenbach and Burton 42 , Reference Jurczyk, Roy and Bajwa 43 ). We hypothesised that supplementation with high doses of glucose (9·2 nmol/egg) while endogenous glucose levels are elevated would contribute to a better adaptation of embryos for hyperglycaemia conditions. A lower dose may mask the stimulus effect by not altering enough the ratio of carbohydrate over other macronutrients, taking into account the overall low glucose level in cyprinid eggs( Reference Lahnsteiner, Urbanyi and Horvath 44 ). The glucose injections (9·2 nmol/egg) increased by 6-fold the glucose levels relative to those in 1 dpf embryos of the saline-injected group (control). In absolute levels, the amount of glucose injected into the egg was similar to basal glycaemia levels found in adult zebrafish fasted for 24 h (about 2·5 mm; 40–45 mg/l)( Reference Eames, Philipson and Prince 45 ). Thus, the achieved glucose supplementation can be considered within the physiological range for this species. At the 1 dpf stage, major processes of organogenesis are ongoing in several systems, such as the emergence of hepatic and pancreatic buds (future key metabolic organs)( Reference Tao and Peng 46 , Reference Tehrani and Lin 47 ), the functioning of the heart and circulatory system and the appearance of muscle spontaneous contractions( Reference Kimmel, Ballard and Kimmel 22 , Reference Saint-Amant and Drapeau 48 ). In addition, the transcriptional regulation of the insulin gene and the gluconeogenic genes fructose-bisphosphate aldolase and phosphoenolpyruvate carboxykinase already occurs at this early stage( Reference Soanes, Achenbach and Burton 42 , Reference Papasani, Robison and Hardy 49 ). Therefore, the stage of 1 dpf can be considered of high genetic plasticity, once important metabolic pathways are newly established or in the process of becoming active, suggesting that possible epigenetic alterations can occur during this period due to early nutritional events( Reference Lillycrop and Burdge 50 ).
To avoid the influence of sexual dimorphism on hepatic gene transcription already reported in zebrafish( Reference Robison, Drew and Murdoch 51 ), the present trial was conducted up to 35 dpf (5 weeks) so that fish could remain immature. Evidence on the proteomic field had demonstrated that zebrafish can mature before reaching 3 months (90 dpf), in few cases from 30 dpf onwards( Reference Gomez-Requeni, Conceicao and Olderbakk Jordal 52 ), suggesting that fish length (growth) can have a stronger effect on sexual maturation rather than age( Reference Spence, Gerlach and Lawrence 53 ). We found that glucose supplementation by microinjection did not impair embryonic development, and that the stimulus along with a drastic increase in carbohydrate intake had no detrimental effects on the growth and survival of juvenile fish. The lack of the negative effects on physiological parameters is in conformity with other nutritional programming studies performed in fish( Reference Fang, Liang and Zhou 16 , Reference Rocha, Dias and Engrola 17 , Reference Geurden, Aramendi and Zambonino-Infante 38 ).
Immediate and persistent effects of early glucose stimulus on gene expression and metabolic utilisation of glucose in juvenile fish
In mammals, prenatal exposure to high glucose levels can permanently alter adult metabolism and trigger diet-related diseases such as diabetes, the metabolic syndrome and obesity( Reference Burdge and Lillycrop 54 ). Despite the lack of full knowledge on the mechanisms involved in metabolic regulation by nutritional factors, there is strong evidence that the genome can be ‘imprinted’ to store the memory of the early nutritional event( Reference Lucas 15 ). However, caution should be taken in extrapolating the programming concept from mammals to fish. Mammals and fish exhibit very distinct embryonic development (in uterus v. ex uterus); thus, stimulus delivery during a sensitive phase becomes more restricted to the control and manipulation of fish, mostly because fish embryos operate as energetically closed systems during yolk reserve consumption( Reference Kamler 19 ). Furthermore, fish have a poor control over glucose homeostasis and exhibit slow metabolic rates for glucose utilisation compared with mammals( Reference Hemre, Mommsen and Krogdahl 2 ). Therefore, the results from the present study addressing metabolic programming by early glucose stimulus will be cautiously discussed in comparison with mammals, whenever possible.
Short-term effect of early glucose stimulus on metabolic gene expression
The short-term effect related to glucose injection (stimulus) on the gene expression of free-swimming 4 dpf larvae that still rely exclusively on endogenous yolk reserves for nutrient supply was assessed. From the metabolic genes analysed, only the expression level of PK (muscle isoform) was up-regulated in the glucose-injected group. In mammals, the expression of PK is regulated by both dietary carbohydrates and hormones (insulin and glucagon) at a pre- and post-translational level( Reference Yamada and Noguchi 55 ). Such dynamic regulation is thought to occur in adult zebrafish according to different feeding conditions: refeeding a commercial diet poorly regulates the postprandial expression of PK, while a high carbohydrate meal induces its expression( Reference Seiliez, Medale and Aguirre 33 ), with the latter being in line with the higher PK mRNA levels after the delivery of the glucose stimulus at 4 dpf. Overall, the early glucose stimulus had a poor short-term effect on the transcript levels of zebrafish larvae, which may be due to the 3 d gap between glucose injection (at 1 dpf) and sampling (at 4 dpf). However, this does not exclude the possibility of a long-term effect of glucose injection at a later life stage, when the fish are confronted again to a challenging nutritional condition. Indeed, sea bass larvae fed diets containing different highly unsaturated fatty acid levels showed no change in the expression levels of several lipogenic enzymes during the first days of stimulus, whereas long-term molecular changes were found in juvenile fish fed a highly unsaturated fatty acid-deficient diet( Reference Vagner, Robin and Zambonino-Infante 39 ). In terms of short-term effects, we obtained less responsiveness of metabolic genes towards glucose stimulus delivered at 1 dpf in comparison with our previous study, where glucose was injected at 0·2 dpf( Reference Rocha, Dias and Engrola 17 ). Although the glucose stimulus was the same in both studies (9·2 nmol/egg), the shift of stimulus delivery towards a ‘window’ of high embryonic development (1 dpf) did not inhibit gene expression, which allowed excluding the hypothesis of cell damage due to glucose overload.
Long-term effect of early glucose stimulus on metabolic gene expression
The long-term effect was evaluated in juvenile fish (35 dpf) after being challenged for 10 d with the HC diet, with the idea that the early glucose injection may generate a ‘metabolic memory’ for improving the future use of carbohydrates. The present study confirms the potential of a single glucose injection (at 1 dpf) to induce persistent molecular changes, as shown by the enhanced expression of genes involved in the first step of glycolysis and gluconeogenesis pathways in the visceral tissue of juveniles conditioned by the glucose stimulus. The increase in the expression level of HK1 and the simultaneous decrease in the expression of PEPCK suggest a higher capacity for glucose phosphorylation as well as a lower glucose production, and thus, the possibility of glucose stimulus to ‘programme’ these two major pathways towards a more efficient control of glucose homeostasis when subjected again to hyperglycaemic conditions. Although not fully understood, the persistent hyperglycaemia observed in several fish species after high carbohydrate intakes has been ascribed to an atypical regulation of hepatic gluconeogenesis( Reference Panserat and Overturf 56 , Reference Panserat and Kaushik 57 ). More specifically, in contrast to mammals, fish transcriptional regulation of gluconeogenic genes does not seem to be down-regulated by high dietary carbohydrate intakes( Reference Panserat, Medale and Breque 12 , Reference Panserat and Kaushik 57 – Reference Panserat, Plagnes-Juan and Breque 59 ). As such, the possibility to programme and decrease this pathway in conditions of hyperglycaemia (through an early glucose stimulus) is encouraging. Here we found that both mitochondrial and cytosolic isoforms of the PEPCK gene were affected by the stimulus at the late juvenile stage. A similar down-regulation of PEPCKc was also reported in zebrafish juveniles from our previous study following glucose injections at 0·2 dpf( Reference Rocha, Dias and Engrola 17 ), which reinforces our hypothesis of early nutritional programming. Also, the expression of the glycolytic HK1 gene, known to be poorly regulated by dietary carbohydrates in fish( Reference Gonzalez-Alvarez, Ortega-Cuellar and Hernandez-Mendoza 60 , Reference Soengas, Polakof and Chen 61 ) but found here to be enhanced by the early stimulus, appears as a relevant indication for the occurrence of genomic imprinting. Furthermore, the beneficial effects of glucose injection at 1 dpf upon HK1 regulation contrast with our data obtained after glucose injection at 0·2 dpf, where HK1 was down-regulated( Reference Rocha, Dias and Engrola 17 ). The analysis of HK1 enzyme activity in viscera is, however, needed to confirm the physiological relevance. A recent study on metabolic programming of adult zebrafish, but exerting the nutritional stimulus at the larval first-feeding stage showed the same molecular pattern of glycolysis stimulation and gluconeogenesis inhibition following early high carbohydrate intakes( Reference Fang, Liang and Zhou 16 ). In contrast, the gene expression of GK that is highly responsive in fish to rises in dietary carbohydrate intake( Reference Panserat, Medale and Blin 7 , Reference Seiliez, Medale and Aguirre 33 , Reference Gonzalez-Alvarez, Ortega-Cuellar and Hernandez-Mendoza 60 , Reference Panserat, Capilla and Gutierrez 62 , Reference Panserat, Fontagne and Bergot 63 ) was not persistently affected by the glucose stimulus, which is in line with our previous data( Reference Rocha, Dias and Engrola 17 ). The lack of programming effects for the GK gene could be related to the lower sensitivity of the GK enzyme to punctual hyperglucidic stimuli delivered during early stages of fish development, as observed in zebrafish and rainbow trout( Reference Fang, Liang and Zhou 16 , Reference Geurden, Aramendi and Zambonino-Infante 38 ).
On the downstream reactions of both pathways, the long-term effect of glucose stimulus on the expression of 6PFK (key glycolytic enzyme) and FBP (the opposing gluconeogenic enzyme) genes was unexpected. Results in viscera demonstrated that both genes were regulated in the reverse way of what was anticipated: the glycolytic flux was reduced (6PFK down-regulated), whereas gluconeogenesis was increased (FBP up-regulated). Given the similarities between fish and mammals on this enzyme-substrate cycle, the programming effect of glucose stimulus on these genes could have been masked by other factors such as hormonal or allosteric control( Reference Enes, Panserat and Kaushik 3 , Reference Pilkis and Granner 58 ). However, the expression of 6PFK was regulated differently according to tissue type: being down-regulated in viscera but up-regulated in the muscle by the glucose injection. In addition, it was the only gene to be positively altered in muscle tissue, as a long-term consequence of the glucose stimulus. So far, these results present the first indication that the 6PFK–FBP loop can be a target of nutritional programming by early glucose stimulus; however, the reason why the ‘memory’ stored at the genome after the stimulus resulted in such an unexpected programming of 6PFK–FBP remains unclear. Finally, the last step of both metabolic pathways regulated by PK (glycolysis) and G6Pase (gluconeogenesis) enzymes was not affected at a molecular level by early glucose injection, suggesting that the stimulus was not suitable for a permanent imprinting of these genes. For the PK gene, the short-term induction recorded at 4 dpf might indicate that some short-term effects related to the stimulus can be reversible at later stages of development. Likewise, previous studies showed no long-term effects of early glucidic stimuli on G6Pase and PK mRNA levels in juvenile rainbow trout and zebrafish( Reference Fang, Liang and Zhou 16 , Reference Rocha, Dias and Engrola 17 , Reference Geurden, Aramendi and Zambonino-Infante 38 ). However, it has been shown that PK gene expression can be up-regulated by the increase in dietary carbohydrates, as in mammals( Reference Seiliez, Medale and Aguirre 33 , Reference Yamada and Noguchi 55 , Reference Kamalam, Medale and Kaushik 64 ), in contrast to G6Pase which, depending on fish species, appears to be poorly or even not regulated( Reference Panserat, Medale and Breque 12 , Reference Panserat, Capilla and Gutierrez 62 , Reference Caseras, Meton and Vives 65 , Reference Panserat, Plagnes-Juan and Kaushik 66 ). Therefore, the regulation of a certain metabolic gene in response to higher carbohydrate intake cannot be used as an indicator of possible long-term effects related to early glucidic stimulus. The different susceptibilities of each gene to epigenetic modifications as well as all the different epigenetic mechanisms that can be involved after an early nutritional event are important factors to be considered for possible programming effects( Reference Lillycrop and Burdge 50 , Reference Canani, Di Costanzo and Leone 67 ).
Concerning the genes involved in lipogenesis and glycogen metabolism, the early glucose stimulus had no long-term effects as all the analysed genes had the same level of expression between the saline- and glucose-injected fish. This result with respect to lipogenesis is in line with our earlier data on zebrafish conditioned to glucose at 0·2 dpf( Reference Rocha, Dias and Engrola 17 ), but disagrees with the hypothesis of increased lipogenesis as a consequence of the early glucose stimulus as anticipated from feeding studies with fish using high dietary carbohydrates( Reference Kamalam, Medale and Kaushik 64 , Reference Shimeno, Kheyyali and Shikata 68 ). Studies on the time course of adaptation of lipogenic enzymes in coho salmon revealed that at least 2–3 weeks are required to cause changes in the activities of these enzymes in response to dietary changes( Reference Lin, Romsos and Tack 69 ). For lipogenesis, our sampling after 10 d of HC dietary challenge may, therefore, not represent metabolically steady-state conditions fully under the influence of the nutritional history of the glucose stimulus. In contrast, since the stimulus did not improve glycogenesis (glucose storage in muscle), as occurred previously in zebrafish( Reference Rocha, Dias and Engrola 17 ), one could consider that glycogen synthesis may have been affected by the reduced amino acid levels in the HC diet. Knowing that amino acids are potent insulin secretagogues in mammals( Reference Armstrong, Bonavaud and Toole 70 ) as in fish( Reference Andoh 9 , Reference Navarro, Rojas and Capilla 11 ), glycogen metabolism could be regulated in a way similar to that in mammals. In this way, the drastic reduction in protein level towards carbohydrate incorporation in the HC challenge diet and the subsequent lowering of amino acids may have limited the full potential of glucose stimulus to program glycogen metabolism in juvenile zebrafish.
Long-term effect of early glucose stimulus on glucose use
The metabolic fate of the tube-fed [14C]glucose tracer revealed that juvenile zebrafish can achieve high absorption levels (over 87 %) of dietary glucose for subsequent retention in tissues or catabolism, regardless of the early nutritional history. Juveniles that were exposed to the glucose stimulus had significantly lower [14C]glucose retention in visceral tissue and, accordingly, higher catabolism, in comparison with the saline-injected group, suggesting an enhancement of glucose oxidation and even a possible decrease in glycogen storage. However, given that no further analysis on glycogen or lipid content in tissues was performed, our hypotheses concerning [14C]glucose retention in tissues are limited. Nevertheless, these results agree with those observed at a molecular level, as fish injected with glucose showed an up-regulation of the expression of HK1 (viscera) and 6PFK (muscle) genes involved in glucose oxidation. Clearly, glucose is an important substrate for oxidation in zebrafish; however, its metabolic utilisation can be enhanced by means of early programming, as demonstrated herein. Such an approach raises the possibility of even greater results upon carnivorous species with less capacity to use dietary carbohydrates as an energy source.
Conclusion
We demonstrated that the late embryo stage is a period of high genetic plasticity and better suitable for nutritional stimulus delivery in zebrafish, when compared with incipient stages of 0·2 dpf. Our data suggest that, at least at a molecular level, the two major pathways for glucose metabolism were permanently modified by early glucose stimulus at specific key metabolic steps. Although few genes were modified by the early stimulus, juvenile fish fed high carbohydrates showed an improved capacity for glucose phosphorylation and lower glucose production in viscera. The metabolic fate of dietary carbohydrates showed that the early glucose injection lowered the retention of [14C]glucose in visceral tissue, thereby promoting higher catabolism by oxidative processes. The present study contributes to the generation of new knowledge on nutritional programming on fish following glucose injection during embryogenesis.
Acknowledgements
The authors acknowledge the EDGE group (Centre of Marine Sciences) for gently conceding zebrafish eggs for the experiment. The authors also thank Patricia Diogo, Carmen Barrios, Helena Teixeira and Rita Colen for their assistance during microinjection and fish rearing, and thank Marianne Cluzeaud for the guidance during the analysis of molecular biology.
The present study was supported by project PTDC/CVT/102481/2008 – ‘ULTRAFISH – Ultrasound to enhance the nutritional status of eggs and yolk-feeding larvae: a novel nutritional tool to program growth potential and metabolic abilities in juvenile fish’, financed by Fundação para a Ciência e Tecnologia (FCT, Portugal). F. R. and S. E. acknowledge the financial support by FCT (Portugal) through grants SFRH/BD/74921/2010 and SFRH/BPD/49051/2008, respectively; J. D., S. E., P. G. and S. P. benefited from travel grants in the framework of Program PESSOA-EGIDE 2011-2012.
The authors' contributions are as follows: F. R. conducted the experiment, performed all the analytical analyses, analysed all data, performed the statistical analysis, prepared the figures and wrote the manuscript; J. D. and S. P. designed the study and supervised the research; F. R. and S. E. were responsible for the metabolic trial with [14C]glucose; F. R. and S. P. performed the gene expression analysis; J. D. formulated and manufactured the diets; S. E., P. G., I. G., M. T. D., J. D. and S. P. contributed to the final content of the manuscript.
None of the authors had conflicts of interest or financial disclosures to declare.








