Child maltreatment is a global public health problem, and indisputably has both immediate and long-lasting pernicious effects on children’s development (Winter et al., Reference Winter, Dittrich, Dörr, Overfeld, Moebus, Murray, Karaboycheva, Zimmermann, Knop, Voelkle, Entringer, Buss, Haynes, Binder and Heim2022). In China, 20%–30% of primary and middle school students have reported experiencing abuse (i.e., caregivers’ physical and/or psychological assaults on children that lead to potential or actual harm), and 44%–47% have reported experiencing neglect (i.e., caregivers’ failure to meet children’s basic physical and/or psychological needs) (Wang et al., Reference Wang, Cheng, Qu, Zhang, Cui and Zou2020). Child maltreatment is biologically embedded and theoretically associated with compromises in stress response systems such as the hypothalamic-pituitary-adrenal (HPA) axis (Koss & Gunnar, Reference Koss and Gunnar2018). Recent decades have witnessed a surge of research on how child maltreatment “gets under the skin” (Holochwost et al., Reference Holochwost, Wang, Kolacz, Mills-Koonce, Klika and Jaffee2020). Nevertheless, more research is needed on how best to conceptualize childhood adversity effects (e.g., threat versus deprivation exposure) on physiological functioning, capture within-individual HPA axis development across sensitive periods, and explore critical individual differences such as sex differences (Gunnar, Reference Gunnar2021; Koss & Gunnar, Reference Koss and Gunnar2018). Importantly, some children who experience maltreatment do not go on to develop significant problems. Thus, a better understanding of resilience is also needed in research on child maltreatment. Building upon the dimensional approach to childhood adversity (McLaughlin & Sheridan, Reference McLaughlin and Sheridan2016; McLaughlin et al., Reference McLaughlin, Sheridan and Lambert2014), in a sample of Chinese boys and girls from a disadvantaged area, this study disaggregates child maltreatment effects in testing whether abuse and neglect are distinctly associated with diurnal cortisol at the baseline assessment and longitudinal changes in diurnal cortisol. The moderating effects of child resilience and sex differences in these associations are also investigated.
Child maltreatment and diurnal cortisol
The HPA axis is one of the core stress response systems that mobilizes energetic resources to help individuals respond to environmental challenges (Koss & Gunnar, Reference Koss and Gunnar2018). Cortisol is the primary end product of the HPA axis, reflecting its overall function. Cortisol levels follow a circadian rhythm in that they are high at awakening (awakening cortisol), rapidly increase to a peak about 30 min after awakening (cortisol awakening response, CAR), and then gradually decline throughout the day (diurnal cortisol slope) (Adam & Kumari, Reference Adam and Kumari2009). The awakening cortisol, CAR, and diurnal cortisol slope are related yet distinct indicators that may provide unique and complementary information about the diurnal cortisol rhythm (Adam et al., Reference Adam, Quinn, Tavernier, McQuillan, Dahlke and Gilbert2017; Stalder et al., Reference Stalder, Lupien, Kudielka, Adam, Pruessner, Wüst, Dockray, Smyth, Evans, Kirschbaum, Miller, Wetherell, Finke, Klucken and Clow2022). Alterations in diurnal cortisol, including flattened diurnal cortisol slope (Adam et al., Reference Adam, Quinn, Tavernier, McQuillan, Dahlke and Gilbert2017) and higher or lower levels of awakening cortisol and CAR (Boggero et al., Reference Boggero, Hostinar, Haak, Murphy and Segerstrom2017; Chida & Steptoe, Reference Chida and Steptoe2009), are associated with impaired health (e.g., depression).
Allostatic load models propose that HPA axis activation is adaptive to deal with fleeting dangers, but that chronic stress (e.g., child maltreatment) may cause repeated activation of the HPA axis, leading to dysregulated HPA axis functioning characterized by hypocortisolism or hypercortisolism (McEwen, Reference McEwen1998). Despite agreement regarding theoretical models, mixed findings have emerged in research on child maltreatment and HPA axis functioning. Even recent meta-analyses (Bernard et al., Reference Bernard, Frost, Bennett and Lindhiem2017) and systematic reviews (Holochwost et al., Reference Holochwost, Wang, Kolacz, Mills-Koonce, Klika and Jaffee2020) suggest heterogeneity in links between maltreatment and HPA axis functioning concerning whether there are associations and whether maltreatment is associated with HPA axis hyper or hypoactivity. Delving deeper into characteristics of the adversity (e.g., abuse versus neglect exposure), examining multiple cortisol indicators (e.g., awakening cortisol, CAR, diurnal cortisol slope), and exploring individual differences in these associations (e.g., moderating factors such as resilience and sex differences) may provide a more nuanced picture of how child maltreatment affects HPA axis functioning.
Prior approaches that assume the effects of different adversities are equal and additive (e.g., Evans et al., Reference Evans, Li and Whipple2013) may be incomplete. These approaches acknowledge the co-occurrence of various adversities, but a focus on the total count of adverse experiences regardless of adversity type may preclude investigation of the unique contributions of different types of adversity (McLaughlin & Sheridan, Reference McLaughlin and Sheridan2016; McLaughlin et al., Reference McLaughlin, Sheridan, Humphreys, Belsky and Ellis2021). Adversity type has been proposed to be critical to delineate the associations between childhood adversity and HPA axis functioning (Miller et al., Reference Miller, Chen and Zhou2007), however, prior research often lacks a theoretical rationale for how to categorize adversity. A recent framework, the dimensional approach to adversity, deconstructs and distinguishes adversity as threat (i.e., presence of harm or threat of harm, such as abuse) versus deprivation (i.e., absence of expected environmental inputs, such as neglect), and argues that threat and deprivation may have distinct effects on neurophysiology (McLaughlin & Sheridan, Reference McLaughlin and Sheridan2016; McLaughlin et al., Reference McLaughlin, Sheridan and Lambert2014). This model has shown value in research on neural development (McLaughlin et al., Reference McLaughlin, Weissman and Bitrán2019), biological aging (Colich et al., Reference Colich, Rosen, Williams and McLaughlin2020), and executive functioning (Johnson et al., Reference Johnson, Policelli, Li, Dharamsi, Hu, Sheridan, McLaughlin and Wade2021), and may be a promising emerging direction in research on adversity and stress physiology.
A handful of empirical studies on stress and HPA axis functioning have applied the dimensional approach and yielded mixed findings. Some have found that threat but not deprivation was associated with adolescents’ blunted cortisol stress responses (Busso et al., Reference Busso, McLaughlin and Sheridan2017; Peckins et al., Reference Peckins, Roberts, Hein, Hyde, Mitchell, Brooks-Gunn, McLanahan, Monk and Lopez-Duran2020). A study on hair cortisol showed that harsh parenting (i.e., threat) from ages 1 to 15 years, and particularly at 15 years, was associated with higher levels of hair cortisol at 15 years, whereas parental disengagement (i.e., deprivation) from ages 1 to 15 years and especially at 1 year was associated with lower levels of hair cortisol at 15 years (Doom et al., Reference Doom, Peckins, Hein, Dotterer, Mitchell, Lopez-Duran, Brooks-Gunn, McLanahan, Hyde, Abelson and Monk2020). Prior research on morning cortisol demonstrated that threat but not deprivation was indirectly associated with higher morning cortisol levels, but only in females and not males (LoPilato et al., Reference LoPilato, Addington, Bearden, Cadenhead, Cannon, Cornblatt, Mathalon, McGlashan, Perkins, Tsuang, Woods and Walker2020). A more recent study investigated how type, timing, and severity of adversity contributed to alterations in multiple diurnal cortisol indicators in late adolescence, which revealed a complicated picture of the relations among adversity and diurnal cortisol (Kessler et al., Reference Kessler, Vrshek-Schallhorn, Mineka, Zinbarg, Craske and Adam2021). Most pertinent to the present study, Kessler et al. (Reference Kessler, Vrshek-Schallhorn, Mineka, Zinbarg, Craske and Adam2021) found that major childhood neglect was associated with flatter diurnal cortisol slope and lower waking cortisol levels in adolescents, whereas major childhood physical abuse was associated with higher waking cortisol. Though complex and focusing on different aspects of HPA axis functioning, such findings may help researchers to hypothesize about whether dimensions of adversity are uniquely related to HPA axis functioning (Busso et al., Reference Busso, McLaughlin and Sheridan2017; Peckins et al., Reference Peckins, Roberts, Hein, Hyde, Mitchell, Brooks-Gunn, McLanahan, Monk and Lopez-Duran2020), whether threat and deprivation are associated with different cortisol indicators (Kessler et al., Reference Kessler, Vrshek-Schallhorn, Mineka, Zinbarg, Craske and Adam2021), and whether the direction of associations are distinct (Doom et al., Reference Doom, Peckins, Hein, Dotterer, Mitchell, Lopez-Duran, Brooks-Gunn, McLanahan, Hyde, Abelson and Monk2020). Developmental timing (Doom et al., Reference Doom, Peckins, Hein, Dotterer, Mitchell, Lopez-Duran, Brooks-Gunn, McLanahan, Hyde, Abelson and Monk2020) and child sex (LoPilato et al., Reference LoPilato, Addington, Bearden, Cadenhead, Cannon, Cornblatt, Mathalon, McGlashan, Perkins, Tsuang, Woods and Walker2020) may also be important when testing these associations.
Longitudinal changes in cortisol during the transition from childhood to adolescence
HPA axis functioning undergoes developmental changes (Koss & Gunnar, Reference Koss and Gunnar2018). Exploring trends in cortisol over time can reveal individual differences and facilitate understanding about developmental stability and change in the HPA axis (Adam & Kumari, Reference Adam and Kumari2009; Shirtcliff et al., Reference Shirtcliff, Allison, Armstrong, Slattery, Kalin and Essex2012). The transition from childhood to adolescence, accompanied by dramatic biological and psychosocial changes, is a sensitive period for the development and recalibration of the HPA axis (Gunnar, Reference Gunnar2021). The mechanism of adrenarche during the transition from childhood to adolescence contributes to salient endocrine and neurobiological fluctuations during this period (Del Giudice et al., Reference Del Giudice, Ellis and Shirtcliff2011). Investigation of within-individual development of the HPA axis during this period may help explain how the HPA axis matures and prepares the body for the transition to adolescence. Moreover, the transition to adolescence constitutes a sensitive period for the onset of psychopathology (Powers & Casey, Reference Powers and Casey2015). Given that the HPA axis is sensitive to adversity and closely related to health problems, testing the relations between adversity and changes in cortisol over time lays the foundation for future research on the emergence and stability of adversity-related health problems.
Research examining stability in diurnal cortisol over time found that diurnal cortisol slope was relatively stable over time, whereas awakening cortisol and CAR showed more temporal variations, and boys’ diurnal cortisol rhythm displayed more temporal stability than that of girls (Kuhlman et al., Reference Kuhlman, Robles, Dickenson, Reynolds and Repetti2019). A six-year longitudinal study found decreased awakening cortisol and flattened diurnal cortisol slope from 9 to 15 years, and compared to boys, girls showed less decreases in awakening cortisol and more flatness in diurnal cortisol slope over time (Shirtcliff et al., Reference Shirtcliff, Allison, Armstrong, Slattery, Kalin and Essex2012). Another study focused on CAR found increases in CAR over three years in adolescents (the mean age was 15 years at baseline), and that girls’ CAR was consistently higher than boys’ CAR (Platje et al., Reference Platje, Vermeiren, Branje, Doreleijers, Meeus, Koot, Frijns, van Lier and Jansen2013). A recent study on Chinese children 7–9 years old at baseline found that boys’ awakening cortisol decreased over three years, whereas boys’ CAR and diurnal cortisol slope, and girls’ awakening cortisol, CAR, and diurnal cortisol slope, were stable across time (Zhang et al., Reference Zhang, Duan, Wan, Zhang, Su, Tao and Sun2020). Though these extant findings are mixed and focus on samples from different cultural contexts with various age ranges, they provide critical insights that indicators of diurnal cortisol may display longitudinal changes and that sex-differentiated development is consistently observed.
A few studies have examined the associations between adversity and within-child development of diurnal cortisol. A 19-year longitudinal study on sexually abused females (6–16 years old at baseline) suggested that abused females showed slower increases in morning cortisol than non-abused females (Trickett et al., Reference Trickett, Noll, Susman, Shenk and Putnam2010). Another study indicated more variations in cortisol levels across 6 years in foster care children (3–6 years old at baseline) than their counterparts (Laurent et al., Reference Laurent, Gilliam, Bruce and Fisher2014). A recent study on previously institutionalized (PI) children (7–15 years old at baseline) found increased CAR and flattened diurnal cortisol slope in PI children over time, but no associations between adversity and longitudinal diurnal cortisol (Reid et al., Reference Reid, DePasquale, Donzella, Leneman, Taylor and Gunnar2021). Interestingly, another longitudinal study revealed that the effects of adversity on cortisol development may change over time (VanTieghem et al., Reference VanTieghem, Korom, Flannery, Choy, Caldera, Humphreys, Gabard-Durnam, Goff, Gee, Telzer, Shapiro, Louie, Fareri, Bolger and Tottenham2021). Specifically, the comparison but not PI children showed age-related increases in morning cortisol before age 13, whereas age-related increases in morning cortisol were observed in the PI but not the comparison group after age 13 (VanTieghem et al., Reference VanTieghem, Korom, Flannery, Choy, Caldera, Humphreys, Gabard-Durnam, Goff, Gee, Telzer, Shapiro, Louie, Fareri, Bolger and Tottenham2021). The limited amount of research, focusing on different diurnal cortisol indicators and different types of adversity, may hamper the ability to draw conclusions about how adversity is related to developmental changes in diurnal cortisol. Nevertheless, these findings highlight the critical role of adversity in shaping HPA axis development and suggest that including more diurnal cortisol indicators may provide a more comprehensive and nuanced understanding of this process.
The moderating roles of child resilience and sex differences
In spite of extensive literature on child maltreatment and HPA axis functioning, studies of individual differences in these associations are relatively sparse. Individuals exposed to adversity may not experience the same pattern of later outcomes. Multifinality, defined as varied outcomes following the same risk factor, is one of the central propositions in developmental psychopathology and applies to the study of individual differences in the negative health sequelae of adversity such as child maltreatment (Cicchetti & Rogosch, Reference Cicchetti and Rogosch2002). Identifying protective factors in the associations between child maltreatment and stress physiology may help illustrate how individuals adapt to stress and provide implications for child maltreatment intervention (Masten, Reference Masten2018). Investigating if the adversity-physiology link differs as a function of demographic variables (e.g., child sex) may also provide a more precise understanding about vulnerable and resilient individuals in the context of adversity.
Resilience involves the processes of adaptation or positive outcomes in the face of adversity (Luthar et al., Reference Luthar, Cicchetti and Becker2000; Masten, Reference Masten2018), and, among other definitions, has been defined at the individual level as a trait-like ability to cope with, adapt to, or recover from stress (Connor & Davidson, Reference Connor and Davidson2003). This study operationalizes resilience at the individual level, which emphasizes personal strengths in the context of adversities (Connor & Davidson, Reference Connor and Davidson2003). Research on resilience has overturned prior deficient-focused models that argued that children experiencing adversity were bound to develop negative outcomes. Several studies focused on the relation between resilience and diurnal cortisol have found direct effects of resilience (i.e., individuals’ capabilities to adapt to adversity) on healthier diurnal cortisol rhythms (Chi et al., Reference Chi, Slatcher, Li, Zhao, Zhao, Ren, Zhu and Stanton2015). Further, individual resilience can serve as a moderator in the association between stress and HPA axis functioning. Prior findings revealed that ego-resiliency (i.e., a personality construct that refers to individuals’ abilities to adapt to stress) buffered the association between negative parent-child interactions and young children’s higher cortisol reactivity (Smeekens et al., Reference Smeekens, Marianne Riksen-Walraven and van Bakel2007). Psychological resilience (i.e., individuals’ capacities to adapt to stressful and challenging situations) was also found to weaken the relation between perceived stress and higher hair cortisol in adults (Lehrer et al., Reference Lehrer, Steinhardt, Dubois and Laudenslager2020). Nevertheless, whether resilience can buffer the associations between child maltreatment and child and adolescent diurnal cortisol remains unknown. The transition from childhood to adolescence is also conceptualized as a window of opportunity to promote health and for the stress physiology system to recover from stress (Gunnar et al., Reference Gunnar, DePasquale, Reid, Donzella and Miller2019). Therefore, this period can be a salient developmental stage in which to test protective factors in the association between child maltreatment and stress physiology, which may have profound implications for interventions aiming at minimizing the adverse effects of abuse and neglect.
Sex differences in the association between adversity and HPA axis are proposed by theories in the field of stress physiology (Del Giudice et al., Reference Del Giudice, Ellis and Shirtcliff2011; Koss & Gunnar, Reference Koss and Gunnar2018), but still need to be explicitly tested in empirical studies. Due to the fluctuations of sex hormones and the interaction between the HPA axis and hypothalamic-pituitary-gonadal axis, sex differences in HPA axis functioning start to emerge and are consolidated during the transition from childhood to adolescence (Koss & Gunnar, Reference Koss and Gunnar2018). A meta-analysis of 81 studies found that compared to boys’, girls’ CAR was higher and diurnal cortisol slope was steeper (Hollanders et al., Reference Hollanders, van der Voorn, Rotteveel and Finken2017). The development of HPA axis functioning from childhood and adolescence also differs in boys and girls, though the specific patterns are not entirely clear (Platje et al., Reference Platje, Vermeiren, Branje, Doreleijers, Meeus, Koot, Frijns, van Lier and Jansen2013; Shirtcliff et al., Reference Shirtcliff, Allison, Armstrong, Slattery, Kalin and Essex2012; Zhang et al., Reference Zhang, Duan, Wan, Zhang, Su, Tao and Sun2020). Moreover, vulnerability and resilience to stress may differ in boys and girls, such that girls’ diurnal cortisol may be more likely to be affected by stressors such as low socioeconomic status (Wright & Bukowski, Reference Wright and Bukowski2021) and threat-related adversity (LoPilato et al., Reference LoPilato, Addington, Bearden, Cadenhead, Cannon, Cornblatt, Mathalon, McGlashan, Perkins, Tsuang, Woods and Walker2020). Examining the associations between child maltreatment and diurnal cortisol and its development, and the moderating role of resilience in boys and girls separately, may help provide a more nuanced picture of how boys and girls physiologically adapt to stress.
It is worth noting that there is a lack of research on child maltreatment and stress physiology in non-Western samples, which limits the ability to generalize prior findings to children and adolescents in non-Western cultures. Parenting practices are embedded in cultural contexts and thus may lead to variations in the effects of child maltreatment in different cultures. Western cultures usually encourage more negotiation and expression of positive affect in parent-child relationships, whereas traditional Asian cultures emphasize more parental authority (Chen et al., Reference Chen, Fu and Yiu2019). In traditional Chinese cultures, children are expected to show the utmost respect and obedience to parents’ authority, and parents’ punitive actions to correct children’s behaviors are accepted and perceived as indicators of parental care and love, although they may lead to child abuse (Chen et al., Reference Chen, Fu and Yiu2019). Also, Chinese parents typically place high demands on children’s academic performance but are likely to overlook children’s psychological needs, which may contribute to higher levels of child neglect (Wang et al., Reference Wang, Cheng, Qu, Zhang, Cui and Zou2020). Furthermore, Chinese collectivism values familism, such that the family is the basic structural and functional unit and constitutes the self (Kwok et al., Reference Kwok, Chai and He2013), which may make family-related stress more distressing for children. In the present study, using a sample of Chinese children from a disadvantaged area builds upon prior research that has been predominantly conducted in Western samples.
The present study
Extant research has documented the detrimental effects of child maltreatment on stress physiology. Nevertheless, the empirical findings on child maltreatment and diurnal cortisol vary regarding the magnitude and direction of effects, as well as the cortisol indicators investigated (Bernard et al., Reference Bernard, Frost, Bennett and Lindhiem2017). Examining specific child maltreatment dimensions and including multiple diurnal cortisol indicators that reflect unique and complementary aspects of diurnal cortisol may benefit a more precise and comprehensive understanding of maltreatment-diurnal cortisol relations. Most of the prior research on child maltreatment and diurnal cortisol has focused on diurnal cortisol at one time point. Though the broader literature on adversity and HPA axis functioning over time has provided evidence that adversity may affect the longitudinal development of the HPA axis (e.g., VanTieghem et al., Reference VanTieghem, Korom, Flannery, Choy, Caldera, Humphreys, Gabard-Durnam, Goff, Gee, Telzer, Shapiro, Louie, Fareri, Bolger and Tottenham2021), there is a paucity of research on child maltreatment and within-individual development in diurnal cortisol. Furthermore, individual differences in the above associations need more elaboration to facilitate the understanding of risk and resilience processes in maltreatment-cortisol relations, for example, whether resilience can moderate the relations between child abuse and neglect and diurnal cortisol, and whether these associations show diverging patterns in boys and girls.
Using a sample of Chinese children and adolescents from a disadvantaged area and building upon the dimensional approach to childhood adversity, the present study contributed to research on the effects of child maltreatment on stress physiology in different time scales by exploring the distinct effects of abuse and neglect on diurnal cortisol at baseline and longitudinal changes in diurnal cortisol over a one-year period. Multiple diurnal cortisol indicators (awakening cortisol, CAR, diurnal cortisol slope) were included to provide a more comprehensive picture of cortisol functioning during the daytime. The moderating role of resilience was also explored to test individual differences in the relations between child maltreatment and diurnal cortisol. The aforementioned research questions were examined in boys and girls separately, especially given that the transition from childhood to adolescence marks a period of the emergence of sex differences in stress physiology (Koss & Gunnar, Reference Koss and Gunnar2018).
Based on the literature reviewed above, the present study hypothesized that the effects of child abuse and neglect on diurnal cortisol might be distinct concerning the direction of associations, whether they are related to different diurnal cortisol indicators, and whether they are related to diurnal cortisol indicators on different time scales (e.g., cortisol at the baseline assessment, longitudinal changes in cortisol). Because prior findings on child maltreatment and diurnal cortisol are mixed and research on longitudinal changes in diurnal cortisol is scarce, the current hypotheses were partially exploratory, such that we did not make a priori hypotheses regarding the specific directions of these associations between child abuse and neglect and cortisol or the effects on specific diurnal cortisol indicators or time scales. Resilience was hypothesized to moderate the associations between abuse and neglect and diurnal cortisol and its longitudinal changes as a stress buffer, such that the effects of child abuse and neglect on cortisol would be more apparent in children and adolescents with lower levels of resilience. Furthermore, associations between child maltreatment and diurnal cortisol and their longitudinal changes were hypothesized to be more observable in girls compared to boys, since prior studies found that stress was more likely to influence diurnal cortisol in girls compared to boys (LoPilato et al., Reference LoPilato, Addington, Bearden, Cadenhead, Cannon, Cornblatt, Mathalon, McGlashan, Perkins, Tsuang, Woods and Walker2020; Wright & Bukowski, Reference Wright and Bukowski2021). Whether and how moderation effects of resilience would vary by sex remained exploratory.
Methods
Participants and procedure
Data in the present study were drawn from an ongoing longitudinal study of risk and adaptation of children and adolescents from rural areas of China. Participants were recruited from two elementary schools in a disadvantaged rural area in Eastern China, and all participants in the current study lived in the school dormitories during the data collection period. The gross regional product per capita in this area (about $2,526 in 2019, the year of the baseline assessment) was far less than the gross domestic product per capita in China ($11,127 in 2019) (National Bureau of Statistics of China, 2020). In this area, a large number of parents migrated to cities for better jobs and higher payment, leading to a sizable group of children and adolescents left behind in town. At the baseline assessment (December 2019; Wave 1, W1), children and adolescents completed self-report questionnaires concerning child maltreatment and resilience during class sessions. Participants also provided their salivary cortisol samples three times a day for three consecutive days under the guidance of trained research assistants. Daily checklists assessing wake-up time and medication usage were collected during days of salivary collection. About one year later (November 2020; Wave 2, W2), children and adolescents provided their salivary samples and daily diaries again following the same procedures as in W1.
There were 304 participants who had valid cortisol data at W1, 269 participants who had valid cortisol data at W2, 261 participants who had valid cortisol data at both waves and 312 participants who had cortisol data at either wave. Following prior research, children and adolescents who had valid cortisol data at either wave were included (Reid et al., Reference Reid, DePasquale, Donzella, Leneman, Taylor and Gunnar2021). Therefore, the current sample consisted of 312 Chinese children and adolescents aged from 9 to 13 years in Grade 4 and Grade 5 (209 boys and 103 girls; M age = 10.80, SD = 0.84) at W1. Independent sample t tests were conducted to examine whether retained and attritted children and adolescents at W2 differed in the demographic covariates and primary study variables at W1. No differences emerged between retained and attritted children and participants in age, sex, subjective socioeconomic status (SSS), abuse, resilience, awakening cortisol, CAR, or diurnal cortisol slope at W1. Children and adolescents who did not participate at W2 reported higher levels of neglect at W1 (p = .018). Participants’ characteristics are presented in Table 1. Children and adolescents were provided with an age-appropriate gift for each wave of data collection. The protocol was approved by the university institutional review board. School administrators and head teachers in participating schools were contacted for permission for recruitment. Primary caregivers of these children and adolescents provided consent, and written informed assent was obtained from all children and adolescents.
Table 1. Descriptive characteristics of sample

a Threshold scores were based on sum scores for each subscale: physical abuse ≥ 8, emotional abuse ≥ 10, physical neglect ≥ 8, and emotional neglect ≥ 15 (Walker et al., Reference Walker, Unutzer, Rutter, Gelfand, Saunders, VonKorff, Koss and Katon1999).
b Abuse represents children who reported physical and/or emotional abuse.
c Neglect represents children who reported physical and/or emotional neglect.
Measures
Child abuse and neglect
Children and adolescents reported on their own abuse and neglect at W1 using the Childhood Trauma Questionnaire-Short Form (Bernstein et al., Reference Bernstein, Stein, Newcomb, Walker, Pogge, Ahluvalia and Zule2003), which has demonstrated good psychometric properties among Chinese children and adolescents (Zhao et al., Reference Zhao, Zhang, Li, Zhou, Li and Yang2005). The current study included emotional abuse (5 items, e.g., “I felt that someone in my family hated me”), emotional neglect (5 items, reverse-worded; e.g., “I felt loved”), physical abuse (5 items, e.g., “People in my family hit me so hard that it left me with bruises or marks”), and physical neglect (5 items, e.g., “I do not have enough to eat”) subscales. Items were rated on a 5-point scale (1 = never, 5 = always). Due to the interest in examining the effects of abuse versus neglect, an abuse composite score was created by averaging the 10 items from the emotional and physical abuse subscales, and a neglect composite score was created by averaging the 10 items from the emotional and physical neglect subscales. Higher scores reflected higher levels of abuse or neglect. In this study, the Cronbach’s alpha for abuse was .87 for both boys and girls; the Cronbach’s alpha for neglect was .75 for boys and .76 for girls.
Resilience
Resilience was assessed at W1 using the Connor-Davidson Resilience Scale (Campbell-Sills & Stein, Reference Campbell-Sills and Stein2007; Connor & Davidson, Reference Connor and Davidson2003), which has demonstrated good psychometric properties among Chinese samples (Cao et al., Reference Cao, An, Yang, Peng, Xu, Xu and Xiang2020). This scale consisted of 10 items (e.g., “Can achieve goals despite obstacles”), rated on a 5-point scale (ranging from 1 = never to 5 = always). In this study, the Cronbach’s alpha was .87 for boys and .90 for girls.
Salivary cortisol
During both W1 and W2 assessments, children and adolescents were instructed to provide their salivary cortisol samples on three consecutive days immediately upon awakening, 30 minutes after awakening, and at bedtime. Participants were instructed to refrain from eating, drinking, and using tobacco for at least 30 minutes prior to the salivary collection. None of the participants reported that they were taking medications that are shown to influence cortisol levels (e.g., steroid medicines; Granger et al., Reference Granger, Hibel, Fortunato and Kapelewski2009). The morning samples (upon awakening, 30 minutes after awakening) were collected at dormitories and the evening samples (bedtime) were collected at classrooms. Participants went back to their dormitories to sleep immediately after the evening sample collection. One day prior to the start of saliva sample collection, the correct procedures of saliva sample collection were shown to all participants. Trained research assistants supervised the sample collection procedures and recorded the sample collection times. At the W2 assessment, a small proportion of adolescents (14 participants out of 269 participants at W2; 5.2%) no longer lived in the dormitories, and they collected saliva samples themselves at home. A research assistant sent reminders to these participants at each scheduled sample collection time to facilitate compliance, and gathered their saliva samples at school the morning following their saliva sample collection. The raw cortisol values and the three cortisol indicators at W2 did not differ in school- and home-collected samples (ps > .05; see Table S1 in the supplementary materials).
The Salivette sampling device (SARSTEDT, Germany) was used to collect saliva samples. Saliva samples were stored at –25°C immediately after collection and sent on ice to the Beijing Protein Innovation Co. Ltd., Beijing, China, to be assayed. Free cortisol concentrations were assayed in singlets using a commercially available enzyme-linked immunosorbent assay (DRG Inc., Germany). The intra- and inter-assay coefficients of variance provided by the manufacturer were less than 10%. Outliers (more than three standard deviations above or below the mean) were winsorized to three standard deviations above or below the mean at each time point [41 samples (1.54%) were affected at W1, and 30 samples (1.30%) were affected at W2]. To accurately examine awakening cortisol and CAR, time compliance for these samples was examined. Samples taken more than 10 minutes earlier or later than the requested collection time or time interval [39 samples (1.48%) at W1 and 39 samples (1.70%) at W2] were considered noncompliant and were excluded in the current analyses (Adam et al., Reference Adam, Hawkley, Kudielka and Cacioppo2006). A total of 4,808 (2,578 at W1 and 2,230 at W2) valid salivary cortisol samples from 312 participants were included in the current analyses. Raw cortisol values were natural log-transformed prior to statistical analysis to account for the positive skew (Adam & Kumari, Reference Adam and Kumari2009). A constant of one was added to cortisol values before the transformation to ensure that all transformed cortisol values were positive.
Covariates
Children and adolescents’ age and SSS at W1 (person level) were included as covariates, as well as wake-up time (day level) considering their possible effects on diurnal cortisol secretion (Adam & Kumari, Reference Adam and Kumari2009; Ursache et al., Reference Ursache, Noble and Blair2015). SSS was assessed using the ladder measure (Adler et al., Reference Adler, Epel, Castellazzo and Ickovics2000). Participants were asked to report how they perceive their position in the social hierarchy on a picture of a 10-rung ladder (from 1 = lowest/worst to 10 = highest/best). Wake-up times were reported by children and adolescents after every awakening saliva sample collection.
Data analytic strategies
Descriptive statistics and bivariate correlations among person-level variables and diurnal cortisol indicators in boys and girls were presented. Log-transformed cortisol values were used to calculate diurnal cortisol indicators. Awakening cortisol samples reflected awakening cortisol. Area under the curve with respect to increase (Pruessner et al., Reference Pruessner, Kirschbaum, Meinlschmid and Hellhammer2003) between the awakening sample and the 30 minute after awakening sample was calculated using log-transformed cortisol values as an index of CAR (Stalder et al., Reference Stalder, Kirschbaum, Kudielka, Adam, Pruessner, Wüst, Dockray, Smyth, Evans, Hellhammer, Miller, Wetherell, Lupien and Clow2016). Diurnal cortisol slope was computed as the simple difference divided by the total time interval (hour) between the awakening and evening samples (Adam et al., Reference Adam, Quinn, Tavernier, McQuillan, Dahlke and Gilbert2017).
Multilevel modelling (MLM) was conducted using the maximum likelihood with robust standard errors (MLR) method in Mplus 8.3 (Muthén & Muthén, Reference Muthén and Muthén2017) to test the associations between abuse and neglect and diurnal cortisol indicators and the moderating role of resilience. MLM accounts for the nested structure of longitudinal cortisol data (day nested in wave, wave nested in person) and enables modelling within-individual changes in diurnal cortisol indicators both across days and across waves. This approach also allows for including participants who missed one wave of data collection. Each diurnal cortisol indicator (awakening cortisol, CAR, diurnal cortisol slope) was tested in a separate model, and models were tested in boys and girls separately. As shown in Equation 1, at level 1 (day level), cortisol indicators were predicted by day level covariates. At level 2 (wave level), coded wave time (0 = W1, 1 = W2) was added to predict level-1 intercepts, such that the coefficient on wave time represented changes in cortisol indicators across waves. At level 3, all person-level predictors and covariates (i.e., age, SSS) were added to predict both the intercept (i.e., cortisol indicator at W1) and the coefficient on wave time (i.e., changes in cortisol indicator across waves) at level 2. Continuous predictors and covariates at the person level were grand-mean centered. Person-level predictors at level 3 included mean-centered abuse, mean-centered neglect, mean-centered resilience, mean-centered abuse × mean-centered resilience, and mean-centered neglect × mean-centered resilience. To better understand patterns of moderation effects, visualization of moderation effects was presented for significant interactions using the Johnson-Neyman technique (Bauer & Curran, Reference Bauer and Curran2005). To facilitate the interpretation of changes in cortisol indicators over time, unconditional multilevel models including wave time (0 = W1, 1 = W2) at level 2 but no other predictors or covariates were also conducted for each cortisol indicator in boys and girls. Full Information Maximum Likelihood estimation was utilized to account for missing data.
Stratified models by child sex could contribute a nuanced and precise understanding of how the effects of child abuse and neglect on diurnal cortisol and the moderating effects of resilience displayed in boys and girls, respectively. Supplemental analyses were also conducted to facilitate the understanding of the statistical significance of potential sex differences. Specifically, multilevel models for the full sample (N = 312) were performed with each of the cortisol indicators, and child sex as well as its interaction terms (i.e., dummy coded sex, mean-centered abuse × dummy coded sex, mean-centered neglect × dummy coded sex, mean-centered resilience × dummy coded sex, mean-centered abuse × mean-centered resilience × dummy coded sex, mean-centered neglect × mean-centered resilience × dummy coded sex) were added as predictors at level 3 in addition to predictors and covariates in multilevel models for boys and girls.
 $$\matrix{ {{\rm{Level }}\ 1{\rm{ }}\ \left( {{\rm{day \ level}}} \right)} \hfill \cr {{\rm{Cortisol \ indicato}}{{\rm{r}}_{{\rm{ij}}}} = {\rm{{\pi}} _{0i{\rm{j}}}} + {\rm{{\pi}} _{1i{\rm{j}}}}\left( {{\rm{day}} \hbox- {\rm{level \ covariates}}} \right) + {{\rm{e}}_{{\rm{tij}}}}} \hfill \cr {{\rm{Level }}\ 2\ \left( {{\rm{wave \ level}}} \right)} \hfill \cr {{{\rm{\pi}} _{0ij}} = {\rm{{\beta}} _{00j}} + {\rm{{\beta}} _{0ij}}\left( {{\rm{wave \ time}}} \right){\rm{ }} + {\rm{{r}}_{0ij}}} \hfill \cr {{\rm{Level}}\ 3{\rm{ }\ }\left( {{\rm{person \ level}}} \right)} \hfill \cr \matrix{ {\rm{\beta} _{00j}} = {\rm{{\gamma}} _{000}} + {\rm{{\gamma}} _{00j}}\left( {{\rm{person}} \hbox- {\rm{level \ variables}}} \right){\rm{ }} + {\rm{{\mu}} _{00j}} \hfill \cr {\rm{\beta} _{0ij}} = {\rm{{\gamma}} _{100}} + {\rm{{\gamma}} _{10j}}\left( {{\rm{person}} \hbox- {\rm{level \ variables}}} \right){\rm{ }} + {\rm{{\mu}} _{10j}} \hfill \cr} \hfill \cr } $$
$$\matrix{ {{\rm{Level }}\ 1{\rm{ }}\ \left( {{\rm{day \ level}}} \right)} \hfill \cr {{\rm{Cortisol \ indicato}}{{\rm{r}}_{{\rm{ij}}}} = {\rm{{\pi}} _{0i{\rm{j}}}} + {\rm{{\pi}} _{1i{\rm{j}}}}\left( {{\rm{day}} \hbox- {\rm{level \ covariates}}} \right) + {{\rm{e}}_{{\rm{tij}}}}} \hfill \cr {{\rm{Level }}\ 2\ \left( {{\rm{wave \ level}}} \right)} \hfill \cr {{{\rm{\pi}} _{0ij}} = {\rm{{\beta}} _{00j}} + {\rm{{\beta}} _{0ij}}\left( {{\rm{wave \ time}}} \right){\rm{ }} + {\rm{{r}}_{0ij}}} \hfill \cr {{\rm{Level}}\ 3{\rm{ }\ }\left( {{\rm{person \ level}}} \right)} \hfill \cr \matrix{ {\rm{\beta} _{00j}} = {\rm{{\gamma}} _{000}} + {\rm{{\gamma}} _{00j}}\left( {{\rm{person}} \hbox- {\rm{level \ variables}}} \right){\rm{ }} + {\rm{{\mu}} _{00j}} \hfill \cr {\rm{\beta} _{0ij}} = {\rm{{\gamma}} _{100}} + {\rm{{\gamma}} _{10j}}\left( {{\rm{person}} \hbox- {\rm{level \ variables}}} \right){\rm{ }} + {\rm{{\mu}} _{10j}} \hfill \cr} \hfill \cr } $$
Results
Preliminary analyses
Descriptive statistics and bivariate correlations among person-level variables and diurnal cortisol indicators in boys and girls are displayed in Table 2. Density plots that illustrate child abuse and neglect data distributions are presented in Figure S1 in the supplementary materials. In both boys and girls, child abuse was positively associated with neglect, and neglect but not abuse was associated with lower levels of resilience. Abuse was associated with lower CAR and steeper diurnal cortisol slope at W2 in girls, but not associated with any cortisol indicators in boys. Neglect was associated with higher awakening cortisol and lower CAR at W1 in girls, and lower CAR at W1 in boys.
Table 2. Descriptive statistics and bivariate correlations for person-level variables and diurnal cortisol indicators
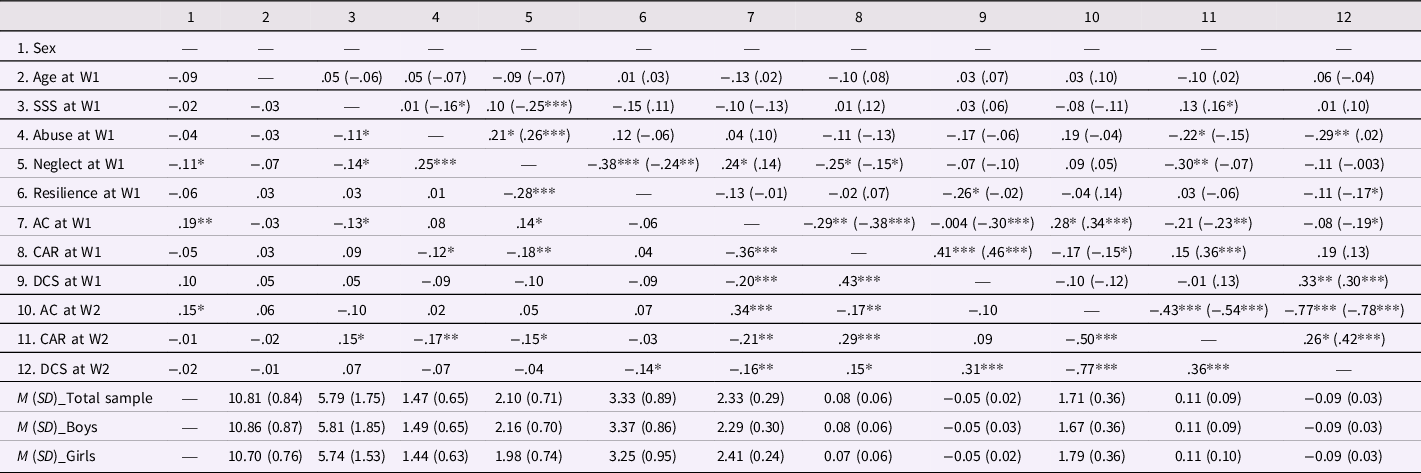
AC = awakening cortisol; CAR = cortisol awakening response; DCS = diurnal cortisol slope; SSS = subjective socioeconomic status.
AC, CAR, and DCS were calculated using transformed cortisol values and mean scores were for cortisol indicators across three days within each wave were used in this table. Sex was dummy coded as 0 = boy, 1 = girl. Correlations above diagonal are for boys (n = 209; in brackets) and girls (n = 103), and correlations below diagonal are for the total sample (N = 312).
*p < .05.
**p < .01.
*** p < .001.
Primary analyses
Among both boys and girls, all three diurnal cortisol indicators changed significantly during the one-year period (Table 3). Awakening cortisol decreased, CAR increased, and diurnal cortisol slope became steeper from W1 to W2. Results of MLM in boys and girls are displayed in Tables 4 and 5, respectively. In examining the effects of abuse versus neglect on diurnal cortisol, no significant associations were found between abuse or neglect and cortisol indicators at W1 or changes in cortisol indicators from W1 to W2 in boys (Table 4). However, in girls, neglect was associated with higher awakening cortisol and lower CAR at W1. Additionally, abuse was associated with girls’ changes in cortisol over time such that abuse was associated with less decreases in awakening cortisol, less increases in CAR, and more steepness in diurnal cortisol slope from W1 to W2 (Table 5). These results indicated that in girls, neglect was associated with alterations in baseline diurnal cortisol (i.e., awakening cortisol and CAR at W1), whereas abuse was associated with changes in diurnal cortisol over time (i.e., longitudinal changes in awakening cortisol, CAR, and diurnal cortisol slope).
Table 3. Multilevel models for changes in diurnal cortisol indicators from W1 to W2
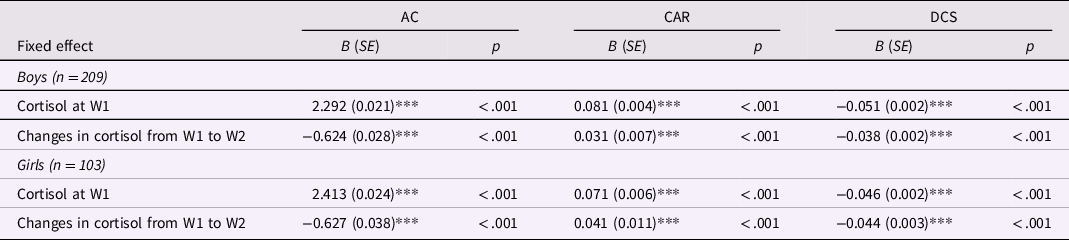
AC = awakening cortisol; CAR = cortisol awakening response; DCS = diurnal cortisol slope.
*p < .05.
**p < .01.
***p < .001.
Table 4. Results of multilevel models for the moderation effects of resilience in the associations between abuse and neglect on diurnal cortisol in boys (n = 209)
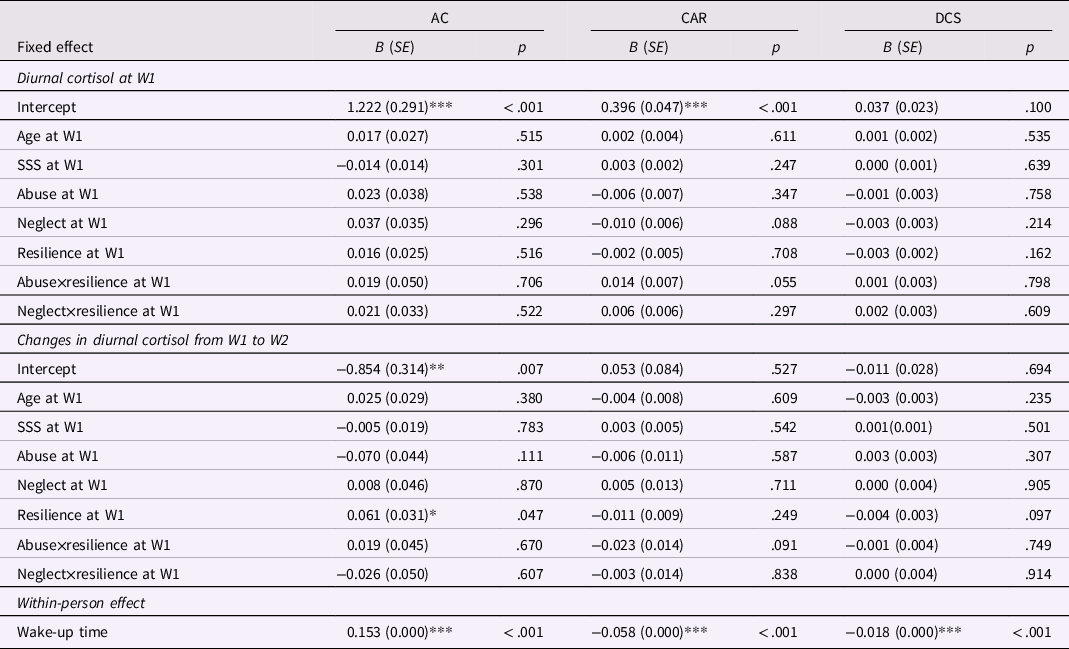
AC = awakening cortisol; CAR = cortisol awakening response; DCS = diurnal cortisol slope; SSS = subjective socioeconomic status.
*p < .05.
**p < .01.
***p < .001.
Table 5. Results of multilevel models for the moderation effects of resilience in the associations between abuse and neglect on diurnal cortisol in girls (n = 103)
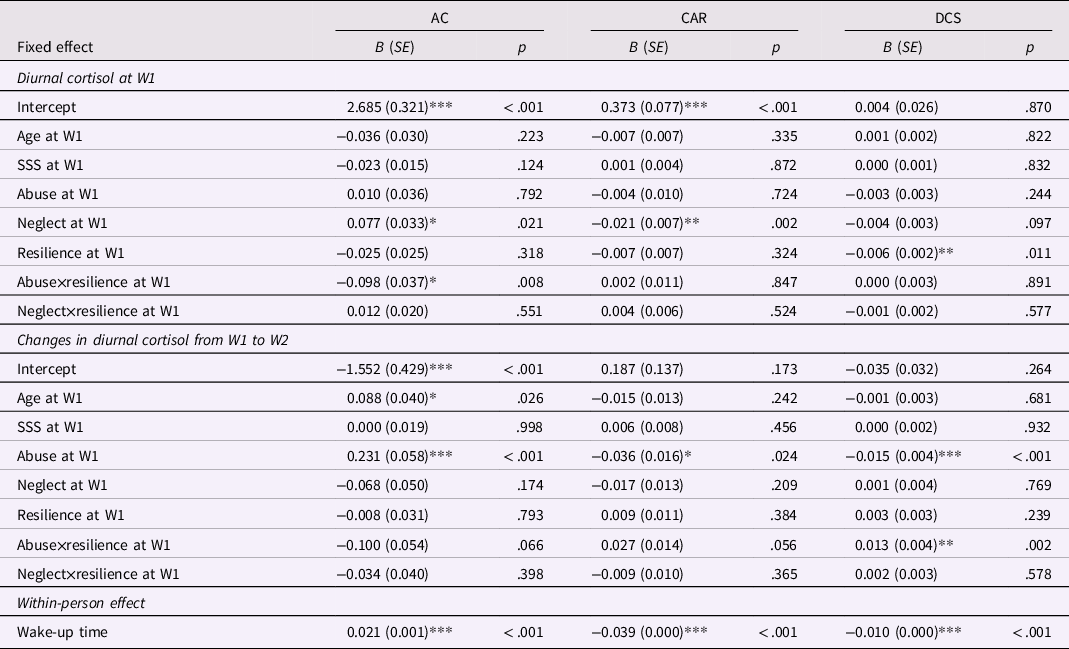
AC = awakening cortisol; CAR = cortisol awakening response; DCS = diurnal cortisol slope; SSS = subjective socioeconomic status.
*p < .05.
**p < .01.
***p < .001.
Concerning the moderating effects of resilience on relations between abuse and neglect and diurnal cortisol, no significant moderation effects were observed in boys. In girls, resilience moderated the association between abuse and awakening cortisol at W1, as well as the association between abuse and changes in diurnal cortisol slope from W1 to W2. The visualization of these observed interaction effects in girls based on the Johnson-Neyman technique is depicted in Figure 1a and b. The patterns of these two moderating effects were different. Abuse was associated with higher awakening cortisol at W1 for girls with resilience levels under 1.18 SD below the mean (about 12% of girls), but was associated with lower awakening cortisol at W1 for girls with resilience levels over 0.93 SD above the mean (about 18% of girls). The association between abuse and changes in girls’ more steepness in diurnal cortisol slope over time was significant for girls with resilience levels under 0.63 SD above the mean (about 68% of girls). These results indicated both the possible benefits and costs of resilience in associations between abuse and diurnal cortisol in girls. Specifically, these two associations between abuse and diurnal cortisol were not evident for girls with moderate levels of resilience, whereas abuse was found to be associated with blunted awakening cortisol at W1 for girls with the highest levels of resilience.

Figure 1. In total, 95% confidence bands for observed sample values of resilience for the unstandardized effects of abuse on awakening cortisol at W1 (a) and the effects of abuse on changes in diurnal cortisol slope from W1 to W2 (b) in girls. The range of girls’ grand-mean-centered resilience was -2.25 ∼ 1.75 in this study. At lower levels of resilience, abuse was associated with higher awakening cortisol at W1 (a; shaded area to the left of the dashed line and about 12% girls) and more steepness in diurnal cortisol slope from W1 to W2 (b; over about one year; shaded area to the left of the dashed line and about 68% girls). At higher levels of resilience, abuse was associated with lower awakening cortisol at baseline (A; shaded area to the right of the right dashed line and about 18% girls) but was no longer associated with changes in diurnal cortisol slope from W1 to W2 (area to the right of the dashed line).
Supplemental analyses (see Table S2 in the supplementary materials) showed statistically significant sex differences in the associations between abuse and changes in awakening cortisol and diurnal cortisol slope over time, as well as the moderation effect of resilience in the link between abuse and changes in diurnal cortisol slope over time. Though the effects of neglect on awakening cortisol and CAR at baseline and the moderation effect of resilience in the link between abuse and awakening cortisol at baseline were only found in girls but not in boys, sex differences in these associations were not statistically significant. Also, though the moderating effect of resilience in relations between abuse and changes in CAR over time significantly varied by sex, this effect was not significant in boys or girls, thus no further interpretations concerning sex differences in this effect were made.
Synthesizing the aforementioned results, the current findings revealed that abuse and neglect may be related to girls’ diurnal cortisol in distinct ways. First, abuse and neglect were associated with girls’ diurnal cortisol at different time scales. Neglect was related to girls’ baseline diurnal cortisol (at W1) but not changes in diurnal cortisol over time (during the one-year period from W1 to W2), whereas abuse was linked to girls’ changes in cortisol over time. Second, considering differential outcomes by type of cortisol indicator, both abuse and neglect were associated with girls’ awakening cortisol and CAR (abuse was associated with changes in awakening cortisol and CAR over time, and neglect was associated with awakening cortisol and CAR at W1), whereas only abuse but not neglect was associated with girls’ diurnal cortisol slope (changes over time). Moreover, the moderating effects of resilience were found in links between abuse and awakening cortisol at W1 and longitudinal changes in diurnal cortisol slope in girls, indicating both the potential benefits and costs of resilience. Finally, no significant associations between abuse and neglect and cortisol indicators were found in boys. The statistical significance of sex differences should be interpreted with caution due to considerations of analytic power when including child sex as a statistical moderator.
Discussion
Accumulating evidence has indicated that child maltreatment alters HPA axis functioning. However, recent meta-analyses (e.g., Bernard et al., Reference Bernard, Frost, Bennett and Lindhiem2017) and systematic reviews (e.g., Holochwost et al., Reference Holochwost, Wang, Kolacz, Mills-Koonce, Klika and Jaffee2020) revealed the intricate associations between child maltreatment and stress physiology, indicating the inherent complexities in clearly defining these associations. The present study attempted to unpack some of this complexity. Among a sample of Chinese children and adolescents from a disadvantaged area, and building upon the dimensional approach to childhood adversity (McLaughlin & Sheridan, Reference McLaughlin and Sheridan2016; McLaughlin et al., Reference McLaughlin, Sheridan and Lambert2014), this study revealed distinct effects of child abuse versus neglect on diurnal cortisol at the baseline assessment and longitudinal changes in diurnal cortisol, with findings varying by cortisol measure and time scale of relations. The moderating effects of resilience in the relations between abuse and diurnal cortisol were also identified. These above associations were found only in girls but not in boys.
The present study found that, in both boys and girls, cortisol circadian rhythms were more apparent over time such that awakening cortisol decreased, CAR increased, and diurnal cortisol slope became steeper during the one-year period. These results were consistent with prior research suggesting a decrease in awakening cortisol over time during preadolescence (Shirtcliff et al., Reference Shirtcliff, Allison, Armstrong, Slattery, Kalin and Essex2012) and the pubertal transition (Zhang et al., Reference Zhang, Duan, Wan, Zhang, Su, Tao and Sun2020), and an increase in CAR across adolescence (Platje et al., Reference Platje, Vermeiren, Branje, Doreleijers, Meeus, Koot, Frijns, van Lier and Jansen2013). These temporal and within-individual changes in diurnal cortisol may reflect the maturation of the HPA axis, which helps prepare children with metabolic resources for the transition to adolescence (Leneman et al., Reference Leneman, Donzella, Desjardins, Miller and Gunnar2018). The steepness of diurnal cortisol slope over time found in this study did not parallel existing findings that diurnal cortisol slope tended to be flatter in samples from Western cultures (Reid et al., Reference Reid, DePasquale, Donzella, Leneman, Taylor and Gunnar2021; Shirtcliff et al., Reference Shirtcliff, Allison, Armstrong, Slattery, Kalin and Essex2012) or was relatively stable in Chinese children (Zhang et al., Reference Zhang, Duan, Wan, Zhang, Su, Tao and Sun2020) across time. Considering that the children and adolescents in this study are from a rural and economically disadvantaged area in China, one interpretation of this finding is that the development of HPA axis functioning may vary by socioeconomic status (Dowd et al., Reference Dowd, Simanek and Aiello2009) or culture (Causadias & Cicchetti, Reference Causadias and Cicchetti2018). For example, in addition to the increasing academic and interpersonal challenges of the transition to adolescence in general, these children from a rural and economically disadvantaged area in China may also be expected to take more household duties as they become adolescents. Steepness in diurnal cortisol slope, which features a more apparent cortisol rhythm and HPA axis activation across the day, may have adaptive values for these children to adjust to expanding expectations and demands.
In girls, neglect was associated with higher awakening cortisol and lower CAR at the baseline assessment. Awakening cortisol and CAR are typically inversely related (Stalder et al., Reference Stalder, Kirschbaum, Kudielka, Adam, Pruessner, Wüst, Dockray, Smyth, Evans, Hellhammer, Miller, Wetherell, Lupien and Clow2016) and thus the effects of neglect found in the present study are consistent with prior research revealing a negative association between deprivation experiences and CAR (Leneman et al., Reference Leneman, Donzella, Desjardins, Miller and Gunnar2018; Quevedo et al., Reference Quevedo, Johnson, Loman, LaFavor and Gunnar2011). Deprivation experiences may interfere with children’s HPA axis functioning, resulting in blunted HPA axis activity (Reid et al., Reference Reid, DePasquale, Donzella, Leneman, Taylor and Gunnar2021). Blunted CAR may be a marker of allostatic overload following chronic stress (O'Connor et al., Reference O'Connor, Thayer and Vedhara2021), which reflects dampened physiological preparation for the coming day and is related to a string of health problems such as posttraumatic stress, fatigue, and burnout (Boggero et al., Reference Boggero, Hostinar, Haak, Murphy and Segerstrom2017; Chida & Steptoe, Reference Chida and Steptoe2009).
In contrast to neglect, abuse in girls was associated with less decreases in awakening cortisol, less increases in CAR, and more steepness in diurnal cortisol slope over time. The distinct effects of abuse on longitudinal changes in different diurnal cortisol indicators may emphasize the advantages of investigating multiple cortisol indicators. The finding regarding CAR is consistent with prior longitudinal research finding that compared to non-abused females, abused females showed lower increases in morning cortisol levels over time (Trickett et al., Reference Trickett, Noll, Susman, Shenk and Putnam2010). The association between abuse and steepness in diurnal cortisol slope over time seems counter-intuitive when considering steep diurnal cortisol slope as an index of better health (Adam et al., Reference Adam, Quinn, Tavernier, McQuillan, Dahlke and Gilbert2017). Nevertheless, prior cross-sectional research indicated that diurnal cortisol slope became steeper with age among previously institutionalized children, who experienced deprivation in early life and were also at high risk of abuse (Flannery et al., Reference Flannery, Gabard-Durnam, Shapiro, Goff, Caldera, Louie, Gee, Telzer, Humphreys, Lumian and Tottenham2017). Steeper diurnal cortisol slope, in general, reflects the activation of the HPA axis. Initial exposure to abuse may activate the HPA axis (reflected in steeper diurnal cortisol slope) to prepare the individual to deal with the stressors at hand, whereas prolonged abuse may eventually lead to down-regulation of the HPA axis, such as HPA axis hypoactivity and flatter diurnal cortisol slope (Miller et al., Reference Miller, Chen and Zhou2007). Given that diurnal cortisol slope is a relatively stable indicator of diurnal cortisol across time, compared to morning cortisol levels and fluctuations (Kuhlman et al., Reference Kuhlman, Robles, Dickenson, Reynolds and Repetti2019; Shirtcliff et al., Reference Shirtcliff, Allison, Armstrong, Slattery, Kalin and Essex2012), it may take a longer time for the effects of abuse on diurnal cortisol slope to become solidified. It is possible that the present study captured a developmental phase in which abuse started to alter diurnal cortisol slope, and the effect was observed as greater activation of the HPA axis (i.e., more steepness in diurnal cortisol slope).
Of note, child abuse in girls inhibited decreases in awakening cortisol and increases in CAR over time but accelerated the longitudinal steepness in diurnal cortisol slope. These seemingly contradictory directions (i.e., abuse slowed down or sped up the longitudinal changes in diurnal cortisol) could be explained in terms of higher awakening cortisol being a reason for steeper diurnal cortisol slope (Adam et al., Reference Adam, Quinn, Tavernier, McQuillan, Dahlke and Gilbert2017). This may also be observed though their correlations, such that higher awakening cortisol was strongly associated with steeper diurnal cortisol slope at W2. A recent meta-analysis revealed that adversity characterized by threat but not deprivation was associated with accelerated biological development (i.e., earlier pubertal timing and advanced cellular aging) in children and adolescents (Colich et al., Reference Colich, Rosen, Williams and McLaughlin2020). The association between abuse and more steepness in diurnal cortisol slope over time found in the present study is consistent with this meta-analysis, since allostatic load has been proposed as a mechanism linking early adversity to accelerated pubertal timing (Colich et al., Reference Colich, Rosen, Williams and McLaughlin2020). According to Life History Theory, harsh environments may accelerate biological development to maximize the opportunity for reproduction prior to mortality (Belsky et al., Reference Belsky, Schlomer and Ellis2012). Nevertheless, whether abuse accelerates or inhibits HPA axis development may depend on the specific cortisol indicator and the age range. A recent longitudinal study identified 13 years old as a “developmental shift” of the effect of previously institutionalized experiences on within-individual changes in morning cortisol, such that comparison children showed more developmental changes in cortisol before this age and previously institutionalized children showed more developmental changes in cortisol after this age (VanTieghem et al., Reference VanTieghem, Korom, Flannery, Choy, Caldera, Humphreys, Gabard-Durnam, Goff, Gee, Telzer, Shapiro, Louie, Fareri, Bolger and Tottenham2021). Our findings on abuse inhibiting the developmental changes in morning cortisol levels were in line with that finding, because the present study mainly captured diurnal cortisol development prior to age 13 (98.1% children and adolescents were 9–12 years old at baseline).
Taking advantage of diurnal cortisol assessed longitudinally, this study found that in girls, child abuse and neglect were differentially associated with diurnal cortisol such that neglect was related to baseline diurnal cortisol whereas abuse was associated with longitudinal changes in diurnal cortisol. The dimensional approach to childhood adversity elucidates that the distinct effects of threat versus deprivation on neurophysiology are driven by differences in core characteristics of these two dimensions (McLaughlin et al., Reference McLaughlin, Sheridan and Lambert2014, Reference McLaughlin, Sheridan, Humphreys, Belsky and Ellis2021). Recent research scrutinized this notion by stating that temporal features of adversity might explain the distinct effects of abuse and neglect (Doom et al., Reference Doom, Peckins, Hein, Dotterer, Mitchell, Lopez-Duran, Brooks-Gunn, McLanahan, Hyde, Abelson and Monk2020; Kessler et al., Reference Kessler, Vrshek-Schallhorn, Mineka, Zinbarg, Craske and Adam2021). Child abuse may occur as a series of intermittent and unpredictable events (e.g., hitting the child, symbolic or verbal aggression towards the child), whereas neglect is more likely to be an early-onset, chronic, and daily occurrence. A recent prospective study revealed that adolescents’ hair cortisol was associated with concurrent but not previous parental harshness, and associated with prior but not concurrent parental disengagement (Doom et al., Reference Doom, Peckins, Hein, Dotterer, Mitchell, Lopez-Duran, Brooks-Gunn, McLanahan, Hyde, Abelson and Monk2020). Accordingly, the current findings may be that child abuse, as a repeated and unpredictable stressor, could continuously shape HPA axis functioning over time, and this process was captured in this study. In contrast, neglect might occur early and its effects on cortisol may be already solidified during late childhood, which could be reflected in our findings that neglect was associated with baseline cortisol but relations with temporal changes in cortisol were not observed. Given that the HPA axis may calibrate itself continuously to stress in the environmental context, this interpretation needs to be tested with longitudinal data covering a greater time span, which could capture more complete and dynamic processes by which child abuse and neglect exert their effects on the HPA axis.
By investigating multiple diurnal cortisol indicators, we were able to get a more complete understanding of diurnal cortisol rhythm and identify the distinct effects of child maltreatment dimensions on specific cortisol indicators in relation to other indicators. We learned that both child abuse and neglect were associated with girls’ awakening cortisol and CAR, though in different time scales, while only abuse but not neglect was associated with girls’ diurnal cortisol slope. Cortisol levels and fluctuations in the morning (i.e., awakening cortisol and CAR) and the cortisol decline across the day (i.e., diurnal cortisol slope) reflect inter-correlated but differentiated components of diurnal cortisol rhythm (O'Connor et al., Reference O'Connor, Thayer and Vedhara2021). Differences in the neural correlates for morning cortisol and diurnal cortisol slope may explain the distinct effects of abuse and neglect, given that the hippocampus plays an important role in regulating CAR (Fries et al., Reference Fries, Dettenborn and Kirschbaum2009), whereas diurnal cortisol slope is associated with multiple central and peripheral nervous systems (Adam et al., Reference Adam, Quinn, Tavernier, McQuillan, Dahlke and Gilbert2017). This is in line with theoretical and empirical work on the dimensional approach to adversity (McLaughlin et al., Reference McLaughlin, Sheridan and Lambert2014, Reference McLaughlin, Weissman and Bitrán2019) suggesting that threat and deprivation have distinct effects on brain structure and functioning. Given that a neural explanation could not be empirically tested in the current study, caution is needed, and more studies are warranted to reveal neural regulatory mechanisms contributing to the distinct effects of abuse and neglect on diurnal cortisol.
Resilience, defined at the individual level in this study, was found to moderate the effects of abuse on girls’ awakening cortisol at baseline and longitudinal changes in diurnal cortisol slope. In girls with lower levels of resilience, abuse was associated with higher awakening cortisol at the baseline assessment and more steepness in diurnal cortisol slope longitudinally, while in girls with higher levels of resilience, abuse was associated with lower awakening cortisol at the baseline assessment. These findings may reveal a complex pattern of the effects of resilience, suggesting both the potential benefits and costs. Resilience can provide individuals with psychological resources to cope with stressors (Chi et al., Reference Chi, Slatcher, Li, Zhao, Zhao, Ren, Zhu and Stanton2015), which may benefit individuals by buffering the negative effects of stress. However, individual resilience may also be accompanied by costs, such that persistent efforts to manage adversity may lead to physiological burdens (Brody et al., Reference Brody, Yu and Beach2016; Chen et al., Reference Chen, Brody and Miller2022). Prior research has revealed a “skin-deep resilience” phenomenon, suggesting that some psychosocially resilient children may gain external success at the cost of elevated physiological burden in the context of adversity, such as higher allostatic loads and faster epigenetic aging (Brody et al., Reference Brody, Yu, Chen, Miller, Kogan and Beach2013; Miller et al., Reference Miller, Yu, Chen and Brody2015; Ming et al., Reference Ming, Zuo, Zhang, Ren, Zhang and Huang2022). Considering that initial exposure to stress may lead to HPA axis hyperarousal but prolonged stress may be related to HPA axis hypoarousal (Miller et al., Reference Miller, Chen and Zhou2007), the moderation of resilience regarding abuse and baseline awakening cortisol in girls may demonstrate both a “stress-buffering” effect and a “skin-deep resilience” phenomenon since associations between abuse and girls’ awakening cortisol were found at both the lower and higher ends of resilience. Abuse may activate the HPA axis and manifest as higher levels of awakening cortisol for girls with lower levels of resilience, and moderate levels of resilience may protect girls’ HPA axes from being altered by abuse, whereas the highest levels of resilience may actually drive the HPA axis to be down-regulated and displayed as blunted awakening cortisol when persistently coping with abuse.
Only the “stress-buffering” but not the “skin-deep” resilience was observed when it came to the relation between abuse and longitudinal changes in diurnal cortisol slope. In line with the interpretations concerning the association between abuse and more steepness in diurnal cortisol slope over time, this may be due to the fact that the diurnal cortisol slope is relatively stable over time, such that we observed how abuse activated, but had not yet down-regulated, the HPA axis, via steep but not yet blunted diurnal cortisol slope. Collectively, in girls, moderate levels of individual resilience may serve as a stress buffer in the relations between abuse and diurnal cortisol, whereas the highest levels of resilience may yield potential costs reflected as blunted awakening cortisol.
Resilience did not moderate the association between neglect and diurnal cortisol. This may be because chronic deprivation can dampen one’s resilience, and thus, resilience makes less difference regarding the effects of neglect. This explanation is consistent with the correlations in this study that only neglect but not abuse was associated with lower levels of resilience. In addition, resilience did not consistently buffer the effects of abuse on other cortisol indicators. Though personal resilience is an important resource in the face of stress, when exposed to severe adversity such as child maltreatment, other coping strategies (e.g., “shift-and-persist” strategies; Chen & Miller, Reference Chen and Miller2012) and social resources (e.g., social support; Gunnar & Hostinar, Reference Gunnar and Hostinar2015) also appear critical in contributing to positive outcomes. Recent research also has revealed that the composites of various internal resources and multiple psychological and social resources can help buffer the effects of stress/adversity on HPA axis functioning (e.g., Chiang et al., Reference Chiang, Ko, Bower, Taylor, Irwin and Fuligni2019; Sun et al., Reference Sun, Jiang, Zilioli, Xie, Chen and Lin2023). Therefore, the accumulation or combination of various individual and contextual resources may also serve a protective role in the association between adversity and cortisol.
The effects of abuse and neglect on diurnal cortisol and the moderating role of resilience were only found in girls but not in boys. These findings are consistent with findings that girls’ HPA axis functioning is more vulnerable to stress (Carpenter et al., Reference Carpenter, Grecian and Reynolds2017), and may also be more sensitive to individual resources. These findings identify girls as a vulnerable group to child maltreatment and indicate that intervention programs aiming at improving regulation of stress physiology and cultivating optimal resilience may benefit maltreated girls. It should be noted, however, that these results do not necessarily mean that boys’ HPA axis functioning will not be influenced by adversity, because adversity-physiology associations may vary according to domains of adversity (e.g., in family versus peer contexts), aspects of HPA axis functioning (e.g., diurnal cortisol versus cortisol stress reactivity), and age ranges.
Limitations and conclusions
The findings from the present study need to be interpreted in the context of the following limitations. First, other characteristics of child maltreatment (e.g., severity, onset time, perpetrator) were not assessed in the present study, which may be important moderators in the association between child maltreatment and HPA axis functioning. It also should be noted that the emotional neglect subscale in the Childhood Trauma Questionnaire-Short Form is based on reverse-worded items. Second, the changes of diurnal cortisol over one year may be influenced by child abuse and neglect that happened during this one-year time frame. However, our lack of measures on past-year child maltreatment at W2 prevented us from testing this possibility. Third, though diurnal cortisol was measured at two time points during a one-year interval, more time points across a longer developmental period would help to fully understand how the HPA axis matures across the transition from childhood to adolescence. Fourth, the current results should also be interpreted in the context of the pandemic, given that our baseline data (December 2019) were collected before the pandemic, and the W2 data (November 2020) were collected amid the pandemic and during the stage of “regular epidemic prevention and control,” which reflected public masking but no quarantine mandates in the data collection areas.
Fifth, this study builds upon the dimensional approach that categorizes adversity as threat and deprivation (McLaughlin & Sheridan, Reference McLaughlin and Sheridan2016; McLaughlin et al., Reference McLaughlin, Sheridan and Lambert2014), but the original assumptions of this approach mainly focus on neural development. It may be fruitful for future work to explore the neural mechanisms that can explain the distinct effects of abuse and neglect on diurnal cortisol. Of note, the dimensional approach conceptualizes adverse experiences in a broader sense than just child maltreatment (McLaughlin & Sheridan, Reference McLaughlin and Sheridan2016; McLaughlin et al., Reference McLaughlin, Sheridan and Lambert2014). Though child maltreatment is one of the most common adversities that asserts profound effects on child development, future studies may benefit from including a wider range of adversities to fully capture the proposed adversity dimensions.
Sixth, resilience in this study was operationalized and measured as personal strengths in the face of adversity, however, recent research has emphasized the multilevel, interacting, and dynamic nature of resilience in a broader sense (Masten et al., Reference Masten, Lucke, Nelson and Stallworthy2021). Since definitions and measures of resilience may vary across cultural contexts, we should be cautious to compare these findings with results from studies using other measures or definitions of resilience, or conducted in different cultural contexts. Seventh, stratified models by sex revealed different patterns of associations in boys versus girls, but not all observed differences were statistically significant when child sex was added as a moderator. Due to analytic power concerns when statistically testing for child sex moderation effects, we should be cautious in interpreting these sex differences. Lastly, one’s pubertal stage can be an important factor that influences or helps explain the associations between maltreatment and cortisol and contributes to sex differences in these associations (Negriff et al., Reference Negriff, Gordis and Susman2023). Considering the age range of participants (i.e., 9–13 years at baseline), it is likely that some participants were experiencing puberty onset (Zhou & Zhang, Reference Zhou and Zhang2015). Though this study controlled for age as a proxy for one’s physical maturation in analyses, an avenue for future research is to test whether the current findings vary for individuals at different pubertal stages, or exploring whether pubertal development serves as a mechanism for the effects of maltreatment on cortisol.
Notwithstanding these limitations, the current findings support, refine, and extend the hypotheses of the dimensional approach of adversity to research on child maltreatment and stress physiology. The distinct effects of abuse and neglect may imply that varying types of maltreatment should be taken into consideration when targeting the amelioration of stress in intervention programs for maltreated children. By focusing on both diurnal cortisol at the baseline assessment and within-individual changes in diurnal cortisol over time, and including multiple cortisol indicators, this study provides a more developmental and comprehensive lens to understand the associations between maltreatment and diurnal cortisol. This study also adds to the extant literature on individual differences in stress physiology by illuminating the moderating effects of resilience and considering the role of child sex, which revealed a complex pattern of potential benefits and costs of resilience in relations between abuse and girls’ diurnal cortisol. Underscoring individual differences in relations between adversity and the HPA axis may help to understand vulnerable and resilient individuals in the face of adversity, and provide targets for child maltreatment intervention. Finally, the present study focused on Chinese children and adolescents from a disadvantaged area, which may complement prior research on stress and physiology that is predominated by samples from Western cultures.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S0954579423001086.
Acknowledgements
We are appreciative of the schools, families and children that participated in our study. We acknowledge Yuyan Wang and other research assistants for their great efforts in collecting the data.
Funding statement
This study was funded by National Natural Science Foundation of China (32071076) awarded to DL and National Institute of Child Health and Human Development (R01HD097189) awarded to EL.
Competing interests
None.









