In 1996 I was commissioned by the Department of Health (DOH) to review the scientific literature regarding the potential therapeutic utility of cannabis and its derivatives. The review was based upon primary sources (identified from a Medline literature search, reference lists supplied by the DOH and the Institute for the Study of Drug Dependence, and personal communications with relevant academics and clinicians). This paper is a greatly shortened version of the review. The 4 years which have elapsed have seen little in the way of new clinical results but considerable advances in cannabinoid basic science (Reference Joy, Watson and BensonInstitute of Medicine, 1999). Government licences have recently been granted for several controlled trials of both synthetic and plant-derived cannabinoids in multiple sclerosis and chronic pain. In January 2000, I was appointed Medical Director of GW Pharmaceuticals, a company established to derive medicinal extracts from standardised cannabis plants.
HISTORY OF THERAPEUTIC USE
The first formal report of cannabis as a medicine appeared in China nearly 5000 years ago when it was recommended for malaria, constipation, rheumatic pains and childbirth and, mixed with wine, as a surgical analgesic (Reference Mechoulam and MechoulamMechoulam, 1986). There are subsequent records of its use throughout Asia, the Middle East, Southern Africa and South America. Accounts by Pliny, Dioscorides and Galen remained influential in European medicine for 16 centuries.
It was not until the 19th century that cannabis became a mainstream medicine in Britain. W. B. O'Shaughnessy, an Irish scientist and physician, observed its use in India as an analgesic, anticonvulsant, anti-spasmodic, anti-emetic and hypnotic. After toxicity experiments on goats and dogs, he gave it to patients and was impressed with its muscle-relaxant, anticonvulsant and analgesic properties, and recorded its use-fulness as an anti-emetic.
After these observations were published in 1842, medicinal use of cannabis expanded rapidly. It soon became available ‘over the counter’ in pharmacies and by 1854 it had found its way into the United States Dispensatory. The American market became flooded with dozens of cannabis-containing home remedies.
Queen Victoria's personal physician wrote (Reference ReynoldsReynolds, 1890), on the basis of more than 30 years' experience, that “Indian hemp, when pure and administered carefully, is one of the most valuable medicines we possess”. He found it incomparable for “senile insomnia”, “ night restlessness” and “temper disease” in both children and adults, but not helpful in melancholia, “very uncertain” in alcoholic delirium, and “worse than useless” in mania. It was very effective in neuralgia, period pains, migraine, “ lightning pain of the ataxic patient” and gout, but useless in sciatica and “hysteric pains”. He found it impressive in clonic spasms and certain epileptiform convulsions related to brain damage, but no good at all in petit mal or “chronic epilepsy”, tetanus, chorea or paralysis agitans. It effectively relieved nocturnal cramps, asthma and dysmenorrhoea.
Reynolds was writing at a time when the zenith of cannabis as prescribed medicine and home remedy was already past. Although Sir William Osler was still recommending it for migraine sufferers in 1913, it was by then in steep decline because of variable potency of herbal preparations, poor storage stability, unpredictable response to oral administration, increasing enthusiasm for parenteral medicines and availability of potent synthetic alternatives, commercial pressures and American concern about recreational use. Cannabis was outlawed in 1928 by ratification of the 1925 Geneva Convention on the manufacture, sale and movement of dangerous drugs. Prescription remained possible until final prohibition under the 1971 Misuse of Drugs Act, against the advice of the Advisory Committee on Drug Dependence.
In the USA, medical use was effectively ruled out by the Marijuana Tax Act 1937. This ruling has been under almost constant legal challenge and many special dispensations were made between 1976 and 1992 for individuals to receive ‘compassionate reefers’. Although this loophole has been closed, a 1996 California state law permits cultivation or consumption of cannabis for medical purposes, if a doctor provides a written endorsement. Similar arrangements apply in Italy and Canberra, Australia.
CANNABINOID PHARMACOLOGY RELEVANT TO THERAPEUTICS
Cannabinol was isolated in 1895 and cannabidiol in 1934, but the most significant discovery was that of ▵9-tetrahydrocannabinol (THC) in 1964. Chromatographic and spectroscopic methods subsequently uncovered many closely related compounds.
Capsules of synthetic THC (dronabinol) have been available for restricted medical use in the USA since 1985. Nabilone, a synthetic THC analogue, was marketed in 1983 and is the only cannabinoid licensed for prescription in the UK, restricted to treatment of nausea and vomiting caused by cytotoxic chemotherapy unresponsive to conventional anti-emetics. Use in other indications is only possible on a ‘named patient’ basis if the drug is supplied by a hospital pharmacy.
In 1988, a specific protein receptor (known as CB1) for THC was discovered in mouse nerve cells. This mediates most of the central nervous system (CNS) responses to cannabinoids, and is abundant in basal ganglia, hippocampus and cerebellum, globus pallidus, substantia nigra and cerebral cortex. An endogenous ligand was identified in 1992 and labelled anandamide (ananda: ‘bliss’ in Sanskrit). Anandamide has analgesic and tranquillising effects in animals, is involved in muscle coordination and affects the secretion and function of certain hormones. Other endogenous agonists almost certainly exist.
In 1993, a second receptor (CB2) was identified in rat spleen macrophages, and this occurs only outside the CNS. There is scope for chemical manipulation of cannabinoids to maximise selectivity for CB2 and so avoid psychoactive effects. It is thought this receptor has relevance for anti-inflammatory and immunosuppressive activity.
Pertwee (Reference Pertwee and Pertwee1995) has suggested that the anandamide system might be concerned with mood, memory and cognition, perception, movement, coordination, posture and skeletal muscle tone, sleep, thermo-regulation, appetite and immune response.
CLINICAL APPLICATIONS
Nausea and vomiting
Many cytotoxic drugs are powerful emetics, and this is the major limiting factor in patients' acceptance of cancer chemotherapy (see Table 1 and Appendix).
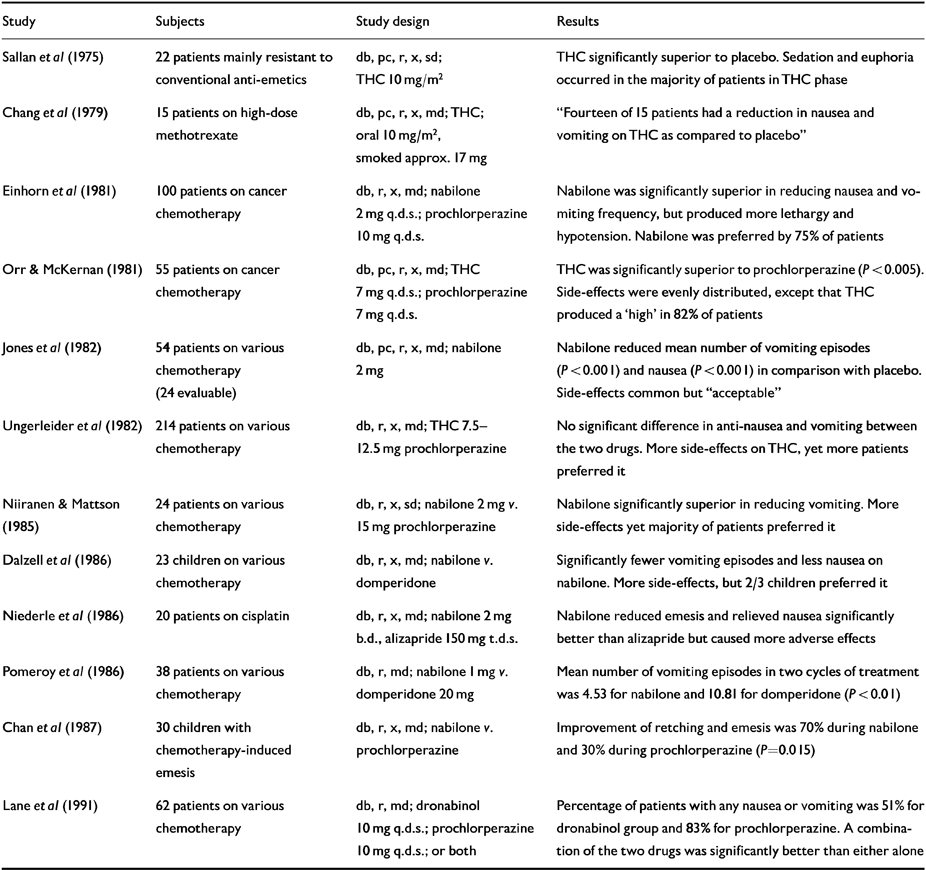
Table 1 Human randomised controlled trials (RCTs): anti-emetic effects
| Study | Subjects | Study design | Results |
|---|---|---|---|
| Sallan et al (Reference Sallan, Zinberg and Frei1975) | 22 patients mainly resistant to conventional anti-emetics | db, pc, r, x, sd; THC 10 mg/m2 | THC significantly superior to placebo. Sedation and euphoria occurred in the majority of patients in THC phase |
| Chang et al (Reference Chang, Shiling and Stillman1979) | 15 patients on high-dose methotrexate | db, pc, r, x, md; THC; oral 10 mg/m2, smoked approx. 17 mg | “ Fourteen of 15 patients had a reduction in nausea and vomiting on THC as compared to placebo” |
| Einhorn et al (Reference Einhorn, Nagy and Furnas1981) | 100 patients on cancer chemotherapy | db, r, x, md; nabilone 2 mg q.d.s.; prochlorperazine 10 mg q.d.s. | Nabilone was significantly superior in reducing nausea and vomiting frequency, but produced more lethargy and hypotension. Nabilone was preferred by 75% of patients |
| Orr & McKernan (Reference Orr and McKernan1981) | 55 patients on cancer chemotherapy | db, pc, r, x, md; THC 7 mg q.d.s.; prochlorperazine 7 mg q.d.s. | THC was significantly superior to prochlorperazine (P < 0.005). Side-effects were evenly distributed, except that THC produced a ‘high’ in 82% of patients |
| Jones et al (Reference Jones, Durant and Greco1982) | 54 patients on various chemotherapy (24 evaluable) | db, pc, r, x, md; nabilone 2 mg | Nabilone reduced mean number of vomiting episodes (P < 0.001) and nausea (P < 0.001) in comparison with placebo. Side-effects common but “acceptable” |
| Ungerleider et al (Reference Ungerleider, Andrysiak and Fairbanks1982) | 214 patients on various chemotherapy | db, r, x, md; THC 7.5-12.5 mg prochlorperazine | No significant difference in anti-nausea and vomiting between the two drugs. More side-effects on THC, yet more patients preferred it |
| Niiranen & Mattson (Reference Niiranen and Mattson1985) | 24 patients on various chemotherapy | db, r, x, sd; nabilone 2 mg v. 15 mg prochlorperazine | Nabilone significantly superior in reducing vomiting. More side-effects yet majority of patients preferred it |
| Dalzell et al (Reference Dalzell, Bartlett and Lilleyman1986) | 23 children on various chemotherapy | db, r, x, md; nabilone v. domperidone | Significantly fewer vomiting episodes and less nausea on nabilone. More side-effects, but 2/3 children preferred it |
| Niederle et al (Reference Niederle, Shütte and Schmidt1986) | 20 patients on cisplatin | db, r, x, md; nabilone 2 mg b.d., alizapride 150 mg t.d.s. | Nabilone reduced emesis and relieved nausea significantly better than alizapride but caused more adverse effects |
| Pomeroy et al (Reference Pomeroy, Fennelly and Towers1986) | 38 patients on various chemotherapy | db, r, md; nabilone 1 mg v. domperidone 20 mg | Mean number of vomiting episodes in two cycles of treatment was 4.53 for nabilone and 10.81 for domperidone (P < 0.01) |
| Chan et al (Reference Chan, Correia and MacLeod1987) | 30 children with chemotherapy-induced emesis | db, r, x, md; nabilone v. prochlorperazine | Improvement of retching and emesis was 70% during nabilone and 30% during prochlorperazine (P=0.015) |
| Lane et al (Reference Lane, Vogel and Ferguson1991) | 62 patients on various chemotherapy | db, r, md; dronabinol 10 mg q.d.s.; prochlorperazine 10 mg q.d.s.; or both | Percentage of patients with any nausea or vomiting was 51% for dronabinol group and 83% for prochlorperazine. A combination of the two drugs was significantly better than either alone |
Many recreational smokers receiving cancer chemotherapy have told their doctors that cannabis relieved their nausea (Reference Grinspoon and BakalarGrinspoon & Bakalar, 1993). Sallan et al's (Reference Sallan, Zinberg and Frei1975) randomised control trial (RCT) compared oral THC and placebo in 22 cancer patients who had proved resistant to conventional anti-emetics. Comparisons using patients' self-reports of nausea and vomiting demonstrated that THC was statistically superior to placebo. THC (10 mg/m2) produced euphoria in the majority of patients, and one-third experienced sedation.
Subsequent RCTs (listed in Table 1) confirmed that natural and synthetic THC is invariably superior to placebo. Comparisons with anti-emetics available in the 1970s and 1980s suggest that THC is either equivalent in effect or better. A combination of prochlorperazine and THC was superior to either drug alone, and nabilone combined with prochlorperazine was better than dexamethazone plus metoclopramide. Although THC and nabilone produced more unwanted effects than comparison drugs, patients generally preferred them.
Children seem to respond well to nabilone and are tolerant of side-effects, but larger studies are required. ▵8-THC performed well in a pilot study (Reference Abrahamov, Abrahamov and MechoulamAbrahamov et al, 1995) involving eight children aged 3-13 years with various blood cancers receiving chemotherapy, 60% of whom had experienced distressing vomiting despite treatment with metoclopramide. ▵8-THC was given orally 2 hours before cytotoxics and repeated 6-hourly. No vomiting was recorded during this treatment and over the following 2 days. Two children were “slightly irritable” and one also showed “slight euphoria”.
In a review of 12 studies involving 600 patients (Reference Penta, Poster and BrunoPenta et al, 1981), THC was “effective” in 8/9 and nabilone in 3/3. The most common side-effects were somnolence (33%), dry mouth (9%), ataxia (8%), dizziness (6%), dysphoria (6%), and orthostatic hypotension (4%). A further review (Reference Levitt and MechoulamLevitt, 1986) incorporating 55 studies, of which 32 were RCTs, showed that low-dose preventive treatment gives better results than targeting established vomiting. Younger patients may respond better than older ones.
Meta-analysis (Reference Plasse, Gorter and KrasnowPlasse et al, 1991) suggested that an optimal balance of efficacy and unwanted effects was achieved with relatively modest doses (7 mg/m2 or less). Sedation and psychotropic symptoms are commonly reported, but are usually mild to moderate in intensity and resolve rapidly on discontinuation. No “persistent or fatal” adverse effects have been reported. Many American oncologists encourage nauseous patients to try cannabis and would prescribe it if it were legal (Reference Doblin and KleimanDoblin & Kleiman, 1991). Mode of action remains uncertain.
Multiple sclerosis and other neurological conditions
Drug therapy of muscle spasticity is generally only moderately effective and is limited by adverse effects (see Appendix). Spasticity is a central feature of multiple sclerosis (MS), cerebral palsy and spinal cord injury. Tremor, ataxia and incontinence also contribute to the high incidence of anxiety and depression in these conditions. Cannabis was often used to treat pain, muscle spasm, cramps and ataxia in the 19th century, and many modern sufferers have reported benefits (Reference Grinspoon and BakalarGrinspoon & Bakalar, 1993).
Most respondents to a questionnaire sent to British and American MS patients reported problems with symptom control (Reference Consroe, Musty and ReinConsroe et al, 1997). Those who smoked cannabis claimed improvements in night-time spasticity and muscle pain (91-98%); night leg pain, depression, tremor, anxiety, spasms on walking, paraesthesiae (80-89%); leg weakness, trunk numbness, facial pain (71-74%); impaired balance (57%); constipation (33%); memory loss (31%).
In a small single-blind comparison with placebo (Reference CliffordClifford, 1983), THC improved tremor and ataxia in most patients. All experienced a ‘high’ at the top dose (15 mg), and two reported dysphoria. Dose-related improvements in dystonia were noted in five patients given cannabidiol 100-600 mg daily for 6 weeks. Hypotension, dry mouth, sedation and light-headedness occurred but were described as mild. Parkinsonian symptoms were aggravated in two subjects.
An RCT by Petro & Ellenberger (Reference Petro and Ellenberger1981) compared the effects of placebo and THC in doses of 5 or 10 mg on muscle tone, reflexes and muscle power in nine MS patients. Both doses of THC reduced spasticity (P < 0.005). One patient receiving THC 10 mg and one patient receiving placebo felt ‘high’ but no other side-effects were recorded. In a small RCT (Reference Ungerleider, Andrysiak and FairbanksUngerleider et al, 1987) with 5-day treatment periods, THC 7.5 mg significantly improved spasticity in comparison with placebo. Nabilone 1 mg on alternate days was compared with placebo in a double-blind randomised cross-over trial with 4-week treatment periods in a single MS patient. Nocturia, muscle spasm and general well-being showed striking improvement during each active treatment period. Mild sedation was noted on active medication.
Cannabidiol had no beneficial effects in 15 patients with Huntington's disease (Reference Consroe, Laguna and AllenderConsroe et al, 1991). Posture and balance were impaired by a single dose of smoked THC in 10 MS patients and 10 non-MS volunteers (Reference Greenberg, Werness and PughGreenberg et al, 1994), but there was no active control to determine the effects of standard anti-spastic medication in this model.
Possible sites of action of cannabinoids in dystonia include basal ganglia, cerebellum, spinal motor neurons, somatic nerves and neuromuscular junction.
Loss of appetite and weight in cancer and AIDS
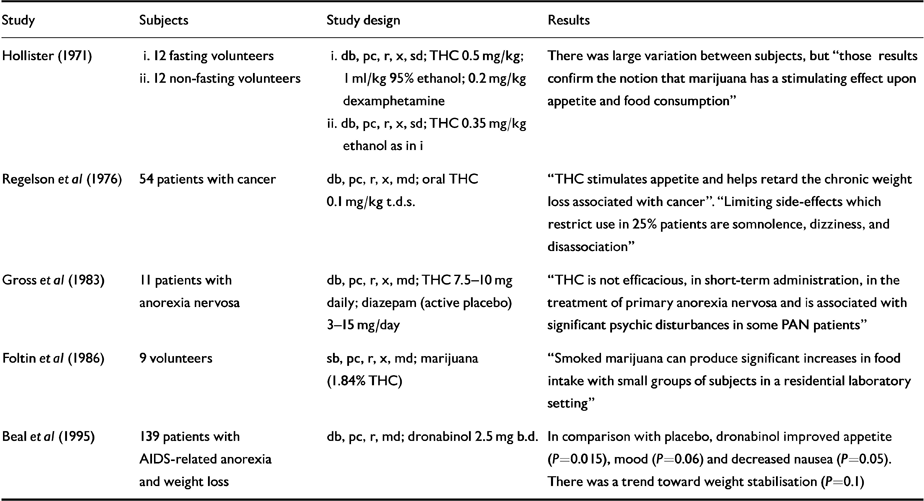
Several studies have investigated effect on appetite and weight (Table 2). The appetite-stimulating effect of cannabis was confirmed in fasting and non-fasting volunteers in an RCT of oral THC with alcohol, amphetamine and placebo (Reference HollisterHollister, 1971). A standardised THC smoking regime over 25 days in a residential laboratory was associated with significant increases in calorie intake and frequency of eating occasions in comparison with placebo.
Table 2 Human randomised controlled trials (RCTs): appetite and weight
| Study | Subjects | Study design | Results |
|---|---|---|---|
| Hollister (Reference Hollister1971) | i. 12 fasting volunteers | i. db, pc, r, x, sd; THC 0.5 mg/kg; 1 ml/kg 95% ethanol; 0.2 mg/kg dexamphetamine | There was large variation between subjects, but “those results confirm the notion that marijuana has a stimulating effect upon appetite and food consumption” |
| ii. 12 non-fasting volunteers | ii. db, pc, r, x, sd; THC 0.35 mg/kg ethanol as in i | ||
| Regelson et al (Reference Regelson, Butler, Schulz, Braude and Szara1976) | 54 patients with cancer | db, pc, r, x, md; oral THC 0.1 mg/kg t.d.s. | “ THC stimulates appetite and helps retard the chronic weight loss associated with cancer”. “Limiting side-effects which restrict use in 25% patients are somnolence, dizziness, and disassociation” |
| Gross et al (Reference Gross, Ebert and Faden1983) | 11 patients with anorexia nervosa | db, pc, r, x, md; THC 7.5-10 mg daily; diazepam (active placebo) 3-15 mg/day | “ THC is not efficacious, in short-term administration, in the treatment of primary anorexia nervosa and is associated with significant psychic disturbances in some PAN patients” |
| Foltin et al (Reference Foltin, Brady and Fischman1986) | 9 volunteers | sb, pc, r, x, md; marijuana (1.84% THC) | “ Smoked marijuana can produce significant increases in food intake with small groups of subjects in a residential laboratory setting” |
| Beal et al (Reference Beal, Olson and Laubenstein1995) | 139 patients with AIDS-related anorexia and weight loss | db, pc, r, md; dronabinol 2.5 mg b.d. | In comparison with placebo, dronabinol improved appetite (P=0.015), mood (P=0.06) and decreased nausea (P=0.05). There was a trend toward weight stabilisation (P=0.1) |
Open studies in cancer patients also showed appetite improvements and slowing of weight loss. Regelson et al's (Reference Regelson, Butler, Schulz, Braude and Szara1976) RCT explored the effect on appetite (and mood) of oral THC in 54 cancer patients over a 2-week period. There were nine with-drawals due to side-effects (six in THC period - dizziness, disassociation, confused thinking, panic, “feelings of disturbance”; three in the placebo period - anxiety, fits, dizziness, lethargy, weakness). Patients receiving THC in the first period gained weight (P<0.05), and those receiving placebo first showed reduced weight loss on transfer to THC (P<0.05). Depression, tranquillity and “ forthrightness” scores all improved on THC. In a quarter of the patients, somnolence, dizziness and disassociation were severe enough to negate these effects.
Many people with AIDS have claimed that smoking marijuana inhibits nausea, improves appetite, reduces anxiety, relieves aches and pains, improves sleep and inhibits oral candidiasis. A small pilot study supported the hypothesis that dronabinol might reduce weight loss or even promote weight gain (Reference Plasse, Gorter and KrasnowPlasse et al, 1991).
Beal et al (Reference Beal, Olson and Laubenstein1995) conducted an RCT over 42 days of treatment with dronabinol 5 mg daily in 139 AIDS patients who had lost at least 2.3 kg. Six receiving dronabinol and three receiving placebo withdrew because of “perceived drug toxicity”. Dronabinol boosted appetite in comparison to placebo (P<0.015) and nausea was reduced (P=0.05). Improvement in mood was a strong trend (P=0.06) and there was a tendency toward weight gain (P=0.1). Dronabinol produced more adverse effects than placebo (P<0.001), but 75% of these were mild or moderate. Most frequent were euphoria (9), dizziness (5), thinking abnormalities (5) and sedation (4).
Further investigation is amply justified. Careful monitoring of possible effects upon the immune system is needed, although a prospective multi-centre study (Reference Kaslow, Blackwelder and OstrowKaslow et al, 1989), which followed nearly 5000 HIV-positive men for 18 months, showed no link between use of psychoactive substances and mean T-cell counts or progression to AIDS.
Pain
Cannabinoids are effective analgesics in animal models with non-opiate mechanisms predominating. There are many anecdotal reports (Reference Grinspoon and BakalarGrinspoon & Bakalar, 1993) of benefits in bone and joint pain, migraine, cancer pain, menstrual cramps and labour.
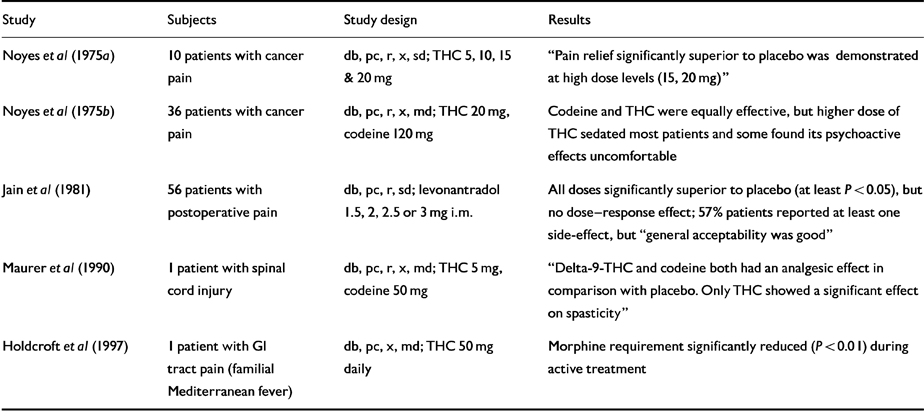
Five small RCTs (Table 3) show that THC is significantly superior to placebo and produces dose-related analgesia peaking at around 5 hours, comparable to but out-lasting that of codeine. Side-effects were also dose-related, and consisted of slurred speech, sedation and mental clouding, blurred vision, dizziness and ataxia. Levonantradol was also superior to placebo and notably long-acting, but almost half the patients reported sedation. Cannabinoids may have considerable potential in neuropathic pain (Reference Joy, Watson and BensonInstitute of Medicine, 1999).
Table 3 Human randomised controlled trials (RCTs): pain
| Study | Subjects | Study design | Results |
|---|---|---|---|
| Noyes et al (Reference Noyes, Brunk and Baram1975a ) | 10 patients with cancer pain | db, pc, r, x, sd; THC 5, 10, 15 & 20 mg | “ Pain relief significantly superior to placebo was demonstrated at high dose levels (15, 20 mg)” |
| Noyes et al (Reference Noyes and Avery1975b ) | 36 patients with cancer pain | db, pc, r, x, md; THC 20 mg, codeine 120 mg | Codeine and THC were equally effective, but higher dose of THC sedated most patients and some found its psychoactive effects uncomfortable |
| Jain et al (Reference Jain, Ryan and McMahon1981) | 56 patients with postoperative pain | db, pc, r, sd; levonantradol 1.5, 2, 2.5 or 3 mg i.m. | All doses significantly superior to placebo (at least P<0.05), but no dose—response effect; 57% patients reported at least one side-effect, but “general acceptability was good” |
| Maurer et al (Reference Maurer, Henn and Dittrich1990) | 1 patient with spinal cord injury | db, pc, r, x, md; THC 5 mg, codeine 50 mg | “ Delta-9-THC and codeine both had an analgesic effect in comparison with placebo. Only THC showed a significant effect on spasticity” |
| Holdcroft et al (Reference Holdcroft, Smith and Jacklin1997) | 1 patient with GI tract pain (familial Mediterranean fever) | db, pc, x, md; THC 50 mg daily | Morphine requirement significantly reduced (P<0.01) during active treatment |
Raised intra-ocular pressure
Glaucoma due to obstructed outflow of aqueous humour or anatomical eye defects is the most common cause of blindness in the Western world. Some RCTs investigating this area are given in Table 4.
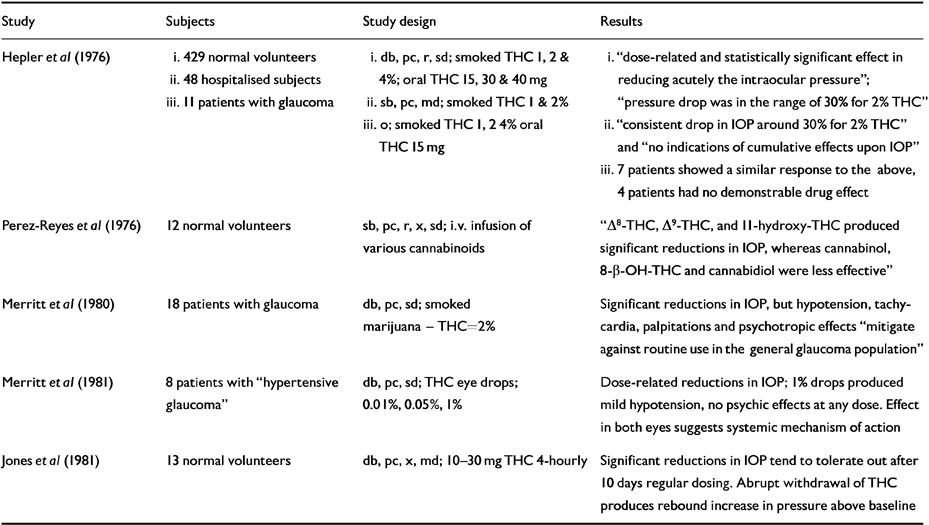
Table 4 Human randomised controlled trials (RCTs): raised intra-ocular pressure (IOP)
| Study | Subjects | Study design | Results |
|---|---|---|---|
| Hepler et al (Reference Hepler, Frank, Petrus, Braude and Szara1976) | i. 429 normal volunteers | i. db, pc, r, sd; smoked THC 1, 2 & 4%; oral THC 15, 30 & 40 mg | i. “dose-related and statistically significant effect in reducing acutely the intraocular pressure”; “pressure drop was in the range of 30% for 2% THC” |
| ii. 48 hospitalised subjects | ii. sb, pc, md; smoked THC 1 & 2% | ii. “consistent drop in IOP around 30% for 2% THC” and “no indications of cumulative effects upon IOP” | |
| iii. 11 patients with glaucoma | iii. o; smoked THC 1, 24% oral THC 15 mg | iii. 7 patients showed a similar response to the above, 4 patients had no demonstrable drug effect | |
| Perez-Reyes et al (Reference Perez-Reyes, Wagner, Wall, Braude and Szara1976) | 12 normal volunteers | sb, pc, r, x, sd; i.v. infusion of various cannabinoids | “Δ8-THC, Δ9-THC, and 11-hydroxy-THC produced significant reductions in IOP, whereas cannabinol, 8-β-OH-THC and cannabidiol were less effective” |
| Merritt et al (Reference Merritt, Crawford and Alexander1980) | 18 patients with glaucoma | db, pc, sd; smoked marijuana - THC=2% | Significant reductions in IOP, but hypotension, tachycardia, palpitations and psychotropic effects “mitigate against routine use in the general glaucoma population” |
| Merritt et al (Reference Merritt, Olsen and Armstrong1981) | 8 patients with “hypertensive glaucoma” | db, pc, sd; THC eye drops; 0.01%, 0.05%, 1% | Dose-related reductions in IOP; 1% drops produced mild hypotension, no psychic effects at any dose. Effect in both eyes suggests systemic mechanism of action |
| Jones et al (Reference Jones, Benowitz and Herning1981) | 13 normal volunteers | db, pc, x, md; 10-30 mg THC 4-hourly | Significant reductions in IOP tend to tolerate out after 10 days regular dosing. Abrupt withdrawal of THC produces rebound increase in pressure above baseline |
There have been many anecdotal reports that street marijuana can relieve glaucoma symptoms and individuals have successfully argued in the USA for legal access to the drug (Reference Grinspoon and BakalarGrinspoon & Bakalar, 1993). A pilot study of smoked marijuana and oral THC (15 mg) in 11 glaucoma patients found an average intra-ocular pressure (IOP) reduction of 30% in seven subjects and no response in four (Reference Hepler, Frank, Petrus, Braude and SzaraHepler et al, 1976).
Randomised controlled trials in volunteers confirmed that oral, injected or smoked cannabinoids produce dose-related reductions of IOP (Reference Hepler, Frank, Petrus, Braude and SzaraHepler et al, 1976; Reference Perez-Reyes, Wagner, Wall, Braude and SzaraPerez-Reyes et al, 1976). Conjunctival engorgement and tear reduction were often noted. THC, ▵8-THC and 11-hydroxy- THC are more effective than cannabinol, while cannabidiol was without effect. Tolerance may develop on multiple dosing.
An RCT in patients showed IOP reductions of similar magnitude following smoked THC along with “alterations in mental status” and tachycardia (Reference Merritt, Crawford and AlexanderMerritt et al, 1980). THC eyedrops produced dose-related IOP reduction with minimal side-effects though parallel reductions in the untreated eye (also seen in animal models) suggested a systemic rather than local mode of action.
Insomnia, anxiety and depression
Randomised controlled trials investigating insomnia, anxiety and depression are given in Table 5.
Table 5 Human randomised controled trials (RCTs): insomnia, anxiety, depression
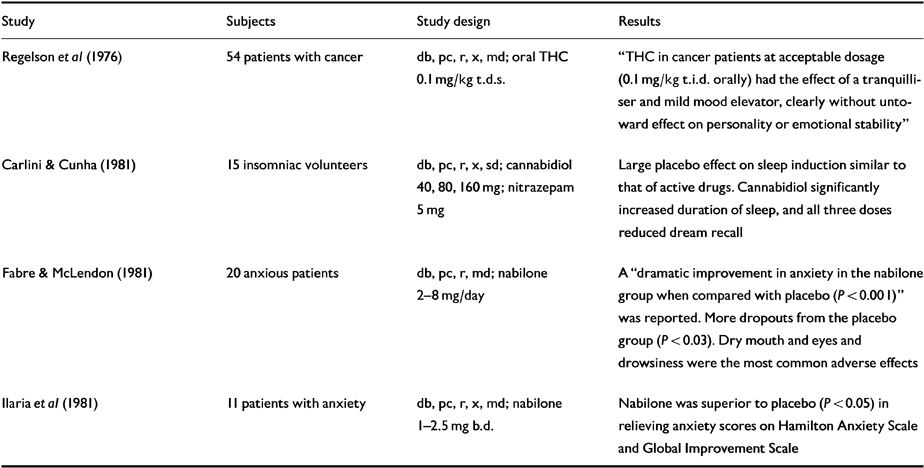
| Study | Subjects | Study design | Results |
|---|---|---|---|
| Regelson et al (Reference Regelson, Butler, Schulz, Braude and Szara1976) | 54 patients with cancer | db, pc, r, x, md; oral THC 0.1 mg/kg t.d.s. | “ THC in cancer patients at acceptable dosage (0.1 mg/kg t.i.d. orally) had the effect of a tranquilliser and mild mood elevator, clearly without untoward effect on personality or emotional stability” |
| Carlini & Cunha (Reference Carlini and Cunha1981) | 15 insomniac volunteers | db, pc, r, x, sd; cannabidiol 40, 80, 160 mg; nitrazepam 5 mg | Large placebo effect on sleep induction similar to that of active drugs. Cannabidiol significantly increased duration of sleep, and all three doses reduced dream recall |
| Fabre & McLendon (Reference Fabre and McLendon1981) | 20 anxious patients | db, pc, r, md; nabilone 2-8 mg/day | A “dramatic improvement in anxiety in the nabilone group when compared with placebo (P < 0.001)” was reported. More dropouts from the placebo group (P < 0.03). Dry mouth and eyes and drowsiness were the most common adverse effects |
| Ilaria et al (Reference Ilaria, Thornby and Fann1981) | 11 patients with anxiety | db, pc, r, x, md; nabilone 1-2.5 mg b.d. | Nabilone was superior to placebo (P < 0.05) in relieving anxiety scores on Hamilton Anxiety Scale and Global Improvement Scale |
Nabilone (1 mg three times daily) produced “dramatic improvements” on the Hamilton Anxiety Scale in 20 anxious patients in comparison to placebo (P<0.001), which were mirrored by other measures (Reference Fabre and McLendonFabre & McLendon, 1981). Seven days into the study, nabilone patients' anxiety scores were halved, and this persisted unchanged throughout treatment. Side-effects included dry mouth, dry eyes and drowsiness. The authors concluded that nabilone is a “very effective anxiolytic deserving of further study”. In a cross-over comparison of nabilone (1-2.5 mg twice daily) and placebo in 11 anxious patients (Reference Ilaria, Thornby and FannIlaria et al, 1981), significant improvements in anxiety scores (P<0.05) were again noted. The only clinically significant adverse effect was postural hypotension with related dizziness, light-headedness or weakness. This was dose-related, experienced by most patients, and tended to tolerate out over time.
Preliminary data suggest that cannabidiol (160 mg) may be an effective hypnotic, and that THC (0.1 mg/kg) may have antidepressant properties in cancer patients and others (Reference Grinspoon and BakalarGrinspoon & Bakalar, 1993).
Epilepsy
Epilepsy afflicts 1% of the world's population. Conventional anticonvulsants provide unsatisfactory control for up to 30% of patients, and all can produce disabling or even life-threatening adverse effects.
The effect of cannabinoids on seizure activity in laboratory animals is complicated. Cannabidiol is a powerful anticonvulsant free of tolerance, but its profile varies between species. THC can produce seizures in big doses or when genetically seizure-sensitive animals are used, yet it is also robustly anticonvulsant in certain seizure models. A lack of stereospecificity suggests that the mechanism may not be related to a single receptor interaction. Serotonin, γ-aminobutyric acid, acetylcholine or prostaglandin systems may be involved.
There are many anecdotal reports of beneficial effects in humans with epilepsy (Reference Grinspoon and BakalarGrinspoon & Bakalar, 1993) but research data are virtually non-existent. Two single-case reports (Reference Keeler and ReiflerKeeler & Reifler, 1967; Reference Consroe, Wood and BuchsbaumConsroe et al, 1975) give confounding information. A young man suffered seizures on his regular medication and began smoking several cannabis cigarettes nightly alongside this. No further seizures occurred while this combination was maintained. In contrast, a man with grand mal epilepsy stopped taking anticonvulsants and suffered no fits for 6 months. He then smoked cannabis on seven occasions over a 3-week period and suffered three fits during this time, although not coincident with actual intoxication.
Only one RCT (Reference Cunha, Carlini and PereiraCunha et al, 1980) exists. Fifteen poorly controlled patients with secondary generalised epilepsy continued with their regular therapy but were also given either cannabidiol or placebo daily for up to 4.5 months while under-going regular clinical and electroencephalogram evaluation. Half the patients on cannabidiol remained “almost free” of fits throughout the experiment, and all but one of the others showed “partial improvement”. All but one of the placebo patients remained entirely un-changed. Somnolence occurred in four patients receiving cannabidiol.
Asthma
Small-scale controlled studies in volunteers with asthma show that oral, smoked and aerosolised THC has comparable bronchodilatory activity to salbutamol, although onset is quicker with the latter. Dose-related tachycardia occurred in some individuals, and subjective intoxication with higher doses. A THC aerosol was free of systemic unwanted effects, but was irritant to the lungs (Reference Tashkin, Reiss and ShapiroTashkin et al, 1977). Nabilone does not produce bronchodilation. Since THC-induced bronchodilation is not mediated through the sympathetic nervous system, synergistic combinations with β2-adrenoceptor stimulants might be possible.
Other possible therapeutic applications
Basic research indicates that THC and analogues inhibit opioid withdrawal (Reference Chesher and JacksonChesher & Jackson, 1985). Anecdotal reports from patients also point to beneficial effects beyond those which could be accounted for by sedative or hypnotic activity. Cannabinoids inhibit primary tumour growth and increase survival in animal tumour models (Reference Harris, Munson, Carchman, Braude and SzaraHarris et al, 1976) by an unknown mechanism. They also show antipyretic and anti-inflammatory activity (Reference Formukong, Evans and EvansFormukong et al, 1989). Mechoulam (Reference Mechoulam and Mechoulam1986) has drawn attention to the lack of modern research directed at possible antihelmintic, antimigraine and oxytocic applications.
DISCUSSION
Therapeutic profile on existing evidence
Tetrahydrocannabinol and nabilone are effective anti-emetics but there are no comparisons with 5-HT3 antagonists, so a role in modern anti-emetic regimes remains to be determined. Currently, only nabilone is licensed in the UK and available for prescription and research. THC (as dronabinol) has recently been rescheduled to permit prescription but remains unlicensed and must be specially imported on a named-patient basis. Delta-8-THC looks worthy of further investigation, particularly in children, and is much simpler to synthesise than THC.
Many individuals with MS have claimed a benefit from cannabis and small controlled trials support this, although effect upon posture and balance requires clarification. THC is an effective analgesic at the expense of sedation with larger doses and may have special merit in neuropathic pain. No conclusions are possible as yet about anticonvulsant potential. Some cannabinoids reduce IOP, though side-effects of products currently available limit application and effects of tolerance are uncertain. The mechanism for bronchodilation probably differs from that of β2-stimulants, so synergistic combinations may be possible.
Cannabis and THC are effective appetite stimulants. Alongside anti-emetic, analgesic, anxiolytic, hypnotic and anti-pyretic properties this suggests a unique role in alleviating symptoms in selected patients with cancer or AIDS. This is a compelling area for future research, although possible effects upon immune function require careful monitoring.
Optimal doses and routes of delivery have not been established. Absorption by the oral route is unreliable. Smoking the drug is generally not a viable option since advantages such as rapid onset, accurate titration of effects and reliability in patients who are vomiting have to be set against the likelihood of lung irritation or damage, and it would in any case be unacceptable to most patients. However, pending availability of more satisfactory preparations, I believe that the existing profile of efficacy and toxicity justifies the provision of a legal supply of standardised herbal material (‘compassionate reefers’) to patients with terminal conditions who currently obtain relief with street cannabis. Sublingual sprays or tablets, nebulisers and aerosols look promising for the future, and THC is effective by the rectal route. Many potentially active cannabinoids have yet to be investigated and the recent identification of a peripheral receptor may lead to new drugs devoid of central nervous system effects.
Cannabis arouses passion in those who support or condemn it, and few people approach the clinical literature with dispassionate objectivity. Poorly controlled research produces ambiguous results which are then interpreted according to the prejudices of the reader. Anecdotes seem to be more readily accepted when they point to adverse rather than positive effects (Reference Hall, Solowij and LemonHall et al, 1994). Yet the known adverse effects of oral cannabinoids are rarely intolerable or life-threatening, in contrast to those associated with some standard therapies. A British Medical Association survey indicated that many UK doctors believe that cannabis should once again be available on prescription (Reference MeekMeek, 1994).
The way forward
A Select Committee of the House of Lords recently examined the scientific information concerning medical cannabis and took verbal and written evidence from a wide range of witnesses. Their conclusion (House of Lords, 1998) published in November 1998, was that, although cannabis should remain a controlled drug, the law should be changed to allow doctors to prescribe “ an appropriate preparation of cannabis if they saw fit”. The government rejected this recommendation on the day of publication.
Under the auspices of the Royal Pharmaceutical Society, large-scale multicentre trials are under way to explore further the efficacy of cannabinoids in relieving spasticity and postoperative pain. A pharmaceutical company has obtained a licence to cultivate medicinal cannabis on a large scale in the UK. By selecting a specific genotype then carefully controlling all other relevant variables such as soil conditions, temperature and humidity, it is possible to obtain levels of purity in plant extracts equal or superior to those of ‘pure’ synthetic cannabinoids. Most of the 60 or so naturally occurring cannabinoids are present in tiny amounts, and synthetic cannabinoids such as nabilone themselves contain up to 5% impurities, some of which are of unknown identity. Whether obtained by synthetic means or by plant extraction, it is essential that cannabinoids for prescription and research in the future should demonstrate excellent purity, stability and bioavailability.
The medicinal properties of cannabis are still mainly delineated by the anecdotal reports of those who believe their symptoms are relieved by its use, and these accounts are often dismissed as wishful thinking or even mischievous. Since the conventional treatments for many of these disorders are both toxic and relatively ineffectual, a more constructive response would be to expose such claims to careful scientific examination and, in the meantime, search for a way to avoid criminalising those who seek only to assuage their own suffering.
APPENDIX
Existing anti-emetics
Phenothiazzines and butyrophenones can cause sedation, movement disorders which may be irreversible, neuroleptic malignant syndrome, dry mouth, blurred vision, urinary retention, hypotension, allergic reactions, jaundice, hypothermia, hormonal disturbances, irreversible eye damage and, rarely, life-threatening anaemias. Domperidone has a more benign profile but is not recommended for long-term use. Metoclopramide produces movement disorders (1% of patients), dizziness and drowsiness. Selective 5-HT3 antagonists (ondansetron, granisetron) are newer and more expensive. Side-effects include constipation, headache, flushing, liver enzyme changes, allergic reactions, visual disturbances, chest pain and dysrhythmias.
Existing neurological treatments
Baclofen alleviates spasticity, but may accentuate muscle weakness. It produces dose-related nausea and vomiting, drowsiness, vertigo, confusion, fatigue and hypotonia. Less commonly, fits, psychiatric disorder and hypotension occur. Sudden withdrawal can cause hallucinations. Diazepam is useful but can worsen weakness or incoordination and cause drowsiness, ataxia, depression, disinhibition and dependence. Dantrolene may cause weakness, hypotonia, drowsiness, dizziness, vertigo and anxiety. Rarely, it damages the liver, and is not recommended in those with co-existing heart or lung disease.
Existing glaucoma treatments
Eye-drops. Miotics can produce blurring of vision, headache, and parasympathetic effects including sweating, bradycardia, colic and bronchospasm. Adrenaline often causes local discomfort. Dipivefrine and guanethidine may cause conjunctival fibrosis on chronic use. Beta-blockers may cause bradycardia, heart block or bronchoconstriction.
Systemic drugs (acetazolamide, dichlorphenamide) can cause hypokalaemia, appetite suppression, paraesthesiae, drowsiness, depression, rashes and, rarely, bone marrow suppression.
Clinical Implications and Limitations
CLINICAL IMPLICATIONS
-
▪ Cannabis and its derivatives show promise of beneficial effects in a number of medical conditions for which standard treatment is less than satisfactory, and further controlled research is fully justified.
-
▪ Cannabis is very safe in overdose, but often produces unwanted effects which are better tolerated by patients with some conditions (e.g. multiple sclerosis, chronic pain, AIDS, cancer) than others (e.g. glaucoma).
-
▪ Optimal formulations, doses and routes of delivery have not yet been established.
LIMITATIONS
-
▪ Because of imposed time constraints, the review is not fully comprehensive, although all accessed sources were incorporated.
-
▪ Much of the evidence is anecdotal, and many of the research studies cited have serious methodological shortcomings.
-
▪ Few researchers (or reviewers) approach the subject of cannabinoid therapeutics in a spirit of dispassionate objectivity.
ACKNOWLEDGEMENT
This review was originally commissioned and funded by the Department of Health. The author thanks Dr Anthony Thorley for his support and encouragement. The views expressed in the paper are those of the author and not necessarily of the Department of Health.








eLetters
No eLetters have been published for this article.