CHDs are the most common developmental anomaly, with incidence rates ranging from eight to 12 per 1000 live births. Reference Hoffman1 Atrial septal defects, ventricular septal defects, and atrioventricular septal defects are major contributors to CHD. Of these, 11.6% are atrial septal defect, 30.7% are ventricular septal defects, and 2% are atrioventricular septal defects. Reference Shabana, Shahid and Irfan2 In recent years, the aetiology of certain CHDs has increased significantly. The majority of the septal defects seen in the human population are the result of genetic modulation by epigenetic factors. Advancements in the understanding of the molecular basis of normal heart development demonstrate the necessity of numerous genes that are predominantly expressed during heart morphogenesis. Reference Bruneau3 The causes of cardiac septation defects in most cases are unknown, but the molecular evolution and gene analysis of the heart have led to the identification of some mutations associated with cardiac septation defects. Many cohort studies have shown that susceptible genes GATA4 and CITED2 are important during heart development. Reference Reamon-Buettner and Borlak4,Reference Bamforth, Bragança and Eloranta5 The mutations of these genes result in the failure of normal development of the heart, thereby leading to septal defects. Reference Garg, Kathiriya and Barnes6,Reference Sperling, Grimm and Dunkel7 However, few studies have focused on the Xinjiang region. This study aimed to identify the nucleotide sequence variation of the GATA4 and CITED2 genes in patients with cardiac septation defects from Xinjiang, China.
Materials and methods
Subjects
A total of 172 patients with cardiac septation defects were recruited from the People’s Hospital of the Xinjiang Uygur Autonomous Region, Xinjiang, China. There were 81 males and 91 females, ranging in age from 3 months to 61 years. These patients with CHD included 120 cases of atrial septal defect, 40 cases of ventricular septal defect, 10 cases of atrial septal defect complicated with ventricular septal defect, and two cases of atrioventricular septal defect. Among them, four cases had a family history of atrial septal defect. A summary of the clinical characteristics of the subjects is shown in Table 1. The evaluation of all subjects included their clinical history, physical examination, electrocardiogram, chest X-ray examination, and echocardiography. Several of them underwent cardiac catheterisation examination. All patients diagnosed with CHD underwent surgery. Syndromic CHDs, such as Noonan, DiGeorge, Holt–Oram, Marfan, Alagille, and Char syndromes, were excluded from this study. In addition, 200 unrelated healthy individuals (94 males and 106 females; age range, 3 months–60 years) were recruited as controls. There was no statistically significant difference in gender ratio or mean age between the CHD and control groups (p > 0.05). The study protocol was approved by the Institutional Research Ethics Committee of People’s Hospital of the Xinjiang Uygur Autonomous Region (KY2021101304). Peripheral blood samples from all participants were obtained after informed consent.
Table 1. Baseline clinical and demographic characteristics of the patients affected with CHDs (n = 172)
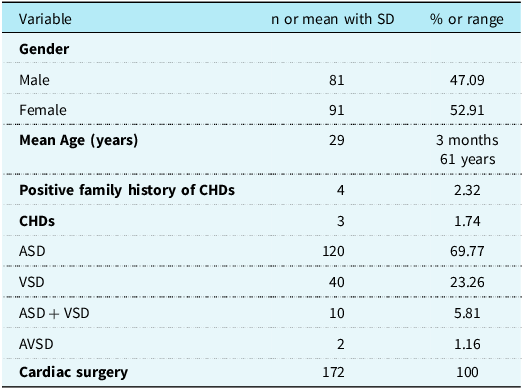
CHD = congenital heart defect; ASD = atrial septal defect; VSD = ventricular septal defect; AVSD = atrioventricular septal defect.
Blood samples from participants were stored in 2 mL vacuum tubes containing Ethylenediaminetetraacetic Acid. The blood samples were immediately transferred to the freezing tubes and stored at −80°C.
Deoxyribonucleic acid extraction
Whole blood genomic deoxyribonucleic acid extraction kit (Tiangen Biochemical Science and Technology Co. Ltd, Beijing, China) was utilised to extract genomic deoxyribonucleic acid among white blood cells. After extraction, an ultramicro UV spectrophotometer was used to detect the concentration and purity of the deoxyribonucleic acid samples, which were then stored in a −20°C refrigerator.
Polymerase chain reaction amplification and deoxyribonucleic acid sequencing
Primers were designed using Primer premier 5.0 for the amplification of the coding exons of the GATA4 (NM_002052.3) and CITED2 (NM_006079.4) genes (Table 2). Polymerase Chain Reaction was performed in 20 μL of standard Polymerase Chain Reaction buffer, containing 2 μL of genomic deoxyribonucleic acid, 1 μL of forward and reverse primers, 10 μL of Goldstar Best MasterMix, and 6 μL of water. The amplification programme was one cycle of an initial denaturation step at 95°C for 10 min; followed by 35 cycles at 95°C for 30 s, 64°C for 30 s, and 72°C for 30 min; and a final extension at 72°C for 10 min. Reactions were performed on an ABI 9700 Polymerase Chain Reaction machine (Applied Biosystems Inc., Carlsbad, CA, USA). Products were run on 1% agarose gel to verify amplification, and purified products were sequenced in the sense direction with an ABI 3730XL deoxyribonucleic acid Analyzer (Applied Biosystems, Inc., Foster City, CA, USA). Deoxyribonucleic acid sequence files were analysed using Chromas and DNAstar software. The pathogenicity of the identified missense variants was assessed in silico using Sorting Intolerant from Tolerant (http://sift.jcvi.org/), Polyphen2 (http://genetics.bwh.harvard.edu/pph2/), and Mutation Taster(www.mutationtaster.org) software.
Table 2. Summary of the primers and size of the GATA4 and CITED2 genes

Genetic and bioinformatics analysis
The protein sequences of GATA4 and CITED2 from other species were obtained from the National Center for Biotechnology Information database. All sequences were aligned using the MUSCLE programme (www.ebi.ac.uk/Tools/msa/muscle/). Sorting Intolerant from Tolerant (http://sift.jcvi.org/), PolyPhen-2 (http://genetics.bwh.harvard.edu/pph2/), and MutationTaster (www.mutationtaster.org) programmes were utilised to validate the mutation prediction of single-nucleotide variants. The structural stability of resultant proteins was predicted by I-Mutant 2.0 (https://folding.biofold.org/i-mutant/i-mutant2.0.html).
Data analysis
The data analysis was performed using SPSS 24.0. The comparison of categorical variables between two groups was conducted using either the chi-square (χ2) test or the Fisher exact test. Statistical significance was determined at a two-tailed p value of < 0.05.
Results
The frequencies of GATA4 and CITED2 variants
Four nonsynonymous variants of GATA4 and CITED2 were identified in the four CHD patients, but not in the healthy control group, resulting in an overall detection frequency of 2.33% in the patient cohort. Among the CHD patients, three reported GATA4 variants were observed in three individuals, accounting for 1.74% of the patient group. Additionally, one novel CITED2 variant was detected in one patient, with a detection frequency of 0.58%. The frequencies of genotypes and alleles for the four missense variants in both CHD patients and controls are presented in Table 3.
Table 3. Frequencies of genotypes and alleles in CHD patients and controls

Phenotypes of GATA4 and CITED2 variants
Our analysis of these patients with CHD identified three reported variants (c.1138G > A, p.V380M; c.1180C > A, p.P394T; and c.1220C > A, p.P407Q) in the GATA4 gene (Fig. 1). The p.V380M variant was discovered in a patient with atrial septal defect. The p.P394T variant was noted in a patient with atrial septal defect. One patient with ventricular septal defect carried both p.V380M and p.P407Q variants. A homozygous variant in the CITED2 gene, c.574A > G(;p. Sl92G);was found in a patient suffering from ventricular septal defect and validated as the first reported variant (Fig. 2). A detailed description of the specific sequence variants of the GATA4 and CITED2 genes and the cardiac phenotype of patients is summarised in Table 4.

Figure 1. Sequencing results of GATA4 variations. a : Heterozygous c.1138 G > A variation. b : Heterozygous c.1180 C > A variation. c : Heterozygous c.1220 C > A variation.

Figure 2. Sequencing result of CITED2 variation. Homozygous c.574 A > G variation. a : Homozygous A/A. b : Homozygous G/G.
Table 4. Detailed description of GATA4 and CITED2 nonsynonymous variations identified in this study

SIFT, “D” meaning deleterious, “T” meaning tolerated; PolyPhen2, “D” meaning probably damaging, “P” meaning possibly damaging, “B” meaning benign; MutationTaster, “A” represents as disease-causing automatic meaning known deleterious reported in HGMD/ClinVar/dbSNP, “D” represents as disease-causing meaning likely deleterious, “N” represents polymorphism or likely harmless, “P” represents polymorphism automatic meaning known harmless. I-Mutant v2.0, DDG: DG(NewProtein)-DG(WildType) in Kcal/mol, DDG < 0: Decrease Stability, DDG > 0: Increase Stability.
# One patient with VSD carried both p.V380M and p.P407Q variants in GATA4 gene.
DDG = Delta Delta G.
Identification of novel variant
We have identified a previously unreported variant in the CITED2 gene. This variant, specifically the homozygous c.574A > G variant, resulted in the p. Sl92G variant. This variant was observed in a patient with ventricular septal defect and has been confirmed as the first reported variant of its kind (Fig. 2). The alignment of CITED2 sequences, depicted in Figure 3, demonstrates evolutionary conservation across multiple species. The p. Sl92G variant of CITED2 was predicted to have deleterious effects, although it was also found to be tolerated and polymorphic. The stability of the resulting proteins was assessed using I-Mutant 2.0. This particular variant has been verified to induce a reduction in the stability of the resulting protein, as indicated in Table 4.
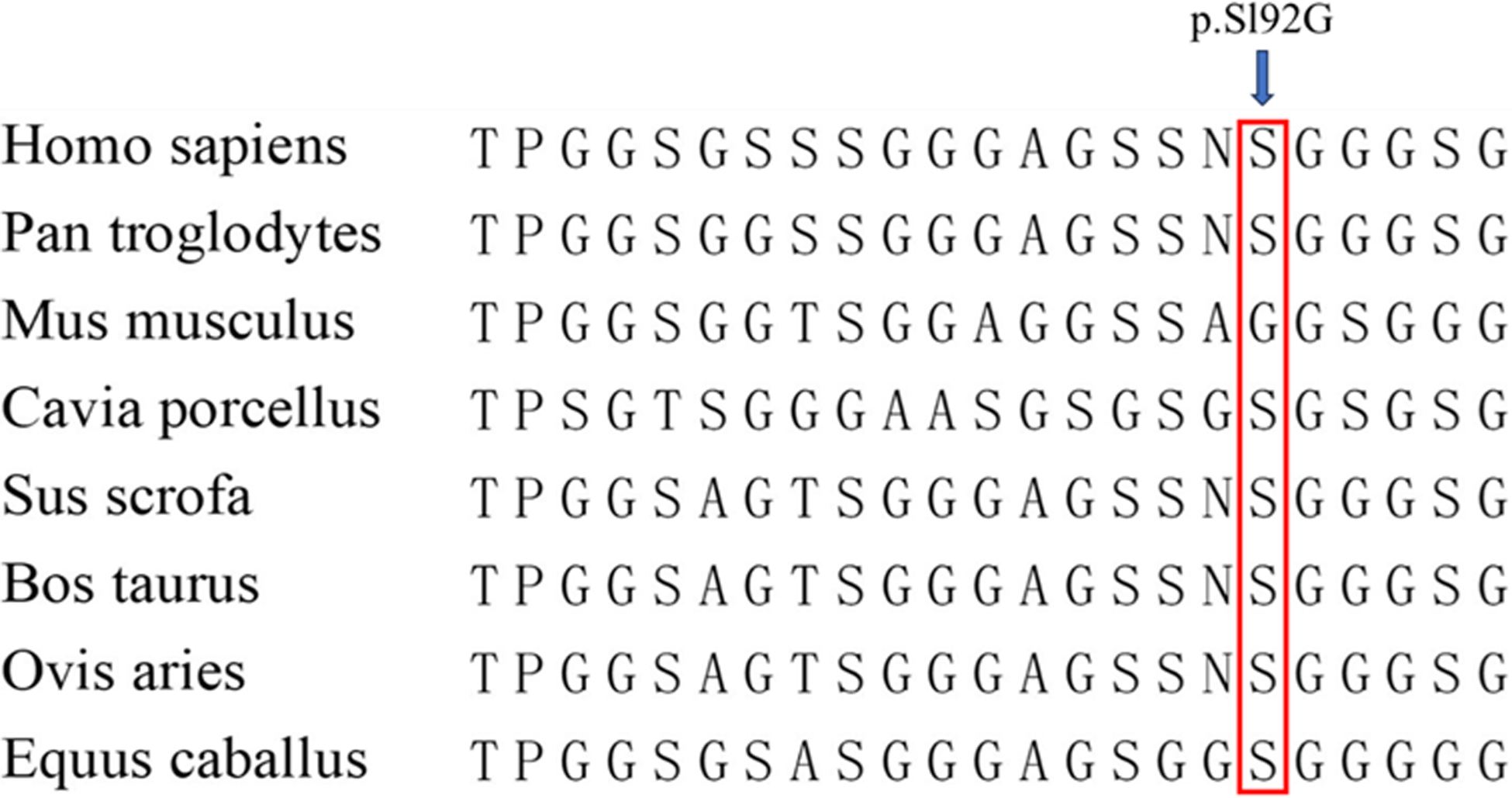
Figure 3. The alignment of the CITED2 sequence. The analysis of amino acid conservation at the variation site indicates that the arrow represents the CITED2 variation site (p.S192G), which demonstrates evolutionary conservation across most species.
Discussion
GATA4 is a master regulator required for ventral morphogenesis and heart tube formation. Reference Molkentin, Lin and Duncan8 The coding region of GATA4 includes two transactivation domains, two forms of type IV zinc-finger motif, and a nuclear localisation signal. The highly preserved N-terminal zinc finger interacted with Fog2, and the C-terminal zinc finger interacted with transcription factors to regulate the expression of cardiac genes. Evidence from these studies confirmed that GATA4 plays an important role embryonic heart development and associated mutations are linked with various cardiac abnormalities. Reference Narita, Bielinska and Wilson9 GATA4 has also been identified as a potential aetiology of human dilated cardiomyopathy. It has been observed that GATA4 mutants demonstrate a noteworthy decrease in transcriptional activity, thereby contributing to the pathogenesis of pathological cardiac hypertrophy. Reference Zhao, Xu and Xu10 Furthermore, a significant correlation has been noted between GATA4 mutations and the prevalence of atrial fibrillation. Reference Jiang, Shen, Fang, Liu and Yang11 There have been more than 100 mutations of GATA4 reported over the past decades. Reference Yu, Lei and Yang12 Its mutation is most widely studied in CHD with various clinical phenotypes, including atrial septal defect, ventricular septal defect, atrioventricular septal defect, tetralogy of Fallot, pulmonary stenosis, patent ductus arteriosus, double outlet right ventricle, and hypoplastic left-heart syndrome. Atrial septal defect is the most common clinical phenotype carrying the GATA4 gene mutation in the CHD family. Reference Okubo, Miyoshi and Baba13,Reference Xiang, Fan and Huang14
We identified three heterozygous variants of GATA4 (p.V380M, p.P394T, and p.P407Q) in the CHD group. These variants have not been previously reported in previous studies. Tang et al. (2006) first found p.V380M variant in a patient with CHD, and they reported it as a disease-causing mutation in CHD. Reference Tang, Xia and Chang15 The p.V380M variant has been implicated in a variety of CHDs, including ventricular septal defect, atrial septal defect, atrioventricular septal defect, and tetralogy of Fallot. Reference Tang, Xia and Chang15–Reference Qian, Xiao and Guo20 In this study, the p.V380M variant was discovered in a patient with atrial septal defect (1/172), but it was not found in our control cohort. The p.V380M variant of the GATA4 gene is rare in cases with CHD in Chinese populations. Reference Tang, Xia and Chang15,Reference Qian, Xiao and Guo20 Liu et al. detected p.V380M variant in four patients (4/600) including tetralogy of Fallot, double outlet right ventricle, PA and ventricular septal defect, and transposition of the great arteries, as well as one in controls (1/300), in China. Reference Liu, Li and Xu21 Several studies have observed the p.V380M variant to be present at a very low frequency in populations from North America, Japan, and India. However, these studies did not establish a correlation between this single-nucleotide variant and CHD. Reference Schluterman, Krysiak and Kathiriya22–Reference Dixit, Narasimhan and Balekundri26 Notably, our analysis demonstrated a statistically significant higher occurrence of the SNP c.1138G > A (p.V380M) in individuals with CHD compared to the control group (Table 5).
Table 5. A list of the three nonsynonymous variants of GATA4 identified among CHD patients and controls in previous studies

ASD = atrial septal defect; AVSD = atrioventricular septal defect; COA = coarctation of aorta; DORV = double outlet right ventricle; LV = left ventricle; PA = pulmonary atresia; PAVSD = pulmonary atresia with ventricular septal defect; PDA = patent ductus arteriosus; PHT = pulmonary hypertension; PS = pulmonary stenosis; PTA = Persistent truncus arteriosus; SV = single ventricle; TGA = transposition of the great arteries; TOF = tetralogy of Fallot; VSD = ventricular septal defect.
One patient with atrial septal defect presented with p.P394T variant for GATA4 in this study. This nucleotide change was not found in the control population. Schluterman et al. identified the p.P394T variant in an individual of Asian ethnicity with CHD (the examined individuals were of variable ethnicity and composed of 76 European Americans, 53 Hispanics, 21 African Americans, and 7 Asians). The variant was not found in the original control population of 159 individuals. The allele frequency was 3.1% in 192 control individuals of Asian ethnicity, which likely represented an SNP in the Asian subpopulation (not specified). Reference Schluterman, Krysiak and Kathiriya22 The p.P394T variant was found in the affected and control populations and likely represented common SNPs in India. Reference Dinesh, Lingaiah and Savitha23,Reference Dixit, Narasimhan and Balekundri26 The variant of the GATA4 gene in Chinese and Japanese populations was rare in cases with CHD and was not identified in any of the controls. Reference Qian, Xiao and Guo20,Reference Liu, Li and Xu21,Reference Kodo, Nishizawa and Furutani24,Reference Yoshida, Morisaki and Nakaji25,Reference Zhang, Li and Shen27,Reference Wang, Lu and Chen28 Some reports did not detect an association between this single-nucleotide variant and CHD in an Indian population (Table 5).
The p.P407Q variant was reported to be pathogenic in the Human Gene Mutation Database and Clin-Var. The p.P407Q variant has been implicated in a variety of CHDs, including ventricular septal defect and tetralogy of Fallot. Reference Qian, Xiao and Guo20,Reference Dixit, Narasimhan and Balekundri26–Reference Peng, Wang and Zhou29 In most Chinese studies, the p.P407Q variant was found only in patients with CHD. Reference Qian, Xiao and Guo20,Reference Zhang, Li and Shen27–Reference Peng, Wang and Zhou29 Only Liu et al. detected p.P407Q variant in four patients (4/600) including tetralogy of Fallot, double outlet right ventricle, PA and ventricular septal defect, and transposition of the great arteries, as well as one in controls (1/300), in China. Reference Liu, Li and Xu21 However, the p.P407Q variant was previously reported as an single-nucleotide variant in Japanese, Canadian, and American populations. Reference Kodo, Nishizawa and Furutani24,Reference Yoshida, Morisaki and Nakaji25 These analyses revealed that p.P407Q variant was found significantly more frequently in CHD cases than in controls (Table 5). One patient with ventricular septal defect carried two variants (p.V380M and p.P407Q) in this study. Two nonsynonymous variations of the GATA4 gene were rarely detected from one patient with CHD. Qian et al. found two variants of GATA4 (p.V380M and p.P407Q) in two patients with tetralogy of Fallot and PFO. Reference Qian, Xiao and Guo20
The identified variations in GATA4 were exclusively observed in the cases group, while no occurrences were found in the control group within our study. The minor allele frequency of three GATA4 variations was assessed using the GnomAD and GnomeAsia databases. Interestingly, in certain studies, these variations were detected in both cases and healthy controls at a significantly higher frequency (Table 5). This observation implies that the GATA4 gene variant may exhibit racial and regional disparities.
The structural stability of the resultant proteins was assessed using I-Mutant 2.0, which provided an expression of the output in terms of free energy change value (Delta Delta G). Among the three variants of GATA4 that were examined, it was confirmed that they caused a decrease in stability of the resultant proteins. Based on a Delta Delta G value < −0.5, it can be inferred that these variants exert a substantial influence on the proteins. The stability of proteins plays a pivotal role in determining their biological activity and functionality, making it a crucial factor. Mutations that result in reduced protein stability could potentially contribute to the onset of CHD.
CITED2 is a cAMP responsive element-binding protein/p300 transactivator, with an Glutamate Glu(E) Aspartate Asp(D) (Glu/Asp)-rich C-terminal domain that functions as an important modulator in heart development. Reference MacDonald, Bamforth and Chen30 Both human genetic and model organism studies have shown that altered expression of CITED2 causes various forms of CHD. CITED2 mutations lead to a constellation of cardiac defects. Since Sperling et al. first uncovered the connection between CHDs and CITED2 mutations in 2005, Reference Sperling, Grimm and Dunkel7 a growing number of mutations have been reported to cause CHD. Reference Chen, Bentham and Cosgrove31–Reference Yadav, Jain and Neelabh38 Most of these mutations are located in the SRJ region, which indicates that this region might be a mutation hot spot in patients with CHD. Reference Sperling, Grimm and Dunkel7,Reference Chen, Bentham and Cosgrove31–Reference Liu, Wang and Wu34,Reference Dianatpour, Khatami and Heidari37 In this study, we have identified a homozygous variation (c.574 A > G, p. Sl92G) of CITED2 in a patient with ventricular septal defect. This variant has been validated as the first reported variant and was also found in the SGJ region. To assess the sequence conservation of CITED2, we conducted multiple sequence alignment (Fig. 3), revealing its evolutionary conservation across most species. Furthermore, the mutation prediction results indicate that the p. Sl92G variant is likely to be disease-causing, as it is deemed deleterious in Sorting Intolerant from Tolerant (Table 4). The structural stability of resultant proteins was predicted by I-Mutant 2.0. The variants of CITED2 were confirmed to cause decrease in stability to the resultant proteins (Delta Delta G value < −0.87), indicating its greater impact towards the proteins. Proteins with decreased stability due to mutation might give rise to CHD. The I-Mutant 2.0 tool was utilised to forecast the structural stability of the resulting proteins. It was determined that the variants of CITED2 induce a reduction in stability of the resulting proteins, as evidenced by a Delta Delta G value below -0.5. This observation suggests that the impact of the variant on the proteins is significant. Consequently, the diminished stability of proteins resulting from mutations may contribute to the development of CHD.
The heterozygous variation (c.574A > G, p. Sl92G, and rs563655306) in CITED2 was previously identified in patients with aortic stenosis Reference Li, Pan and Guan32,Reference Xu, Wu and Li33 and ventricular septal defect (https://www.hgmd.org). Li et al. found that p. Sl92G in CITED2 results in the loss of the ability to mediate the expression of VEGF by disabling competitive binding with HIF1-α to cAMP responsive element-binding protein/p300. Reference Li, Pan and Guan32 Therefore, p. Sl92G mutations affect the transcriptional regulatory properties of PITX2C and VEGF, and this effect is closely related to cardiac development. These data provide important evidence that the mutation of CITED2 may play a causative role in the development of CHD in humans. Further investigation is warranted to explore the in silico and in vitro functional validation of variations in CITED2.
Conclusion
Our study indicated that nonsynonymous sequence variants in the GATA4 and CITED2 genes are rare and do not appear to play any significant pathogenic role in some patients with cardiac septal defects. We found that the p.V380M, p.P394T, and p.P407Q variants of the GATA4 gene differed greatly in many studies. This gene may differ among races and regions, and it may not be an important contributor to subjects with sporadic CHD in Xinjiang. Our findings also supported the notion that the p. Sl92G variant of the CITED2 gene could be pathogenic. These findings show that most CHDs have a multifactorial aetiology. GATA4 and CITED2 gene mutations may be the causes of idiopathic CHD in several patients. Further studies are warranted to unravel the genetic basis of cardiac defects.
Acknowledgements
The authors want to thank all patients and their families for participating this study.
Financial support
This research was supported by institutional funds.
Competing interests
None.
Ethical standards
The study was approved by the Ethics Committee of People’s Hospital of the Xinjiang Uygur Autonomous Region.











