Pancreatic cancer is the most fatal of all gastrointestinal cancers, with a survival rate of only 6 % at 5 years( 1 ). This cancer is characterised by a high mortality rate, rapid progression, and resistance to chemotherapy and radiation( Reference Li, Albrecht and Li 2 ). There is growing evidence on the role of chronic inflammation in pancreatic cancer( Reference Farrow and Evers 3 , Reference Jackson and Evers 4 ), as in other neoplasms( Reference Touvier, Fezeu and Ahluwalia 5 , Reference Keibel, Singh and Sharma 6 ). While inflammation typically occurs as part of the body's response to tissue insult/injury( Reference Keibel, Singh and Sharma 6 , Reference Pan, Lai and Dushenkov 7 ), chronic inflammation is a persistent condition in which tissue destruction and repair occur simultaneously( Reference Coussens and Werb 8 , Reference Philip, Rowley and Schreiber 9 ).
Results from human studies on the association between the risk of pancreatic cancer and inflammation have been mixed so far. A case–control study has shown that the levels of C-reactive protein and IL-6 were higher among pancreatic cancer patients than among those without cancer( Reference Moses, Maingay and Sangster 10 ) and, in another case–control study, pancreatic adenocarcinoma patients have been shown to have higher levels of TNF-α and interferon-γ than those with chronic pancreatitis and healthy volunteers( Reference Talar-Wojnarowska, Gasiorowska and Smolarz 11 ). Combined results from three cohort studies conducted in the USA have also shown that inflammation is associated with pancreatic cancer over a long follow-up period (>10 years)( Reference Jacobs, Newton and Silverman 12 ). However, in a nested case–control study from the European Prospective Investigative Cohort, no associations were observed between C-reactive protein and IL-6 and the risk of pancreatic cancer, although there was a positive association with soluble TNF receptor 1 among women( Reference Grote, Kaaks and Nieters 13 ). A combined analysis of five cohort studies has shown no association between inflammatory markers and the risk of pancreatic cancer( Reference Bao, Giovannucci and Kraft 14 ). Pancreatic cancer has been positively associated with chronic pancreatitis( Reference Duell, Lucenteforte and Olson 15 ) but inversely with allergy, a condition known to be related to inflammation( Reference Gandini, Lowenfels and Jaffee 16 – Reference Olson, Hsu and Satagopan 19 ).
Diet represents a complicated set of exposures that often interact and whose cumulative effect modifies both inflammatory responses and health outcomes. According to the latest report from the World Cancer Research Fund. American Institute for Cancer Research, which was based on the systematic reviews of cohort and case–control studies, both decaffeinated and caffeinated coffee have not been found to be associated with the risk of pancreatic cancer, while results for other food items, such as red and processed meat, or nutrients such as total fat and saturated fat, are still unclear( 20 ). Although several studies have been conducted, the relationship between diet and the risk of pancreatic cancer is still unclear( Reference Ji, Chow and Gridley 21 – Reference Jansen, Robinson and Stolzenberg-Solomon 24 ). In a case–control study conducted in the USA, nutrients from fruits and vegetables have been found to reduce the risk of pancreatic cancer( Reference Jansen, Robinson and Stolzenberg-Solomon 24 ). Similarly, consumption of dietary antioxidants has been found to be protective against developing pancreatic cancer in the European Prospective Investigative Cohort-Norfolk study( Reference Banim, Luben and McTaggart 25 ). In a pooled analysis carried out using data from fourteen cohort studies, consumption of dairy foods, Ca or vitamin D during adulthood was not associated with the risk of pancreatic cancer( Reference Genkinger, Wang and Li 26 ), and no association has been observed between intake of vegetables, fruits, carotenoids and vitamins C and E and the risk of pancreatic cancer in The Netherlands Cohort Study( Reference Heinen, Verhage and Goldbohm 23 ). In the National Institute of Health–American Association of Retired Professionals cohort study conducted in the USA, a high-quality diet, as indicated by increasing Healthy Eating Index 2005 scores, has been found to be protective against pancreatic cancer( Reference Arem, Reedy and Sampson 27 ), and in a case–control study conducted in Italy, the Mediterranean diet score has been inversely associated with pancreatic cancer( Reference Bosetti, Turati and Dal Pont 28 ). These results further stress the importance of studying diet in its entirety rather than studying individual dietary components. In a case–control study conducted in California, a positive association was observed between the risk of pancreatic cancer and a Western dietary pattern, characterised by a higher intake of red and processed meat, salty high-fat snacks, sugary beverages, sweets, high-fat dairy products, eggs and refined grains( Reference Chan, Gong and Holly 22 ). In an Italian case–control study, a starch-rich dietary pattern has been associated with the increased risk of pancreatic cancer, while inverse associations have been reported for diets rich in vegetables, fruits and vitamins( Reference Bosetti, Bravi and Turati 29 ). However, the evidence is still inconsistent in two cohort studies( Reference Heinen, Verhage and Goldbohm 23 , Reference Jansen, Robinson and Stolzenberg-Solomon 24 ). The possible relationship between diet-related inflammation and the risk of pancreatic cancer has not yet been investigated.
The paucity of research related to diet and inflammation is probably due to logistical issues resulting from methodological complexity involved in linking diet, inflammation and cancer risk in the same study. In an effort to fill obvious methodological gap, researchers at the University of South Carolina's Cancer Prevention and Control Program developed a dietary inflammatory index (DII), which can be used in diverse populations in order to predict the levels of inflammatory markers associated with diet and related health outcomes( Reference Shivappa, Steck and Hurley 30 , Reference Shivappa, Steck and Hurley 31 ). Developing the DII involved careful review and scoring of the scientific literature on diet and inflammation, and obtaining datasets from around the world with which individuals’ dietary intakes could be compared( Reference Shivappa, Steck and Hurley 30 ).
The purpose of the present study was to examine the association between the DII and the risk of pancreatic cancer in a case–control study conducted in Italy. We hypothesised that subjects consuming a pro-inflammatory diet, as represented by a higher DII score, will exhibit an increase in the levels of circulating inflammatory cytokines, resulting in a higher risk of developing pancreatic cancer.
Methods
Full details of the case–control study have been given elsewhere( Reference Polesel, Talamini and Negri 32 ). Briefly, the study was conducted between 1991 and 2008 in the province of Pordenone and in the greater Milan area, northern Italy. Cases were 326 patients (174 men, 152 women; median age 63 years) admitted to the major teaching and general hospitals in the areas under study with incident pancreatic carcinoma. Controls were 652 patients (348 men, 304 women; median age 62 years) admitted to the same hospitals as cases for a wide spectrum of acute conditions other than neoplastic or digestive tract diseases related neither to known risk factors for pancreatic cancer nor to long-term dietary modifications. Controls were frequency-matched to cases by study centre, sex and age ( ± 5 years), with a control:case ratio of 2:1. More than 95 % of cases and controls who were approached agreed to participate in the study.
Information on sociodemographic characteristics, anthropometric measures, lifestyle habits, including smoking and alcohol drinking, personal medical history, and family history of cancer in first-degree relatives was assessed during the hospital stay of the subjects using a standard questionnaire administered by trained interviewers. To determine the usual diets of the subjects during the 2 years before cancer diagnosis or hospital admission (for controls), an interviewer-administered FFQ was used, which included seventy-eight foods and beverages, as well as a range of the most common Italian recipes. The subjects were asked to indicate the average weekly frequency of consumption of each dietary item; intakes less than once per week, but at least once per month, were coded as 0·5 per week. Nutrient and total energy intakes were determined using an Italian food composition database( Reference Gnagnarella, Parpinel and Salvini 33 ). The FFQ showed satisfactory validity( Reference Decarli, Franceschi and Ferraroni 34 ) and reproducibility( Reference Franceschi, Negri and Salvini 35 , Reference Franceschi, Barbone and Negri 36 ), with Spearman's correlation coefficients ranging between 0·60 and 0·80 for most food items and nutrients.
The present study was conducted according to the guidelines laid down in the Declaration of Helsinki, and all eligible patients agreed to participate in the study by signing an informed consent approved by the internal review boards of the study hospitals.
The FFQ-derived dietary information was used to calculate the DII scores for all of the subjects, using a procedure described in detail elsewhere( Reference Shivappa, Steck and Hurley 30 ). Briefly, to compute the DII, dietary data for each study subject were first linked to a global database derived from surveys conducted in eleven countries worldwide that included data on the consumption of forty-five food/nutrient parameters used in the computation of the DII (i.e. foods such as garlic and onions, nutrients such as saturated fat and vitamin C, and other food components such as flavonoids)( Reference Shivappa, Steck and Hurley 30 ). For each subject and each food parameter, a z score was derived by subtracting the ‘standard global mean’ from the amount reported and dividing this value by the standard deviation. To minimise the effect of ‘right skewing’ (a common occurrence with dietary data), this value was then converted to a centred percentile score, which was then multiplied by the respective food parameter effect score (derived from a literature review and scoring of 1943 articles) to obtain the food parameter-specific DII score of the subjects. All food parameter-specific DII scores were then summed to create the overall DII score for each subject in the study. The higher DII scores reflected a greater pro-inflammatory potential of the diet. The study had data on thirty-one of the forty-five food parameters studied for the development of the DII: carbohydrate; protein; fat; alcohol; fibre; cholesterol; SFA; MUFA; PUFA; n-3; n-6; niacin; thiamin; riboflavin; vitamin B6; Fe; Zn; vitamin A; vitamin C; vitamin D; vitamin E; folic acid; β-carotene; anthocyanidins; flavan-3-ol; flavonol; flavonones; flavones; isoflavones; caffeine; tea. A more detailed description of the validation of the DII score, based on both dietary recalls and a structured questionnaire similar to a FFQ, is available elsewhere( Reference Shivappa, Steck and Hurley 31 ). BMI was calculated as weight (in kg) divided by height (in m) squared, and was categorised into normal weight (BMI < 25·0 kg/m2), overweight (25·0 kg/m2≤ BMI < 30·0 kg/m2) and obese (BMI ≥ 30·0 kg/m2).
The DII was analysed both as a continuous variable (i.e. a one-unit increment in the DII corresponds to approximately 7 % of its global range) and by quintiles of exposure. The DII also was examined across the strata of factors such as age, education, BMI, smoking status, alcohol drinking and history of diabetes using the ANOVA test for continuous variables or the χ2 test for categorical variables. OR and the corresponding 95 % CI were estimated using logistic regression models, adjusting only for age, and then additionally for sex, study centre, year of interview (continuous), education, ( < 7, 7–11 and ≥ 12 years) and major recognised risk factors for pancreatic cancer, i.e. BMI ( < 25·0 kg/m2, 25·0–29·9 kg/m2 and ≥ 30·0 kg/m2), smoking status (never smoker, ex-smoker and current smoker), alcohol drinking (0, < 21 and ≥ 21 drinks/week) and history of diabetes (yes/no). Energy adjustment was performed by using the residual method( Reference Ferrari, Rinaldi and Jenab 37 ). Linear tests for trend were performed using the median value within each quintile as an ordinal variable. Stratified analyses were carried out across the strata of the following factors: sex; BMI; smoking status. Statistical tests were performed using SAS® 9.3 (SAS Institute, Inc.). All P values were two-sided.
Results
Table 1 shows the distribution of 326 cases of pancreatic cancer and 652 controls according to study centre, sex, age and other selected covariates. By design, cases and controls had similar distribution by study centre, sex and age. Compared with the controls, the cases had a higher level of education, were more frequently heavy drinkers and smokers, had normal BMI and reported more frequently a history of diabetes.
Table 1 Distribution of 326 patients with pancreatic cancer and 652 controls according to study centre, sex, age, education, and other selected variables in a case–control study conducted in Italy between 1991 and 2008 (Number of subjects and percentages)

* The sum does not add up to the total because of some missing values.
The mean DII value among the cases was 0·26 (sd 1·44) and among the controls was − 0·13 (sd 1·40), indicating a more pro-inflammatory diet for cases. The characteristics of the subjects across the quintiles of the DII are presented in Table 2. There were small differences in sociodemographic, anthropometric and lifestyle habits across the quintiles of the DII. There were a higher percentage of current smokers in the higher quintiles of the DII than that in the first quintile.
Table 2 Characteristics of 652 controls across the quintiles of the energy-adjusted dietary inflammatory index (DII), in a case–control study conducted in Italy between 1991 and 2008 (Mean values and standard deviations; number of subjects and percentages)
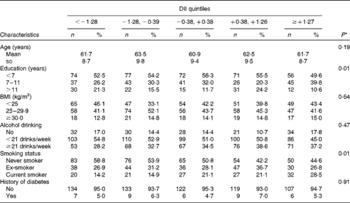
* ANOVA and χ2 tests were used for continuous and categorical variables, respectively.
The OR of pancreatic cancer according to quintiles and continuous DII are shown in Table 3. In age-adjusted models, positive associations were observed between the DII and pancreatic cancer (ORcontinuous 1·22, 95 % CI 1·11, 1·34 and ORquintile5 v. 1 2·10, 95 % CI 1·35, 3·24; P trend= 0·004). Significant positive associations were found in multivariate analysis, with an ORcontinuous of 1·24 (95 % CI 1·11, 1·38). Similarly, when the analysis was carried out with the DII expressed as quintiles, subjects in the second, third, fourth and fifth quintiles of the DII had increased OR for pancreatic cancer (ORquintile2 v. 1 1·70, 95 % CI 1·02, 2·80; ORquintile3 v. 1 1·91, 95 % CI 1·16, 3·16; ORquintile4 v. 1 1·98, 95 % CI 1·20, 3·27; ORquintile5 v. 1 2·48, 95 % CI 1·50, 4·10) compared with those in the lowest quintile of the DII (P trend= 0·0015).
Table 3 Odds ratios of pancreatic cancer for the energy-adjusted dietary inflammatory index (DII) among 326 cases and 652 controls, in a case–control study conducted in Italy between 1991 and 2008 (Odds ratios and 95 % confidence intervals)

* Continuous OR for a one-unit increment of the DII, corresponding to approximately 7 % of its global range.
† Reference category.
‡ Adjusted for age, sex, study centre, year of interview, education, BMI, smoking status, alcohol drinking, and history of diabetes.
When stratified by sex, a significant association between the DII and the risk of pancreatic cancer was observed for women (ORquintile5 v. 1 3·71, 95 % CI 1·73, 7·94; P trend= 0·001; Table 4). When stratified by BMI, a significant association between the DII and the risk of pancreatic cancer was observed for normal-weight subjects (BMI < 25 kg/m2: ORquintile5 v. 1 2·24, 95 % CI 1·03, 4·86; P trend= 0·16) and overweight subjects (BMI 25·0–29·9 kg/m2: ORquintile5 v. 1 2·32, 95 % CI 1·03, 5·21; P trend= 0·005). When stratified by smoking status, a significant association between the DII and the risk of pancreatic cancer was observed for never smokers (ORquintile5 v. 1 2·32, 95 % CI 1·08, 4·99; P trend= 0·01) and ex-smokers (ORquintile5 v. 1 3·37, 95 % CI 1·22, 9·35; P trend= 0·07).
Table 4 Odds ratios* of pancreatic cancer for the energy-adjusted dietary inflammatory index (DII), stratified by sex, BMI and smoking status, among 326 cases and 652 controls in a case–control study conducted in Italy between 1991 and 2008 (Odds ratios and 95 % confidence intervals)
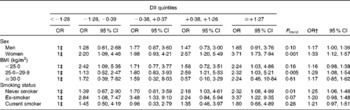
* Adjusted for age, sex, study centre, year of interview, education, BMI, smoking status, alcohol drinking, and history of diabetes.
† Continuous OR for a one-unit increment of the DII, corresponding to approximately 7 % of its global range.
‡ Reference category.
Discussion
The present study demonstrated a positive association between the DII and the risk of pancreatic cancer, with risk estimates being statistically significant for the DII expressed both as a continuous variable and by quintiles. This result supports the hypothesis that people eating a pro-inflammatory diet are at a higher risk of developing pancreatic cancer.
Some dietary factors have been shown to increase the risk of developing pancreatic cancer, including animal protein and (red) meat( Reference Genkinger, Wang and Li 26 , Reference Paluszkiewicz, Smolińska and Dębińska 38 , Reference Lucenteforte, Talamini and Bosetti 39 ). Other dietary factors, such as vegetables and fruits, have been shown to reduce the risk( Reference Banim, Luben and McTaggart 25 , Reference Jansen, Robinson and Stolzenberg-Solomon 24 , Reference Paluszkiewicz, Smolińska and Dębińska 38 ). In particular, previous research conducted in this case–control study database has revealed a strong positive association between the risk of pancreatic cancer and increased intake of (red) meat, other animal products, (refined) cereals and sugars( Reference Polesel, Talamini and Negri 32 ), and for a Western dietary pattern( Reference Bosetti, Bravi and Turati 29 ); however, fruit and vegetable intake( Reference Polesel, Talamini and Negri 32 ) and increasing adherence to a Mediterranean diet( Reference Bosetti, Turati and Dal Pont 28 ) appeared to exert a protective effect( Reference Bosetti, Bravi and Turati 29 ). A normal human diet consists of both pro-inflammatory and anti-inflammatory foods/nutrients. The DII score is designed to take this into account. Thus, it more accurately reflects the potential inflammatory effect of diet as a whole. Thus far, the DII has been successful in predicting C-reactive protein in the Seasonal Variation of Blood Cholesterol Study( Reference Shivappa, Steck and Hurley 31 ) and IL-6 in a case–control study conducted in Australia( Reference Wood, Shivappa and Berthon 40 ). An older version of this index has been used by a group in The Netherlands to predict inflammatory biomarkers (i.e. C-reactive protein, IL-6, IL-8, TNF-α and serum amyloid A) and soluble intercellular adhesion molecule-1( Reference Hebert, Shivappa and Tabung 41 , Reference van Woudenbergh, Theofylaktopoulou and Kuijsten 42 ). Using the data from the National Health and Nutrition Examination Survey, the new version of DII also has been used to compare the inflammatory potential of diet among workers assigned to different shifts( Reference Wirth, Burch and Shivappa 43 ). Additionally, the DII has been shown to predict prostate cancer among Italian men( Reference Shivappa, Bosetti and Zucchetto 44 ) and colorectal cancer among women in the Iowa Women's Health Study( Reference Shivappa, Prizment and Blair 45 ).
One of the possible mechanisms responsible for the association between a high-inflammatory diet and the risk of pancreatic cancer is increased production of cytokines in cellular organelles, which can lead to the release of proteolytic enzymes and reactive oxygen species. These, in turn, can produce genotoxic effects such as DNA strand breaks, sister chromatid exchanges, mutations, and formation of adducts with DNA( Reference Farrow and Evers 3 ). Another mechanism is through the effect of growth factors: chronic inflammation in the pancreas leads to an increased release of growth factors such as platelet-derived growth factor and transforming growth factor-α, which leads to increased proliferation of pancreatic cells( Reference Kalthoff, Roeder and Brockhaus 46 ).
The influence of diet on cancer is difficult to estimate, and challenges in assessing dietary exposures are greatest in case–control studies. Potential selection and recall bias of hospital-based case–control studies also should be considered. Dietary habits of hospital controls may be different from those of the general population; however, we were careful to exclude from the control group all diagnoses that could be associated with long-term dietary modifications. A recent diagnosis of cancer may have influenced recall of diet for the cases, although we asked for dietary habits 2 years before cancer diagnosis, and the role of diet in pancreatic cancer was unknown in the general Italian population at the time of the study. The use of a similar interview setting for both cases and controls would reduce potential information bias. Some of the associations observed in the present study, in particular the inverse ones with vegetables, and the direct ones with (red) meat, are apparently stronger than those reported in most cohort investigations. However, inverse relationships with folate and flavonoids in vegetables have been reported in cohort studies as well( Reference Larsson, Giovannucci and Wolk 47 , Reference Nothlings, Murphy and Wilkens 48 ). Some have reported positive associations with the consumption of meat and total sugar( Reference Nothlings, Murphy and Wilkens 49 , Reference Stolzenberg-Solomon, Cross and Silverman 50 ).
One of the strengths of the present study is the use of a validated( Reference Decarli, Franceschi and Ferraroni 34 ) and reproducible( Reference Franceschi, Negri and Salvini 35 , Reference Franceschi, Barbone and Negri 36 ) FFQ, which allowed for a comprehensive assessment of major nutrient sources in the Italian diet, although some measurement error inherent in the FFQ may be present. Moreover, we had detailed information on major recognised risk factors for pancreatic cancer. Notwithstanding the limitations of case–control studies in general, we believe that our findings on a positive association between the DII and the risk of pancreatic cancer are plausible and could be related to immune and hormonal factors( Reference Li, Albrecht and Li 2 – Reference Jackson and Evers 4 , Reference Moses, Maingay and Sangster 10 , Reference Talar-Wojnarowska, Gasiorowska and Smolarz 11 ). Also, the association is too strong to be accounted for by bias or confounding only, with an OR of 2·48 in the highest quintile.
In conclusion, the present study on the risk of pancreatic cancer and the DII in a Southern European population indicates a possible role of diet in the risk of developing pancreatic cancer, which we hypothesised would be through the process of inflammation. However, further confirmatory results from other studies conducted in different populations with different study designs (e.g. cohort studies) are required to establish this association.
Acknowledgements
The present study was funded by the Italian Foundation for Cancer Research (FIRC) and the IRCCS Centro di Riferimento Oncologico, Aviano (Ricerca Corrente 2009), Italy. J. R. H. was supported by an Established Investigator Award in Cancer Prevention and Control from the Cancer Training Branch of the National Cancer Institute (K05 CA136975).
The author's contributions are as follows: N. S. was involved in the calculation of the DII, performed all the analyses, and drafted the first version of the manuscript; C. B. and C. L. V. helped with the analyses, data acquisition interpretation of the data, and critical revision of the manuscript; A. Z. and D. S. contributed in data acquisition and to the interpretation of the data and drafting of the manuscript; J. R. H. provided expertise and oversight throughout the process. All authors revised and approved the final version of the manuscript.
The authors declare that there are no conflicts of interest.







