Alcohol has long been recognised as an aetiological agent in severe and enduring mental illness. One 19th-century physician-superintendent (Reference YellowleesYellowlees 1874) estimated that half of the male patients under his care at the Glamorgan County Asylum were there as a result of their heavy drinking. It is difficult, from the diagnostic perspective of today, to be confident about the proportion of cases labelled as ‘alcohol dementia’ or ‘alcohol mania’ that would now meet strict criteria for alcohol-related brain damage (ARBD).Footnote a Undoubtedly, individuals who would have met these criteria were being admitted to asylums before the description by Korsakoff, in 1881, of his eponymous syndrome, which was adopted by British psychiatry within a few years. It should be noted that the terms ‘dementia’ and ‘mania’ were then used in a technically different way from today.
A concern of psychiatrists in the late 19th century was that these disease entities could be inherited in a worsening pattern by the next generation, the so-called degeneration theory. Interestingly, links were made between inebriety and imbecility that are not made to the same extent today. One wonders whether, long before the description of the fetal alcohol syndrome in the 1970s, these early psychiatrists, with their concerns for physical appearance in relation to mental disability, were observing this phenomenon without realising it. Moving forwards in time, it is probably true that ARBD was a minor concern in the interwar years of the 20th century, coinciding with extremely low levels of alcohol consumption in an era of relative prohibition. In the past 65 years, as a consequence of rising alcohol consumption in many countries, ARBD has again become a major concern. Along with the increasing numbers of cases, the closure of medium- and long-stay psychiatric wards has made the issue of placement of this group of patients highly contentious. The most recent development, as outlined below, is that of specialist provision for them.
Illustrative case history
As a starting point for this article we present a typical case history, which is an amalgam of a large number of cases seen over a number of years. It is presented in the light of recent service developments in ARBD care in one city in the UK (Glasgow), which we feel has improved the management of this patient group.
Case vignette: A.B.
A.B., a 55-year-old single male, presented to the accident and emergency (A&E) department of a general hospital, having been found wandering in the street, unable to find his way home. Physical examination in A&E revealed an alcohol withdrawal syndrome, with evidence of stigmata of alcoholic liver disease. He was not overtly jaundiced and his liver was palpable on examination. He was admitted to a general medical ward for management of the alcohol withdrawal, and although no behavioural disturbance was noted, he required observation to prevent him leaving the ward. He received a full course of parenteral B vitamins, but was still confused 2 days after detoxification. A brain computed tomography (CT) scan revealed global atrophy, with no intracerebral pathology of note. His Mini-Mental State Examination (MMSE) score was 27/30, with loss of recall. He was referred to liaison psychiatry staff and a diagnosis of Korsakoff syndrome was made.
One evening A.B. was found wandering around the hospital, attempting to find his way home, and refused to return to the ward on a voluntary basis. He was detained under the Mental Health Act and admitted to a psychiatry ward under the care of a general adult psychiatrist. Six weeks after his initial presentation he continued to show markedly impaired recent memory, and a referral was made to the ARBD team for detailed neuropsychological and functional assessment. His full-scale IQ, using the Wechsler Adult Intelligence Scale – Revised (WAIS-R) (Reference WechslerWechsler 1981), was 106, but his memory quotient, using the Wechsler Memory Scale (WMS-R) (see Reference HarperHarper 1998) was 70. His premorbid IQ was estimated at 115. Tests of executive function showed clear deficits.
A.B. had no family, and occupational therapy assessment indicated that he would not be able to return to independent living. Referral was made to Social Services to access a suitable placement. He was discharged to ‘core and cluster’ supported accommodation, with input from the ARBD team, based within addiction services.
His care progressed from the ‘core’ 24-hour staffed facility to a nearby ‘cluster’ flat with less intensive support, but with help from the ‘core’ facility if required. He maintained abstinence from alcohol, and 1 year later was living in his own flat, with daily support from a voluntary organisation. His finances were controlled by guardianship order (legal measures).
Nosology/definition of ARBD
The difficulty in defining ARBD is exemplified by the definitions within current classification systems. ICD-10 and DSM-5 view it as a number of discrete entities, as opposed to a continuum (see section on neuropsychology on p. 309).
In ICD-10 the various symptoms of ARBD are classified under amnesic syndrome (F10.6), dementia (F10.73) or other persisting cognitive disorder (F10.74) (World Health Organization 1992). In DSM-5, they come under alcohol-induced persisting amnestic disorder (291.1) or alcohol-induced persisting dementia (291.2) (American Psychiatric Association 2013). It is worth noting that the DSM-5 categories ignore the possibility of slow recovery (over 1–2 years).
Addiction specialists use the term ARBD more widely to describe a spectrum of alcohol-related cognitive impairment, ranging from less severe impairment, through alcohol amnestic (or amnesic) syndrome (Wernicke–Korsakoff syndrome) to more widespread and severe impairment (dementia) (Reference Jacobson and LishmanJacobson 1987). Furthermore, clinical experience indicates that a significant proportion of patients present with a ‘mixed picture’ of aetiological factors, with multiple withdrawals, repeated head injuries, mild vascular insults and hepatic impairment, in addition to alcohol neurotoxicity and thiamine (vitamin B1) deficiency.
Aetiology and natural history of ARBD
Alcohol is thought to have a direct neurotoxic effect on the brain. Alcohol-dependent individuals have been shown to have decreased brain weight at post-mortem, predominantly related to reductions in white matter volume (Reference Harper and MatsumotoHarper 2005). A number of factors are likely to contribute to this, most importantly thiamine deficiency. Reference LishmanLishman (1986) postulated that the cumulative damage caused in ARBD was likely to be due to alcohol excess in addition to thiamine deficiency, the so-called Lishman hypothesis. Our clinical experience and evidence from neuropathology (Reference HarperHarper 1998) would support this view, with similar, although significantly larger, changes seen in the brains of individuals with Korsakoff syndrome compared with those of alcohol-dependent controls with no history of the syndrome.
Other factors that contribute to the effects of alcohol on cognitive impairment (and general functioning) are summarised in Box 1. As would be expected, a number of these are shared with predisposition to alcohol misuse/dependency.
BOX 1 Factors to consider in alcohol-related cognitive impairment
Demographic factors
-
• Age
-
• Gender
-
• Relationship status
-
• Social class
-
• Family history
-
• Ethnicity/genetics
Comorbid neurological and medical conditions
-
• Head injury
-
• History of withdrawal seizures
-
• Cerebrovascular disease (especially small vessel disease), underlying dementia of any cause
-
• Fetal alcohol syndrome
-
• Liver disease
-
• Malnutrition (including specific vitamin deficiencies)
Substance-misuse history
-
• Age at onset of dependent/binge drinking
-
• Duration of alcohol misuse/dependency
-
• Number of previous detoxifications
-
• Continuous v. binge drinking
-
• History of blackouts
-
• Periods of abstinence
-
• Other drugs with neurotoxic effects
-
• Type of alcohol consumed
Natural history of ARBD: the centrality of thiamine
As illustrated in the fictitious case vignette of A.B., and borrowing largely from the pioneering work of Victor, Adams & Collins (Reference Victor, Adams and CollinsVictor 1971), the course of ARBD is varied. Not all individuals who develop Korsakoff syndrome (alcohol amnestic syndrome, defined on p. 309) will have experienced a clear antecedent Wernicke episode (10% in their cohort did not). The classic triad of eye signs, ataxia and confusion in Wernicke’s encephalopathy is rarely seen and any one of these features should lead to a presumed diagnosis and treatment with parenteral thiamine (operational criteria for Wernicke’s encephalopathy are given in Box 2, p. 308). In our clinical experience, at least half of patients with ARBD will present with a number of medical and neurological comorbidities, which may exacerbate the impairments already present.
BOX 2 Patients at risk of thiamine deficiency and/or neurotoxicity
Factors predisposing to thiamine deficiency
-
• Weight loss in past year
-
• Reduced body mass index
-
• General clinical impression of nutritional status
-
• High carbohydrate intake
-
• Recurrent episodes of vomiting in the past month
-
• Co-occurrence of other nutritionally related conditions (polyneuropathy, amblyopia, pellagra, anaemia)
Factors predisposing to neurotoxicity
-
• Genetic predisposition to alcohol dependence and neurotoxic effects of alcohol
-
• Quantity/frequency of alcohol use
-
• Severity of dependence
-
• Frequent episodes of acute intoxication
-
• Withdrawal syndromes
-
• Concurrent drug use, particularly cocaine
-
• Alcohol-related liver damage
Early signs/symptoms of thiamine deficiency
-
• Loss of appetite
-
• Nausea/vomiting
-
• Fatigue, weakness, apathy
-
• Giddiness, diplopia
-
• Insomnia, anxiety, difficulty in concentration
-
• Loss of memory
Operational criteria for Wernicke encephalopathy: two of the following four
-
• Dietary deficiencies
-
• Oculomotor abnormalities
-
• Cerebellar dysfunction
-
• Either altered mental state or mild memory impairment
(Reference Thomson, Cook and GuerriniThomson 2008, with permission)
What is clear is that the most important factor altering the course of the condition is continued alcohol use. If a patient presents with ARBD, continued abstinence will facilitate either improvement or stability in their cognitive function. If further deterioration is seen, other causes should be sought. Elderly individuals ‘labelled’ as having ARBD may in fact be in the throes of an independent progressive dementing process, which will manifest in continued decline with abstinence.
Demographic factors
Age
Adolescent alcohol use, and alcohol misuse and ARBD in early adulthood
In recent years, studies investigating the effect of heavy alcohol consumption on the adolescent brain have shown evidence of white matter and subtle neurocognitive abnormalities (Reference Clark, Thatcher and TapertClark 2008). The heavy use of alcohol at this stage in development may affect structures involved in behavioural, emotional and cognitive regulation (e.g. the prefrontal cortex and limbic system), predisposing these individuals to further use. A recent systematic review of the literature on neuroimaging studies of the effect of alcohol on brain structure in adolescents and younger adults (under 40 years of age) (Reference Welch, Carson and LawrieWelch 2013) concluded that quantitative changes are found and that changes in the hippocampus in adolescent problem drinkers are of particular concern.
Ageing and alcohol use
In the neuropathological studies carried out by Reference HarperHarper (1998), the cerebral atrophy observed in the brains of chronic alcoholics typically resembled that seen in the brains of older adults. This gave rise to the premature ageing hypothesis and, later, the increased vulnerability hypothesis (Reference Oscar-Berman, Marinkovic, Kalechstein and van GorpOscar-Berman 2007).
The premature ageing hypothesis postulates that the brains of alcohol-dependent individuals are subject to accelerated ageing compared with the brains of normal controls. The increased vulnerability hypothesis proposes that the brains of older alcoholics are more susceptible to the toxic effects of alcohol than the brains of their younger counterparts. Most recent work would support the second hypothesis, but this has not translated into a model for understanding clinically significant ARBD in older adults (see below).
Contrary to popular opinion, there is a lack of conclusive evidence to suggest beneficial effects of small to moderate amounts of alcohol on cognition in older age. Studies suggesting this have relied on secondary outcome measures from larger studies and are open to major confounders: more recent evidence suggests no relationship (Reference Cooper, Bebbington and MeltzerCooper 2009).
Much has been written on the possible consequences of alcohol use by older adults, with some suggesting a ‘silent epidemic’ (Reference Gupta and WarnerGupta 2008). In spite of the hypotheses above, and increased rates of alcohol use in the general population, there has been no evidence of such an ‘epidemic’. Scottish data, for instance, show only a modest increase (Reference Jauhar and SmithJauhar 2009). To date, there is no neuropathological evidence to support a primary alcohol-related dementia of old age (probably reflecting the aetiological importance of thiamine deficiency). There continues to be a misconception that rates are high in elderly people, with a commonly quoted figure of 21% (Reference Smith and AtkinsonSmith 1995). More recent work, incorporating diagnoses of frontotemporal dementia and more robust clinical measures (Reference Oslin and CaryOslin 2003), found evidence of ‘alcohol-related dementia’ in only 16 patients (10.1%) from a nursing home population.
Gender
As with alcohol-related liver disease, women appear to be more susceptible to the effects of alcohol on the central nervous system, although the precise extent of this is not clear. Evidence suggests that, on average, women develop ARBD 10 years younger than men, after a shorter drinking career (Reference Victor, Adams and CollinsVictor 1971). In older women who start drinking in later life, a partially reversible form of ARBD appears to be more common (Reference CuttingCutting 1978).
Genetics
It was once postulated that Wernicke–Korsakoff syndrome might be the result of a genetic variant of the thiamine-dependent enzyme transketolase (crucial for glucose metabolism in the areas of the brain affected by Wernicke–Korsakoff pathology). This was presumed to be an autosomal recessive condition that would only manifest under conditions of thiamine deficiency. However, with the advent of molecular genetic techniques it has been shown that transketolase in humans is non-variant.
More recently, other candidate genes that may predispose to ARBD have been suggested. These fall into three broad categories: genes for alcohol-metabolising enzymes; genes for proteins involved in neurotransmitter systems (e.g. gamma-aminobutyric acid, GABA); and genes for proteins involved in the transport of thiamine (Reference Guerrini, Thomson and GurlingGuerrini 2009).
The potential benefits of further work in this area include the possibility of identifying those at higher risk of developing thiamine deficiency, and therefore at higher risk of developing clinically significant ARBD.
Comorbid physical conditions
One of the most clinically relevant comorbid physical problems is the relationship between alcoholic liver disease and ARBD. Alcohol-misusing patients with comorbid cirrhosis have been shown to have more brain damage as measured by cerebral atrophy than those without cirrhosis on post-mortem examination (Reference Harper and KrilHarper 1985).
Cerebral atrophy is measured by subtracting brain volume from intracranial volume to derive pericerebral space, which will be increased with cerebral tissue loss. In their study of patients with alcohol dependency, Harper & Matsumoto (2009) found that those with associated liver disease had greater pericerebral space than those with Wernicke–Korsakoff syndrome, who themselves had higher pericerebral space than those with neither Wernicke–Korsakoff syndrome nor associated liver disease. This would suggest an additive effect of alcoholic liver disease on neural tissue damage, distinct from hepatic encephalopathy alone. This comorbidity has become much more clinically relevant to the problem of ARBD in the past 20 years, given the marked increase in alcohol cirrhosis in the UK.
Furthermore, anyone involved with alcohol-dependent patients will acknowledge the high rate of minor (and major) head injuries, cerebrovascular events and increased risk of haemorrhagic stroke, and such events should be checked for in their medical history if they present with cognitive impairment.
Fetal alcohol spectrum disorder/’minimal’ brain dysfunction
The longitudinal course of fetal alcohol syndrome/alcohol-related neurodevelopmental disorder has not been well defined, and its prevalence in the ARBD population is unclear. There is an older literature on minimal brain dysfunction predisposing to alcohol dependence, implying that care is needed in establishing whether neuropsychological deficits are primarily due to alcohol-related damage. Ideally, neuropsychological assessment should therefore try to ascertain premorbid functioning and educational attainment, enabling comparison with current function.
Substance misuse history
Binge v. continuous drinking
There is evidence of short-term cognitive impairment in the hours and days following a one-off heavy drinking session beyond the period of intoxication. This builds on animal work that shows decreased rates of hippocampal neurogenesis in rats exposed to binge levels of ethanol (Reference Nixon and CrewsNixon 2002). Longer-term effects on memory are still to be clearly defined in binge drinkers compared with more continuous drinkers, and extrapolation from rat studies (e.g. Reference George, Sanders and FreilingGeorge 2012) to humans has to be treated with caution. Whether in a binge or continuous fashion, the overall weekly amount of alcohol consumed beyond 21 UK units of alcohol per week for men and 14 units for women has been shown to have a positive relationship with the likelihood of cognitive impairment (Reference HeffernanHeffernan 2008).
Future research is also needed to identify more precise markers of recovery, and the clinical correlates of any ongoing damage, in those who are abstinent and those who continue to drink, taking into account differing patterns of use.
‘Blackouts’
The presence of alcohol blackouts has also attracted significant interest. Blackouts are defined as episodes of amnesia, during which individuals are capable of participating even in salient, emotionally charged events – as well as more mundane events – that they later cannot remember (Reference GoodwinGoodwin 1995).
Recent studies imply increased vulnerability to the effects of alcohol on memory in those with a history of blackouts, although a direct relationship has yet to be established (Reference WhiteWhite 2003). Blackouts are most likely a proxy for severity of the alcohol use disorder and may imply higher overall consumption levels.
It should also be borne in mind that other substances (e. g. benzodiazepines) have an additive effect on memory (and psychosocial functioning) in those who are acutely intoxicated with alcohol.
Type of preferred alcoholic drink
Those who prefer spirits and wine as opposed to beer appear to have more pronounced ARBD, although the precise mechanisms for this are unclear. A recent imaging study showed that increased levels of plasma homocysteine were correlated with brain atrophy in alcoholics who preferred spirits and wine, compared with those preferring beer (even though the beer drinkers had higher total alcohol use) (Reference Wilhelm, Frieling and HillemacherWilhelm 2008). The authors postulated that levels of circulating homocysteine may be an important intermediary in producing excitotoxic neuronal damage in those who drink wine and spirits.
Clinical evaluation and re-evaluation
The clinical assessment of patients presenting with suspected or established ARBD is at times complex, and requires assiduous history-taking and examination. Often a corroborative history (from family or care home/hospital staff) is invaluable in teasing out the temporal nature of deficits and functional decline, in addition to the alcohol and substance misuse history.
Physical examination is helpful in assessing the degree of vitamin deficiency and the presence of peripheral neuropathy. The latter may indicate a sensitivity to central nervous system involvement; in Victor et al’s original sample, 82% of those with Wernicke–Korsakoff syndrome had clinical evidence of polyneuropathy (Reference Victor, Adams and CollinsVictor 1971). Symptoms and signs of thiamine deficiency are shown in Box 2.
The assessment of cognitive function can be complicated, since many patients will only recently have stopped drinking and may be suffering from the sequelae of alcohol withdrawal and its treatment. Although some advocate 6 weeks of abstinence before assessment, there does not appear to be significant evidence to support this (Reference Brandt, Butters and RyanBrandt 1983). DSM-5 reflects this view, stating that alcohol withdrawal does not usually last longer than 4 weeks. In our experience, assessment after 4 weeks using an instrument such as the Addenbrooke’s Cognitive Examination (ACE) (Reference Mioshi, Dawson and MitchellMioshi 2006) in addition to a test of executive function such as the Frontal Assessment Battery (FAB) (Reference Dubois, Slachevsky and LitvanDubois 2000) should provide sufficient information on which to base an initial working diagnosis of ARBD.
Other instruments can be recommended, such as the Behavioural Assessment of the Dysexecutive Syndrome (BADS), although one instrument may not be enough to show impairment in all affected individuals (Reference Ihara, Berrios and LondonIhara 2000).
Psychiatric comorbidity
Anecdotally, rates of depression and anxiety in individuals with ARBD are elevated and can aggravate the functional deficit. It is worth assessing for and treating these conditions following a period of abstinence. From clinical experience, occasional psychotic phenomena (and at times alcohol hallucinosis) are seen in a small number of these patients, though there is a dearth of research on this association. Treatment of comorbid psychiatric conditions follows guidance similar to that for other neuropsychiatric conditions.
Multidisciplinary assessment
In our experience, a multidisciplinary team approach for individuals with ARBD is vital. Other disciplines involved include occupational therapy (functional and home assessment), dietetics (psychoeducation, given the predominance of thiamine deficiency), physiotherapy (given the rates of peripheral neuropathy and ataxia) and clinical psychology (both for therapeutic interventions and neuropsychological assessment).
Care needs to be taken in re-evaluating these patients over time, as slow adaptation to an environment may occur without any apparent improvement on more objective psychometric measures. We know of patients in whom recovery had been assumed and a more independent placement had been arranged, with the patient then failing to cope in the new environment.
Neuropsychology
There still exists a degree of controversy as to whether ARBD exists on a continuum, with cognitive deficits found in alcohol-dependent individuals without a definite ARBD diagnosis. Ryback’s continuity hypothesis states that such deficits do exist on a continuum, and increase with years of heavy drinking (Reference RybackRyback 1971). More recent work (Reference Pitel, Beaunieux and WitkowskiPitel 2008) supports the hypothesis and suggests that there exists a continuum of severity in both episodic memory and working memory deficits in Wernicke–Korsakoff syndrome and alcohol-dependent individuals without Wernicke–Korsakoff syndrome.
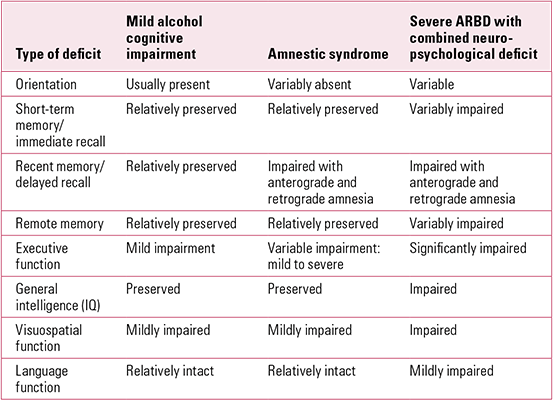
The major deficits associated with mild alcohol-related cognitive impairment, alcohol amnestic syndrome/Korsakoff syndrome (ARBD) and severe ARBD with combined cognitive impairments are summarised in Table 1.
TabLE 1 The spectrum of neuropsychological deficits seen in alcohol-related brain damage (ARBD)
Amnestic syndrome
We can define amnestic syndrome operationally as a syndrome associated with chronic prominent loss of recent memory and preservation of immediate recall; remote memory is sometimes also impaired. Disturbances of sense of time and ordering of events are usually evident, as are difficulties in learning new material. Confabulation may be marked, but is not invariably present. Other cognitive functions are usually well preserved and amnesic defects are out of proportion to other disturbances.
However, as Table 1 underlines, it is unusual to find this syndrome without at least some dysexecutive features in those with an alcohol-related aetiology. In-depth neuropsychological assessment is always helpful in people with ARBD, and especially where the clinical picture is unclear and other comorbidities are questioned.
Neuroimaging
There is currently no diagnostic test for ARBD, and until recently the majority of neuroimaging findings did not satisfactorily correlate with neuropsychological function. In the context of more modern imaging, and in-depth neuropsychology, correlations have been found, specifically linking executive function to magnetic resonance imaging (MRI) findings in detoxified alcoholics, implicating the frontocerebellar circuit in executive and working memory deficits (Reference Chanraud, Martelli and DelainChanraud 2007). It has also been proposed that damage to the Papez circuit (which includes the hippocampus) may affect episodic memory in this population.
Newer MRI modalities (e.g. diffusion tensor imaging) have been employed in research studies looking at specific white matter tracts (which, given the neuropathology associated with ARBD, would seem reasonable). Although the literature is more sparse than in other psychiatric conditions (e.g. schizophrenia), findings are promising, with clear abnormalities noted in the tracts linking the prefrontal cortex to the limbic system.
For more in-depth information relating to brain imaging and alcohol, the interested reader is directed to a relatively recent systematic review that summarises the effects of alcohol on the brain through a number of imaging modalities (Reference Bühler and MannBühler 2011).
Newer techniques may lead to a way of defining the specific pathology in individual cases, given the localised nature of the damage in ARBD.
Clinical management
Clinically, we would advocate that every patient presenting with suspected ARBD should have a brain scan to exclude other pathologies that are not uncommon in this population (e.g. subdural haemorrhage, vascular insults). Computed tomography may be used, but MRI is preferable given the better resolution offered, specifically of subcortical structures, and the lack of exposure to ionising radiation. MRI holds the prospect of defining positive evidence for ARBD, particularly where structures such as the mamillary bodies and mamillothalamic tracts are affected, so its value is not restricted solely to excluding other pathology. The extra expense of neuroimaging is justifiable given the cost of incorrect diagnosis, which for some can lead to expensive and unnecessary long-term institutional care without any attempt at rehabilitation to less costly semi-independent or independent living.
Services
Patients with ARBD have often been a source of dispute in terms of service provision (Reference WilsonWilson 2011). Over the past decade, however, there have been attempts in some parts of the UK to implement specific care pathways for this group. National guidelines and recommendations have been published in Scotland (Reference Cox, Anderson and McCabeCox 2004; Scottish Executive 2007), and Glasgow saw an ARBD team established in 2005. A specialist unit and service have also been established in Cheshire and Wirral, in north-west England (Reference Wilson, Halsey and MacphersonWilson 2012). For articles on these services and care pathways, and on others in the UK, particularly Northern Ireland, see Neuropsychiatry News (Section of Neuropsychiatry, Royal College of Psychiatrists 2013).
Of particular interest is the recommendation of the Scottish Executive (2007) that provision for this patient group should be within a general neuropsychiatric/brain injury rehabilitation service. In reality, where such a service does exist, the ARBD group tends to be excluded. This is illustrated by a more recently published report in Scotland that suggests a managed care network for ARBD, equivalent to that intended for patients with acquired brain injury (Mental Welfare Commission for Scotland 2010).
An alternative model is to have a designated community team, with residential provision, including input from the voluntary sector where relevant. This is the model that currently exists in Glasgow. A specialist team comprising a psychiatrist, clinical psychologist, occupational therapist, psychiatric nurses and social care staff advises on management after detailed initial assessment, and continues to track patients and advise on care for at least 2 years after confirmed diagnosis. The aim of this team is also to provide behavioural intervention to maximise each patient’s functioning.
The experience in Cheshire and Wirral and in Glasgow suggests that the adaptation or development of local supported accommodation and residential care is preferable to placing individuals in the private sector (often at greater expense and at a distance from relatives and friends).
Legislation
The use of legislation (in Scotland, the Mental Health (Care and Treatment) (Scotland) Act 2003 and the Adults with Incapacity (Scotland) Act 2000; in England and Wales, the Mental Health Act 1983 and the Mental Capacity Act 2005) is another area that can arouse debate. For example, should such measures be applied to those with a pure amnesic deficit when they can clearly and consistently come to a decision on their wishes in a specific situation, but are later unable to recall the discussion and ensuing decision-making? The application of such measures is more clear-cut where other cognitive deficits are obviously present. In our clinical experience, a careful functional assessment of ability in relation to specific tasks is essential. All patients with alcohol dependence, particularly older patients, require consideration as to whether they have ARBD, as the consequences of ignoring this can leave clinicians open to censure (see Mental Welfare Commission for Scotland 2006).
The working of the Adult Support and Protection (Scotland) Act 2007 is also starting to highlight the inadequacy of provision for this patient group.
Treatment and prevention in those at risk of ARBD
Although the precise links between alcohol, thiamine deficiency and brain damage are complex, there is little doubt that, at a population level, the rates of ARBD are higher in areas where alcohol consumption and social deprivation are highest. Adding thiamine to alcoholic drinks (beer) has been considered in the UK, USA and Australia, but there is little evidence to suggest a change in prevalence with thiamine supplementation. Such an intervention is beset by other problems, such as comorbid vitamin deficiencies and the type of alcohol preferred (see earlier).
To date (and probably as a reflection of its multifactorial presentation) there is no reliable screening measure for ARBD, although screening measures do exist for thiamine deficiency (Reference Sgouros, Baines and BloorSgourous 2004).
Thiamine and magnesium supplementation
It is important to identify patients with alcohol dependence, given the evidence supporting treatment with parenteral thiamine and managed detoxification for prevention of brain damage. This has been reflected in guidelines issued by the Royal College of Physicians (Reference Thomson, Cook and TouquetThomson 2002), and links well to work in Australia (Reference Ambrose, Bowden and WhelanAmbrose 2001) that showed beneficial effects of thiamine supplementation on working memory.
The optimal use of this treatment has yet to be agreed on, a Cochrane review pointing out the lack of definitive randomised controlled trial evidence on dosing (Reference Day, Bentham and CallaghanDay 2013). What has been made clear is that, in a significant proportion of patients with alcohol dependence (who are malnourished), absorption of oral thiamine is exceptionally poor and has been estimated at one-third of that in healthy controls (Reference Thomson, Cook and GuerriniThomson 2008). Features that should precipitate prophylactic treatment with parenteral thiamine are outlined in Box 2.
What should be stressed is that parenteral thiamine can be continued past the recommended 5-day period of use (500 mg three times daily) if clinical improvement is seen. Furthermore, concerns as to a risk of anaphylaxis with parenteral use are overestimated and should not be a major barrier to providing treatment in the community.
There is also a suggestion that magnesium supplementation be given, as magnesium is required for utilisation of thiamine and is deficient in a large number of alcohol-dependent individuals.
Neuroprotective agents during detoxification
The use of neuroprotective agents during detoxification has yet to be firmly established despite tantalising evidence for drugs such as acamprosate (Reference De Witte, Littleton and ParotDe Witte 2005).
Treatment and rehabilitation in established ARBD
A number of principles from the generic neurorehabilitation field should be incorporated into the design of ARBD units and the care plans of those with ARBD.
The environment
In addition to basic features (simplicity of design and clear signposting), care should be taken to ensure that residents have their own rooms, which they can personalise with items that help autobiographical memory and with aids for prospective memory. These would include a personal wall chart, with a timetable for the week ahead.
Memory aids
As with other forms of memory disorder, there is no reason to deprive this patient group of access to the latest memory aids. These range from the simple – for example, a diary and a whiteboard – to the more complex electronic technology. There is some evidence to suggest the use of radio-paging or mobile phone technology as a memory prompt in acquired brain injury (Reference Wilson, Emslie and QuirkWilson 2001), and it would sound reasonable for this to be considered for those with alcohol amnestic syndrome/ARBD. For further information see the Headway factsheet Coping with Memory Problems: Practical Strategies (www.headway.org.uk/Factsheets.aspx).
Errorless v. errorful/trial-and-error learning
As proposed in a review of Korsakoff syndrome in Advances (Reference Smith and HillmanSmith 1999) and confirmed by recent evidence, though with some exceptions (Reference Kessels, van Loon and WesterKessels 2007), the body of work on rehabilitation of those with acquired brain injury suggests that errorless learning has a significant role to play for both verbal and non-verbal tasks.
Put simply, errorless learning consists of directing the individual to a desired response when it is clear they do not have a ready answer to a practical task/question. Errorful or trial-and-error learning is just what its name implies: the individual reaches the desired response by trial and error. Clinical experience has suggested that, although care home staff are aware of the benefits of errorless learning, it is debatable whether it is consistently used.
Distributive practice
Distributive practice is another important technique in memory enhancement, in relation to a new environment or task. It involves presenting information in small component parts and repeating it on a number of occasions until it is retained. It is important to present the information in manageable ‘chunks’.
Rehabilitation for dysexecutive symptoms
Patients with ARBD almost invariably have some frontal lobe damage and, depending on the specific areas of the frontal lobe involved, a number of symptom clusters can result. Careful assessment of the pattern of dysexecutive symptoms will guide neurorehabilitation focused on these deficits. Techniques exist in the brain injury literature, such as goal management training, which can be combined with mobile phone text alerts as part of an individualised programme of rehabilitation. See Royal College of Psychiatrists (2014) for further information on this (pp. 33–41) and a range of other topics covered in this article.
Pharmacological intervention
There is still no established drug treatment for the cognitive deficits of ARBD. However, where psychiatric comorbidity is suspected, and in particular where depression is suspected on the basis of behavioural change such as self-neglect and loss of interest in previously enjoyed activity, a trial of an antidepressant is indicated. Selective serotonin reuptake inhibitors should be favoured over antidepressants with sedative and anticholinergic properties.
Conclusions
The effects of alcohol on cognitive function have never been more relevant to psychiatry in the UK, given the current prevalence of excess alcohol use. There are clear diagnostic and management approaches for people with ARBD that all general and old age psychiatrists should be aware of, and it is hoped that by utilising these, better care can be given to this patient group. We hope that this article, in conjunction with the recently published Royal College of Psychiatrists’ report on this topic (Royal College of Psychiatrists 2014), will help to further educate colleagues on the management of this neglected condition and will advance the cause of improving service provision for the individuals affected by ARBD.
MCQs
Select the single best option for each question stem
-
1 The most common cause of Wernicke–Korsakoff syndrome is deficiency of:
-
a vitamin C
-
b vitamin B1
-
c magnesium
-
d vitamin B12
-
e vitamin B6.
-
-
2 The rate of ‘alcohol-related dementia’ in the elderly population is estimated at around:
-
a 30%
-
b 40%
-
c 50%
-
d 5%
-
e 10%.
-
-
3 In all forms of alcohol-related brain damage, neuropsychological deficits are seen in the domain of:
-
a executive function
-
b language function
-
c IQ
-
d orientation
-
e semantic memory.
-
-
4 The proportion of people with Korsakoff syndrome who have evidence of peripheral neuropathy is about:
-
a 20%
-
b 40%
-
c 10%
-
d 50%
-
e 80%.
-
-
5 The degree of alcohol-related brain damage is increased by comorbid disease of the:
-
a liver
-
b pancreas
-
c heart
-
d spleen
-
e kidney.
-
MCQ answers
| 1 | b | 2 | e | 3 | a | 4 | e | 5 | a |
Acknowledgements
We would like to acknowledge the advice of Jon Evans, Professor of Clinical Neuropsychology, Glasgow University, concerning memory rehabilitation, and thank Dr Allan Thomson for permission to reproduce Box 2.




eLetters
No eLetters have been published for this article.