Introduction
Major depressive disorder (MDD) (lifetime prevalence 13–16%) is a complex constellation of disturbances in cognition, behaviour and physical functioning (Rizvi & Kennedy, Reference Rizvi and Kennedy2012). MDD is the third leading cause of global disease burden and a leading cause of disability worldwide (Mathers et al. Reference Mathers, Fat and Boerma2008). Depression is clinically and aetiologically heterogeneous (Hasler, Reference Hasler2010). A major research effort aims to elucidate its biological underpinnings, and thus to develop more effective treatments.
Numerous structural magnetic resonance imaging (MRI) studies have sought to identify the key brain areas involved in the pathogenesis of depressive symptoms. Voxel-based morphometry (VBM) is a fully automated method for analysing neuromorphological MRI data which allows unbiased investigation of differences in brain structure between patients and healthy subjects (Ashburner & Friston, Reference Ashburner and Friston2001). It has accuracy comparable with manual volumetry (Davies et al. Reference Davies, Scahill, Graham, Williams, Graham and Hodges2009) and overcomes the technical limitations of region-of-interest approaches. VBM has been widely used in characterizing subtle changes in brain structure in a variety of neurological and psychiatric diseases. VBM studies have reported that MDD is associated with widespread local abnormalities in many brain regions, such as the frontal gyrus (Wagner et al. Reference Wagner, Koch, Schachtzabel, Reichenbach, Sauer and Schlösser2008; Scheuerecker et al. Reference Scheuerecker, Meisenzahl, Koutsouleris, Roesner, Schopf, Linn, Wiesmann, Bruckmann, Moller and Frodl2010; Salvadore et al. Reference Salvadore, Nugent, Lemaitre, Luckenbaugh, Tinsley, Cannon, Neumeister, Zarate and Drevets2011), anterior cingulate gyrus (Tang et al. Reference Tang, Wang, Xie, Liu, Li, Su, Liu, Hu, He and Blumberg2007; Wagner et al. Reference Wagner, Koch, Schachtzabel, Reichenbach, Sauer and Schlösser2008; Salvadore et al. Reference Salvadore, Nugent, Lemaitre, Luckenbaugh, Tinsley, Cannon, Neumeister, Zarate and Drevets2011) and hippocampus (Wagner et al. Reference Wagner, Koch, Schachtzabel, Reichenbach, Sauer and Schlösser2008; Zou et al. Reference Zou, Deng, Li, Zhang, Jiang, Huang and Sun2010; Arnone et al. Reference Arnone, McKie, Elliott, Juhasz, Thomas, Downey, Williams, Deakin and Anderson2012). However, these reports are inconsistent in detail, probably because of small sample sizes, heterogeneous subjects and methodological differences. Identifying consistent results from VBM studies of MDD patients through meta-analysis is therefore of particular importance.
New analytical tools have made possible whole-brain meta-analyses using the spatial coordinates of reported structural imaging findings to produce consensus neuroanatomical maps of abnormalities. Three recent meta-analyses of VBM studies found that MDD was characterized by volume reductions in the bilateral anterior cingulate cortex (ACC) (Bora et al. Reference Bora, Fornito, Pantelis and Yucel2012; Du et al. Reference Du, Wu, Yue, Li, Liao, Kuang, Huang, Chan, Mechelli and Gong2012; Lai, Reference Lai2013), right middle and inferior frontal gyrus (Du et al. Reference Du, Wu, Yue, Li, Liao, Kuang, Huang, Chan, Mechelli and Gong2012), parahippocampal gyrus and hippocampus (Du et al. Reference Du, Wu, Yue, Li, Liao, Kuang, Huang, Chan, Mechelli and Gong2012). However, most of the patients included in these were receiving antidepressant drugs at the time of scanning. It is recognized that the inclusion of medicated patients may bias meta-analyses and limit the interpretability and generalizability of the results.
Animal and human studies yield primary evidence on the effect of therapy on grey matter. For example, 3 weeks of agomelatine administration affects neurogenesis in the dorsal and ventral hippocampus of adult rats (Banasr et al. Reference Banasr, Soumier, Hery, Mocaer and Daszuta2006); similarly, 4 weeks of tianeptin administration protects against the decreased hippocampal volume produced by stress in tree shrews (Czeh et al. Reference Czeh, Michaelis, Watanabe, Frahm, de Biurrun, van Kampen, Bartolomucci and Fuchs2001). Medication-related differences in regional brain volumes have been reported in patients with mood disorders (Brambilla et al. Reference Brambilla, Nicoletti, Harenski, Sassi, Mallinger, Frank, Kupfer, Keshavan and Soares2002; Chang et al. Reference Chang, Karchemskiy, Barnea-Goraly, Garrett, Simeonova and Reiss2005; Lavretsky et al. Reference Lavretsky, Roybal, Ballmaier, Toga and Kumar2005; Savitz et al. Reference Savitz, Nugent, Bogers, Liu, Sills, Luckenbaugh, Bain, Price, Zarate, Manji, Cannon, Marrett, Charney and Drevets2010; Smith et al. Reference Smith, Chen, Baxter, Fort and Lane2013). Adult patients with MDD show increased grey matter in the left dorsolateral prefrontal cortex (DLPFC) after 12 weeks of selective serotonin reuptake inhibitor (SSRI) treatment (Smith et al. Reference Smith, Chen, Baxter, Fort and Lane2013), while in geriatric MDD patients antidepressant exposure is associated with increased orbitofrontal grey matter (Lavretsky et al. Reference Lavretsky, Roybal, Ballmaier, Toga and Kumar2005). In addition, a meta-analysis has reported that the greater the proportion of medicated individuals in the depressed sample, the larger is the abnormality in amygdala volume (Hamilton et al. Reference Hamilton, Siemer and Gotlib2008). Furthermore, there is growing evidence that decreased levels of brain-derived neurotrophic factor and other neurotrophic factors in MDD could contribute to the atrophy of limbic structures such as the hippocampus and prefrontal cortex, and that the neurotrophic actions of antidepressants could reverse neuronal atrophy and cell loss (Duman & Monteggia, Reference Duman and Monteggia2006). It is therefore conceivable that antidepressant medication itself could cause regional grey matter volume changes in MDD. Thus studies of unmedicated patients could better elucidate brain abnormalities that are directly related to the disease itself.
Direct study of unmedicated depressed patients faces substantial practical and ethical problems (Blease, Reference Blease2011). Although published VBM studies on medication-free MDD patients have recently become sufficiently numerous to warrant specific meta-analysis, no such meta-analysis of grey matter volumes has yet been published. Therefore, the goals of this study were twofold: first, to establish the most prominent and replicable grey matter abnormalities in medication-free MDD patients; and, second, to examine the effects of demographics and clinical characteristics on grey matter volumes in MDD, using a new coordinate-based meta-analytic technique called effect-size signed differential mapping (ES-SDM). ES-SDM combines several useful features of earlier methods, such as activation likelihood estimation and multilevel kernel density analysis. It also introduces some novel improvements: to avoid any voxel appearing significant in opposite directions, both positive and negative differences are reconstructed in the same signed differential map; the use of effect sizes allows combination of reported peak coordinates with statistical parametric maps, thus allowing more detailed and accurate meta-analyses; lastly, complementary analyses such as jack-knife, subgroup and meta-regression analyses are readily used to assess the robustness and heterogeneity of the results (Radua et al. Reference Radua, Mataix-Cols, Phillips, El-Hage, Kronhaus, Cardoner and Surguladze2012b ).
Method
Inclusion of studies
A systematic strategy was used to search for relevant studies published in Pubmed, Embase, Web of Science and Science Direct up to March 2013, using the keywords ‘depression’ or ‘depressive disorder’ or ‘unipolar depression’ plus ‘voxel-based morphometry’ or ‘VBM’ or ‘voxel*’ or ‘morphometry’. The reference lists of these studies were checked to identify further studies for inclusion. Studies were included according to the following criteria: (1) used VBM to analyse whole-brain grey matter changes in MDD patients; (2) compared MDD patients with healthy control subjects; and (3) investigated medication-free MDD patients, who either had never received antidepressive medications (i.e. were medication-naive), or who underwent a wash-out period before MRI scanning. Findings based on small volume correction were excluded. Theoretical papers and reviews were excluded, as well as studies that reported adolescent or late-life depression. For studies where multiple independent patient samples were compared with healthy controls, the appropriate coordinates were included as separate datasets. For studies using overlapping samples, the study with the most subjects was included.
Recorded variables
For each included study we recorded: numbers, sex and mean age of subjects; illness duration, depression symptom severity and mean numbers of episodes; drug status; the percentage of patients who had ever received psychotherapy; the statistical threshold of the main findings, and the method employed to correct whole-brain results for multiple comparisons.
Meta-analysis of regional differences in grey matter
Regional differences in grey matter were analysed using ES-SDM (http://www.sdmproject.com), a voxel-based meta-analytic approach that uses the reported peak coordinates to re-create maps of the effect size of group differences in grey matter. For peak coordinates, the re-creation is based on first converting the peak t value to Hedges' effect size and then applying a non-normalized Gaussian kernel to the voxels close to the peak. In ES-SDM both positive and negative differences are reconstructed in the same map, which prevents a particular voxel appearing to be significant in opposite directions. Importantly, negative studies are also included in the meta-analysis. Jack-knife sensitivity analysis was used to test the replicability of results and meta-regression methods to characterize the impact of key clinical variables on brain structure (Radua et al. Reference Radua, Mataix-Cols, Phillips, El-Hage, Kronhaus, Cardoner and Surguladze2012b ).
ES-SDM has been described in detail elsewhere (Radua et al. Reference Radua, Via, Catani and Mataix-Cols2011, Reference Radua, Borgwardt, Crescini, Mataix-Cols, Meyer-Lindenberg, McGuire and Fusar-Poli2012a , Reference Radua, Mataix-Cols, Phillips, El-Hage, Kronhaus, Cardoner and Surguladze b ) and applied to meta-analysis studies on neuropsychiatric disorders including schizophrenia and bipolar disorder (Bora et al. Reference Bora, Fornito, Radua, Walterfang, Seal, Wood, Yucel, Velakoulis and Pantelis2011; Palaniyappan et al. Reference Palaniyappan, Balain, Radua and Liddle2012). Here we summarize its main features. First, we ensured that the same threshold was used throughout the whole brain in each included study (although not necessarily the same in different studies), in order to avoid biases towards liberally thresholded brain regions (Radua & Mataix-Cols, Reference Radua and Mataix-Cols2009). Second, an effect-size signed map of the differences in grey matter was separately recreated for each study. Third, the mean map was obtained by calculating the mean of the study maps, weighted by the inverse of each study variance and accounting for inter-study heterogeneity. Statistical significance was assessed by a permutation test (Radua et al. Reference Radua, Mataix-Cols, Phillips, El-Hage, Kronhaus, Cardoner and Surguladze2012b ).
Sensitivity analysis
A systematic whole-brain voxel-based jack-knife sensitivity analysis was used to test the replicability of the results. This involves repeating the main analysis 14 times, discarding a different study each time: if a brain region remains significant in all or most of the combinations of studies it can be concluded that this finding is highly replicable.
Subgroup analysis
We conducted a subgroup analysis of first-episode medication-naive MDD patients. Strategically, recruiting such individuals helps to reduce confounders such as illness duration and previous antidepressant treatment that may dilute the neuropathological findings in MDD. Because medication withdrawal will not immediately reverse any medication-related grey matter changes, we compared these results with a subgroup analysis of MDD patients who underwent a wash-out period before MRI scanning.
Meta-regression analysis
The following variables were explored by meta-regression: the percentage of female MDD patients (i.e. in the whole MDD group in each study); mean age of patients; illness duration; and the mean number of illness episodes. Depression symptom severity could not be explored by meta-regression due to a lack of consistent depression-scale data. The percentage of MDD patients who had ever received psychotherapy could not be explored by meta-regression because data were available for fewer than nine studies (Radua & Mataix-Cols, Reference Radua and Mataix-Cols2009). As described in previous meta-analyses, in order to minimize the detection of spurious relationships, we decreased the probability threshold to 0.0005 required abnormalities to be detected both in the slope and in one of the extremes of the regressor, and discarded findings in regions other than those detected in the main analyses (Radua & Mataix-Cols, Reference Radua and Mataix-Cols2009). Furthermore, regression plots were visually inspected to discard fittings driven by too few studies (Radua & Mataix-Cols, Reference Radua and Mataix-Cols2009).
Results
Included studies and sample characteristics
The search strategy identified 1144 studies, of which 11 studies (Tang et al. Reference Tang, Wang, Xie, Liu, Li, Su, Liu, Hu, He and Blumberg2007; Wagner et al. Reference Wagner, Koch, Schachtzabel, Reichenbach, Sauer and Schlösser2008; Cheng et al. Reference Cheng, Xu, Chai, Li, Luo, Yang, Li, Shan, Xu and Xu2010; Jia et al. Reference Jia, Huang, Wu, Zhang, Lui, Zhang, Amatya, Kuang, Chan, Kemp, Mechelli and Gong2010; Lai et al. Reference Lai, Hsu and Wu2010; Scheuerecker et al. Reference Scheuerecker, Meisenzahl, Koutsouleris, Roesner, Schopf, Linn, Wiesmann, Bruckmann, Moller and Frodl2010; Zou et al. Reference Zou, Deng, Li, Zhang, Jiang, Huang and Sun2010; Salvadore et al. Reference Salvadore, Nugent, Lemaitre, Luckenbaugh, Tinsley, Cannon, Neumeister, Zarate and Drevets2011; Arnone et al. Reference Arnone, McKie, Elliott, Juhasz, Thomas, Downey, Williams, Deakin and Anderson2012; Ma et al. Reference Ma, Ding, Li, Guo, Long, Liu, Gao, Zeng, Zhao and Chen2012; Zhang et al. Reference Zhang, Yao, Zhu, Wang and Zhong2012), including 14 datasets, ultimately met inclusion criteria, with 400 unmedicated MDD patients (142 male and 258 female; mean age 33.8 years) and 424 healthy controls (163 male and 261 female; mean age 32.4 years). Of two studies using overlapping samples, the one with the most subjects was included after email discussion with the authors (Arnone et al. Reference Arnone, McKie, Elliott, Juhasz, Thomas, Downey, Williams, Deakin and Anderson2012). Fig. 1 shows a flow diagram of the identification and attrition of studies. Table 1 summarizes clinical and demographic data from all included studies. In no study was there any significant difference in age and sex between the MDD group and the healthy controls.

Fig. 1. Identification and attrition of studies. Search terms were ((depression OR depressive disorder OR unipolar depression) AND (voxel-based morphometry OR VBM OR voxel* OR morphometry)). MDD, Major depressive disorder; VBM, voxel-based morphometry; HC, healthy controls.
Table 1. Demographic and clinical characteristics of subjects in the 14 voxel-based morphometry datasets included in the meta-analysis
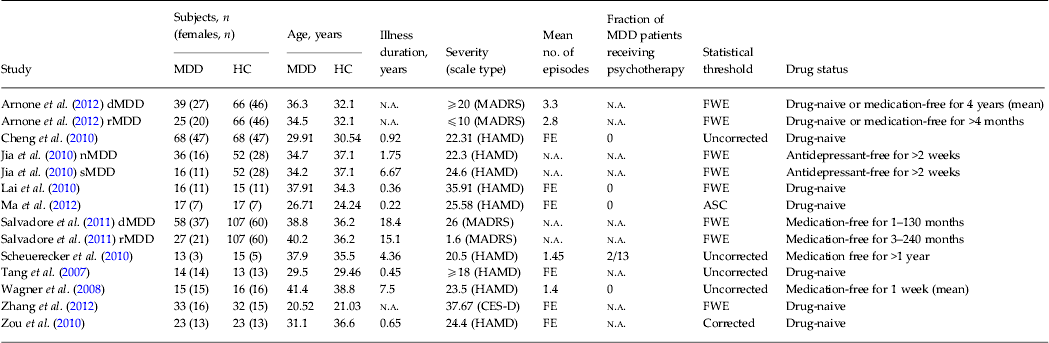
MDD, Major depressive disorder; HC, healthy control; dMDD, currently depressed subjects; n.a., not available; MADRS, Montgomery–Asberg Depression Rating Scale; FWE, familywise error correction; rMDD, subjects currently in remission; HAMD, Hamilton Depression Rating Scale; FE, first episode; nMDD, depressed patients without a history of suicide attempts; sMDD, depressed patients with a history of suicide attempts; ASC, AlphaSim correction; CES-D, Center for Epidemiological Studies Depression Scale.
Pooled meta-analysis of all included medication-free studies
A group comparison of MDD patients with healthy controls across the 14 datasets revealed grey matter volume reductions in the left inferior frontal gyrus, the right middle frontal gyrus extending to the inferior frontal gyrus, the right superior frontal gyrus, the left superior frontal gyrus extending to the middle frontal gyrus, and the bilateral parahippocampal gyrus and hippocampus (Table 2 and Fig. 2). No grey matter volume increases were found.

Fig. 2. Regions showing reduced grey matter in medication-free major depressive disorder patients compared with healthy controls. L, Left; R, right; SFG, superior frontal gyrus; IFG, inferior frontal gyrus; MIFG, middle frontal gyrus; PG, parahippocampal gyrus; HP, hippocampus.
Table 2. Regional differences in grey matter volume between MDD patients and HC subjects identified by the present meta-analyses (voxelwise p < 0.005 and FWHM 20 mm)

MDD, Major depressive disorder; FWHM, full width at half maximum; SDM, signed differential mapping; HC, healthy control; BA, Brodmann area.
Jack-knife sensitivity analysis
Whole-brain jack-knife sensitivity analysis showed that grey matter reduction in the right middle frontal gyrus and the right superior frontal gyrus was highly replicable, being preserved throughout all 14 combinations of datasets. The results in the left inferior and superior frontal gyrus remained significant in all but one combination, those in the left parahippocampal gyrus and the hippocampus in all but two combinations, and those in the right parahippocampal gyrus and hippocampus in all but three combinations.
Subgroup meta-analysis of first-episode medication-naive studies
In six datasets recruiting first-episode medication-naive MDD patients the subgroup analysis revealed grey matter volume increase relative to controls in the right thalamus, and decrease in the right superior and middle frontal gyrus, the right parahippocampal gyrus and hippocampus extending to the amygdala, and the left parahippocampal gyrus and hippocampus (Table 2).
Subgroup meta-analysis of medication wash-out studies
The wash-out subgroup analysis of eight datasets showed grey matter volume increase relative to controls in the bilateral anterior cingulate cortex, and decrease in the left inferior and middle frontal gyrus extending to the superior frontal gyrus, the right middle and superior frontal gyrus, and the right parahippocampal gyrus (Table 2).
Meta-regression analysis
The percentage of female patients with MDD (i.e. in the whole MDD group in each study) was negatively associated with grey matter volume in the right hippocampus (r = 0.814, permutation-derived p < 0.0001) (Fig. 3). However, this result should be interpreted with some caution, being driven only by four studies (Tang et al. Reference Tang, Wang, Xie, Liu, Li, Su, Liu, Hu, He and Blumberg2007; Wagner et al. Reference Wagner, Koch, Schachtzabel, Reichenbach, Sauer and Schlösser2008; Lai et al. Reference Lai, Hsu and Wu2010; Scheuerecker et al. Reference Scheuerecker, Meisenzahl, Koutsouleris, Roesner, Schopf, Linn, Wiesmann, Bruckmann, Moller and Frodl2010). The mean age of patients, illness duration, and the mean number of episodes were not associated with MDD-related grey matter changes, at least linearly.

Fig. 3. Meta-regression results show that the percentage of female major depressive disorder patients is negatively correlated with grey matter in the right hippocampus (RHP). In the graphs, the effect sizes needed to create this plot have been extracted from the peak of maximum slope significance, and each study is represented as a dot, whose size reflects sample size. Large dots indicate samples with >40 patients; medium dots, 20–40 patients; and small dots, < 20 patients. The regression line (meta-regression signed differential mapping slope) is shown.
Discussion
This is to our knowledge the first meta-analysis of VBM studies in medication-free MDD patients, examining how age, sex and clinical characteristics affect grey matter volumes without the interference of medication effects. The medication-free MDD patients, compared with healthy control subjects, displayed significantly and robustly reduced grey matter in the prefrontal and limbic regions. Notably, increasing percentage of female patients correlated with decreasing grey matter volumes in the right hippocampus. Although most of the results of the two subgroup meta-analyses were consistent, there were some differences. Increased grey matter volume in the right thalamus compared with controls was only seen in the first-episode medication-naive studies, which might suggest that increased right thalamus volume is a trait for MDD, i.e. directly related to MDD per se. Conversely, increased grey matter volume in the bilateral ACC compared with controls was only seen in the subgroup meta-analysis of wash-out studies, which is at least consistent with the idea that increased ACC volume is an effect of medication. To our knowledge, there are no prospective randomized controlled studies of the long-term effects of antidepressants on brain structure, and this finding in the ACC merits further investigation. This result differs from that of previous meta-analyses which showed decreased grey matter volume in the bilateral ACC (Bora et al. Reference Bora, Fornito, Pantelis and Yucel2012; Du et al. Reference Du, Wu, Yue, Li, Liao, Kuang, Huang, Chan, Mechelli and Gong2012; Lai, Reference Lai2013) and decreased grey matter in the left thalamus (Du et al. 2012), compared with controls, which might be explained by the fact that all of these studies included both medicated and unmedicated MDD patients.
Findings in the hippocampus and parahippocampus
The reduction in grey matter volume in the bilateral hippocampus and parahippocampal gyrus in medication-free MDD patients, compared with controls, which we identified in the present study is consistent with a recent meta-analysis of traditional MRI and computed tomography studies on depression (Kempton et al. Reference Kempton, Salvador, Munafo, Geddes, Simmons, Frangou and Williams2011). The hippocampus and parahippocampus are key regions of the limbic system that play a central role in the regulation of emotions, motivation, memory and affective dimension of pain (Fang et al. Reference Fang, Jin, Wang, Li, Kong, Nixon, Zeng, Ren, Tong, Wang and Hui2009; Fossati, Reference Fossati2012); they are extensively connected with other cortical and subcortical structures in the frontal and temporal lobes and play an important role in cognitive processes of MDD (Price & Drevets, Reference Price and Drevets2010). Thus grey matter damage in these regions may be related to memory function impairment and emotion processing, which is the main clinical manifestation of MDD. Furthermore, in a 3-year longitudinal study, hippocampal grey matter volume declined significantly more in MDD patients than in controls, and also significantly more in patients who did not remit during the period than those who did, suggesting that depression may lead to changes in hippocampal morphologic structure (Frodl et al. Reference Frodl, Koutsouleris, Bottlender, Born, Jager, Scupin, Reiser, Moller and Meisenzahl2008).
Findings in the prefrontal cortex
Our finding of decreased grey matter in medication-free MDD patients in the superior, middle and inferior frontal gyrus compared with controls corresponds to the right DLPFC and left DLPFC and ventrolateral prefrontal cortex. Postmortem studies have demonstrated reductions in glial cell density and neuronal size in the deeper cortical layers of the PFC in MDD which may underlie these grey matter abnormalities detected by neuroimaging (Rajkowska et al. Reference Rajkowska, Miguel-Hidalgo, Wei, Dilley, Pittman, Meltzer, Overholser, Roth and Stockmeier1999; Rajkowska, Reference Rajkowska2000; Cotter et al. Reference Cotter, Mackay, Chana, Beasley, Landau and Everall2002). Furthermore, there is evidence that increased glutamatergic transmission in depression may cause excitotoxic damage and cellular loss, dendritic reshaping and consequent grey matter morphometric abnormalities (Price & Drevets, Reference Price and Drevets2010). The lateral prefrontal lobe has been theoretically identified as important in the pathophysiology of MDD, being associated with cognition dysfunction and emotion regulation in depression (Levesque et al. Reference Levesque, Eugene, Joanette, Paquette, Mensour, Beaudoin, Leroux, Bourgouin and Beauregard2003; Ochsner et al. Reference Ochsner, Ray, Cooper, Robertson, Chopra, Gabrieli and Gross2004; Phan et al. Reference Phan, Fitzgerald, Nathan, Moore, Uhde and Tancer2005; Matsuo et al. Reference Matsuo, Glahn, Peluso, Hatch, Monkul, Najt, Sanches, Zamarripa, Li, Lancaster, Fox, Gao and Soares2007; Walter et al. Reference Walter, Wolf, Spitzer and Vasic2007; Price & Drevets, Reference Price and Drevets2010). Moreover a recent longitudinal therapeutic study on SSRI-treated MDD patients has shown grey matter changes in the left DLPFC, which is associated with top-down neuronal modulation of emotion (Smith et al. Reference Smith, Chen, Baxter, Fort and Lane2013); a defect in the regulation of negative affect due to DLPFC dysfunction is a plausible mechanism for DLPFC involvement in depression (Koenigs & Grafman, Reference Koenigs and Grafman2009). Therefore, the identified grey matter abnormality in the prefrontal gyrus may be part of the structural underpinning of the prefrontal dysfunction in MDD patients.
Sex effects
The percentage of female MDD patients was negatively correlated with grey matter volume in the right hippocampus, which indicates that female medication-free MDD patients tend to have smaller right hippocampus. Other structural imaging studies have shown significant sex effects consistent with the present results: greater hippocampal volume in male compared with female MDD patients (Steffens et al. Reference Steffens, Byrum, McQuoid, Greenberg, Payne, Blitchington, MacFall and Krishnan2000; Frodl et al. Reference Frodl, Meisenzahl, Zetzsche, Born, Groll, Jager, Leinsinger, Bottlender, Hahn and Moller2002; Kronmuller et al. Reference Kronmuller, Schroder, Kohler, Gotz, Victor, Unger, Giesel, Magnotta, Mundt, Essig and Pantel2009), and specifically greater right hippocampal volumes in male than female psychotropic-naive patients with familial MDD (MacMaster et al. Reference MacMaster, Mirza, Szeszko, Kmiecik, Easter, Taormina, Lynch, Rose, Moore and Rosenberg2008). Sex differences are seen in epidemiological and clinical aspects of MDD: rates of MDD are higher in females than males (Kessler et al. Reference Kessler, Chiu, Demler, Merikangas and Walters2005); most studies report greater sensitivity in women to the depressogenic effect of stressful life events (Post et al. Reference Post, Weiss, Smith, Rosen and Frye1995); female MDD patients show higher rates of suicide attempts and co-morbid anxiety disorders, while male MDD patients are more likely to show psychomotor agitation and have co-morbid substance abuse disorders (Roeloffs et al. Reference Roeloffs, Fink, Unutzer, Tang and Wells2001; Marcus et al. Reference Marcus, Young, Kerber, Kornstein, Farabaugh, Mitchell, Wisniewski, Balasubramani, Trivedi and Rush2005). The sex difference in right hippocampus volume in MDD may contribute to differences in epidemiological and clinical aspects of depression. However, other reports (Vakili et al. Reference Vakili, Pillay, Lafer, Fava, Renshaw, Bonello-Cintron and Yurgelun-Todd2000; MacQueen et al. Reference MacQueen, Campbell, McEwen, Macdonald, Amano, Joffe, Nahmias and Young2003; Neumeister et al. Reference Neumeister, Wood, Bonne, Nugent, Luckenbaugh, Young, Bain, Charney and Drevets2005) found no such sex difference. The heterogeneous findings of structural imaging studies can perhaps be explained by the highly variable female:male ratio among studies.
Limitations and conclusions
This study has several limitations. First, peak-based meta-analyses are based on summarized (i.e. coordinates from published studies) rather than raw statistical brain maps, and this approach may result in less accurate results (Nakao et al. Reference Nakao, Radua, Rubia and Mataix-Cols2011). Second, the main meta-analysis included primary studies enrolling medicated MDD patients who underwent a wash-out period before MRI scanning and the results of the two subgroups were to some extent different. Clearly the best way to minimize the effects of illness duration and antidepressant treatment is to focus on drug-naive MDD patients. However, the sample size of our drug-naive subgroup analysis is necessarily small, which limits the generalizability of the results. Meta-analyses of first-episode drug-naive MDD are needed when the number of published VBM studies on such patients permits. Third, the meta-regression finding that the percentage of female MDD patients was negatively correlated with grey matter volume in the right hippocampus was driven by only four studies.
In summary, our findings indicate that medication-free MDD patients have significantly and robustly reduced grey matter mainly in prefrontal and limbic brain regions excluding the confounding influences of medication status. The subgroup meta-analysis results suggest that increased right thalamus volume may be directly related to MDD, while increased ACC volume may be an effect of medication. Meta-regression results suggest that female medication-free MDD patients may have smaller right hippocampus, which may constitute the structural underpinnings of the sex differences observed in both epidemiological and clinical aspects of MDD. Future studies will benefit from the use of a longitudinal approach to track patients from disease onset and through follow-up and examine the dynamic effect of medication on brain structure in MDD patients. Longitudinal studies that investigate the dynamic brain structure change of competent MDD patients who reject all therapies are also encouraged.
Acknowledgements
This work was supported by the National Natural Science Foundation (grant nos. 81030027, 81227002 and 81220108013), the National Key Technologies R&D Programme (programme no. 2012BAI01B03), and the Programme for Changjiang Scholars and Innovative Research Team in University (PCSIRT, grant no. IRT1272) of China.
Declaration of Interest
None.







