Impact statement
Good water quality is mandatory in different phases of a successful aquaculture production, water intake, water use and waste discharges. However, unsustainable aquaculture practices can result in low yields and cause negative impacts on the environment and the human community. This review provides assessments of water quality in different aquaculture systems, and the impacts of their effluents on the natural water bodies. To optimize aquaculture production, and minimize their impacts on the environment, effective management of the water quality and wastes in aquaculture is needed. Major constraints in adequate aquaculture wastewater treatment, including high capital and operation costs of waste treatment systems, lack of incentives for waste treatment and lack of legislation and enforcement in discharges of raw aquaculture wastes, should be overcome. Possible solutions include technological innovations in production systems and wastewater treatments, increased professionals in water quality control and waste management, improved legislation, certification, financial assistance and incentives to farmers along the aquaculture industrial chains can be applied for a sustainable aquaculture sector. If water quality management can be effectively carried out, it would have a great long-term impact on the aquaculture industry.
Introduction
Aquaculture is the fastest-growing food-production sector, and its sustainable growth is vital to food security, ecosystem health, uninterrupted natural resource utilization, biodiversity conservation and socioeconomic resilience. In the face of declining capture fishery resources and rising demand for fish and fishery products, aquaculture has become the main source of aquatic food/protein supply and contributes to the food security of the global population (Boyd et al., Reference Boyd, McNevin and Davis2022; Troell et al., Reference Troell, Costa‐Pierce, Stead, Cottrell, Brugere, Farmery, Little, Strand, Pullin, Soto, Beveridge, Salie, Dresdner, Moraes-Valenti, Blanchard, James, Yossa, Allison, Devaney and Barg2023). However, there are concerns about the impacts of aquaculture activities on the environment and natural resources, such as habitat destruction, exploitation of wild fish stocks, fishmeal/fish oil requirements and waste disposal (Bull et al., Reference Bull, Cunha and Scudelari2021; Klootwijk et al., Reference Klootwijk, Alve, Hess, Renaud, Sørlie and Dolven2021). Different aquaculture systems (extensive, semi-intensive and intensive); types of systems (closed, semi-open and open); different cultured species and stocking densities can generate different environmental impacts (Figure 1). Environmental impacts can occur through three different processes such as consumption of natural resources, culture procedures/practices and generation of wastes. Each ecosystem has its own carrying capacity, and working within the limit is crucial to avoid negative impacts. The transition of traditional cultural practices to an intensified cultural system involves increased waste that requires proper treatment to avoid pollution and deleterious impacts on the environment (da Silva et al., Reference da Silva, Rotta, Fornari and Streit2022). With the high demand for aquaculture products, more farms are opting for intensive culture systems, which tend to affect the environment more than extensive and semi-intensive systems due to large amounts of waste containing toxins, drugs and chemicals in the former system (Zhang et al., Reference Zhang, Ma, Huang, Liu, Lu, Peng and Li2021; Nagaraju et al., Reference Nagaraju, Malegole, Chaudhary and Ravindran2022). Thus, unsustainable aquaculture activities could result in widespread habitat destruction, loss of biodiversity, declined fishery and other aquatic resources in the surrounding area (Valiela et al., Reference Valiela, Bowen and York2001; Polidoro et al., Reference Polidoro, Carpenter, Collins, Duke, Ellison, Ellison, Farnsworth, Fernando, Kathiresan, Koedam, Livingstone, Miyagi, Moore, Nam, Ong, Primavera, Salmo, Sanciangco, Sukardjo, Wang and Yong2010; Herbeck et al., Reference Herbeck, Unger, Wu and Jennerjahn2013; Cardoso-Mohedano et al., Reference Cardoso-Mohedano, Lima-Rego, Sánchez-Cabeza, Ruiz-Fernández, Canales-Delgadillo, Sánchez-Flores and Paez-Osuna2018).

Figure 1. Different aquaculture production systems in closed (tanks, ponds, and raceways) and open ecosystems (cages and extractive culture systems in lakes, rivers, and coastal waters).
In aquaculture production systems, poor water quality due to accumulation of toxic compounds, including ammonia, nitrite and hydrogen sulfide, together with low dissolved oxygen, hypoxic conditions, harmful algal blooms (HABs) and pathogenic bacteria can greatly affect the fish health through bacterial infections, poor growth and stress rendering them less tolerant to handling. Diseases in aquaculture systems are closely related to the environmental health. Uncontrolled diseases can rapidly decimate operations and can cause high mortality in aquaculture systems. Lusiastuti et al. (Reference Lusiastuti, Prayitno, Sugiani and Caruso2020) attributed the disease outbreaks, mass fish mortality and low aquaculture production to poor water quality associated with environmental degradation and climate change. Climate change can affect the aquaculture industry through flooding (too much water), drought (too little water) and changes in water quality. Decline in pH due to ocean acidification could seriously affect aquaculture, especially those in the coastal areas (Guo et al., Reference Guo, Huang, Luo, You and Ke2023). Hassan et al. (Reference Hassan, Rashid, Muhaimeed, Madlul, Al-Katib and Sulaiman2022) noted that improving water quality, maintaining stable environmental factors and controlling water exchange would reduce the occurrence of fish diseases in aquaculture production systems.
Untreated or improperly treated aquaculture discharges with high nutrient concentrations can cause eutrophication and water quality deterioration, hypoxia and HABs in adjacent water bodies (Zhang et al., Reference Zhang, Zhu, Mo, Liu, Wang and Zhang2018; Purnomo et al., Reference Purnomo, Patria, Takarina and Karuniasa2022). HABs can be a serious concern in coastal and inland waters (rivers, lakes and reservoirs) that receive aquaculture effluents. Lukassen et al. (Reference Lukassen, de Jonge, Bjerregaard, Podduturi, Jørgensen, Petersen, David, da Silva and Nielsen2019a) reported that the off-flavor compounds produced by the HABs especially geosmin in tilapia produced in cage aquaculture increased the risk of decreasing fish quality and value. Hu et al. (Reference Hu, Li, Ye, Chen, Xu, Dong, Liu and Li2022) reported that Lake Datong, a shallow lake in China, became eutrophic and its water quality deteriorated after the introduction of aquaculture.
Extraction of ground water for aquaculture can cause saltwater intrusion and salinization in coastal areas (Gopaiah et al., Reference Gopaiah, Chandra and Vazeer2023). All these environmental changes could affect the livelihoods of the local communities (da Silva et al., Reference da Silva, Rotta, Fornari and Streit2022; Nagaraju et al., Reference Nagaraju, Malegole, Chaudhary and Ravindran2022; Menon et al., Reference Menon, Arunkumar, Nithya and Shakila2023). Kim et al. (Reference Kim, Kim, Lee and Lee2022a) reported that an increasing number of farms in the coastal area resulted in the release of organic wastes derived from excess feed and fish metabolites. Yang et al. (Reference Yang, Zhao, Tong, Tang, Lai, Li and Tang2021) and Chiquito-Contreras et al. (Reference Chiquito-Contreras, Hernandez-Adame, Alvarado-Castillo, de J. Martínez-Hernández, Sánchez-Viveros, Chiquito-Contreras and Hernandez-Montiel2022) reported that approximately 27% to 49% of the feeds supplied to aquaculture production ponds are converted to fish products while the rest goes to wastes that are usually discharged into the nearby water bodies, and eventually form one of the factors that negatively affect the aquaculture value chain.
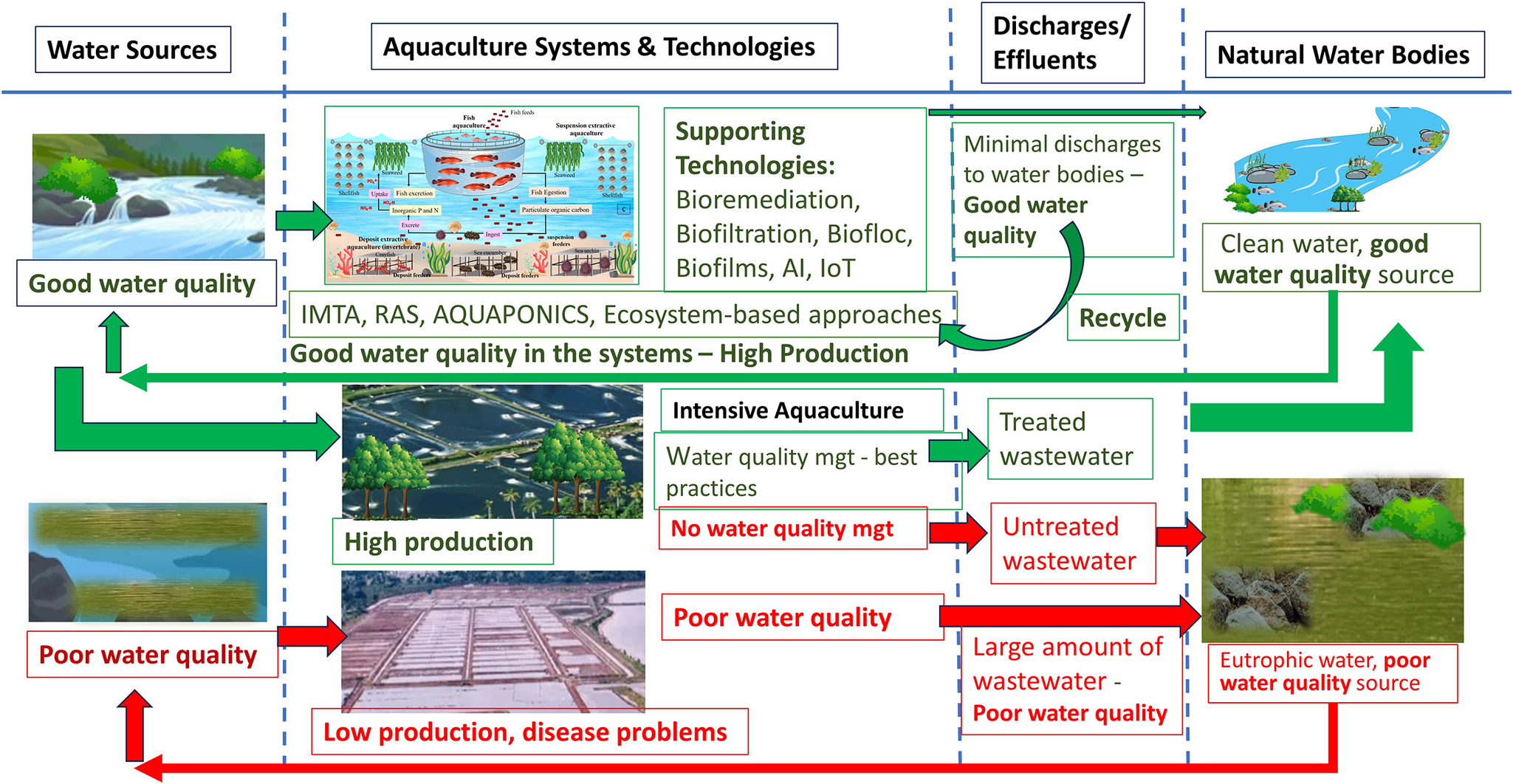
Water treatment technologies that are technically feasible, environmentally promising and financially profitable can be integrated into different aquaculture systems to make aquaculture industry a sustainable sector and contributes to the circular economy. Aquaculture wastes can be recovered and recycled using various technologies such as bioremediation, aeration, biocoagulation and biofiltration applied in various production systems such as recirculating aquaculture system (RAS), integrated multi-trophic aquaculture (IMTA) and aquaponics (aquaculture and hydroponics). In these circular economic activities, aquaculture wastes can generate additional products such as seaweeds, herbs, vegetables, mollusks and other by-products, while generating a clean water source that can be recycled and used for the fed culture (Figure 2). Legal instruments and authoritative interventions are also necessary for regulating aquaculture waste discharge and ensuring producers consider environmental impact and water quality management in their operations and practices. This review assessed the impacts of different production systems on the water quality and suggested possible approaches such as the use of environmentally friendly technological innovations and good governance in improving water quality management for a sustainable aquaculture industry.

Figure 2. Recycling of aquaculture wastes to create various economically important outputs and maintain good water quality for aquaculture production.
Pollution and threats to water quality in aquaculture systems
Most aquaculture systems require a thorough understanding of water quality and waste management for accurate treatment decisions to ensure healthy cultured organisms with high yields (Davidson et al., Reference Davidson, Redman, Crouse and Vinci2022). Ssekyanzi et al. (Reference Ssekyanzi, Nevejan, Kabbiri, Wesana and Stappen2022) reported that in Sub-Saharan Africa, limited knowledge of water quality is one of the main factors contributing to low production (<1% of global production) and slow growth of the aquaculture sector.
Major factors contributing to the deteriorating environment and water quality in the aquaculture industry include nutrients (17%), other pollutants, including emerging pollutants (12%), habitat loss (16%), HABs (9%), lack of treatment technologies (8%) and socioeconomic factors (38%) (Theuerkauf et al., Reference Theuerkauf, Morris, Waters, Wickliffe, Alleway and Jones2019). Nutrients play a major role in eutrophication, resulting in massive proliferation of HABs, such as cyanobacteria and dinoflagellates and high mortality of cultured organisms in cultured systems (Table 1). Cyanobacterial blooms are also commonly associated with toxic–odor compounds such as geosmin and 2-methylisoborneol (2-MIB) which impart an unpleasant taste to water and cultured organisms. Marques et al. (Reference Marques, da Silva, de Oliveira, Cunha and Sobral2018) and Ryan et al. (Reference Ryan, Palacios, Encina, Graeber, Osorio, Stubbins, Woelfl and Nimptsch2022) noted the negative impacts of an intensive aquaculture farm on effluent water quality due to excessive nutrients, especially phosphorus and nitrogen.
Table 1. Major problems and mitigating measures in water quality management in aquaculture production systems

Emerging pollutants such as microplastics (Table 1) can cause health implications such as reduced feeding rate, gill malfunction, reduced reproductive capacity and immune suppression of cultured animals (Mallik et al., Reference Mallik, Xavier, Naidu and Nayak2021). In aquaculture, plastic debris from aquaculture farms, rafts, cages, nets and other related production structures are sources of microplastics (Chen et al., Reference Chen, Jin, Tao, Wang, Xie, Yu and Wang2018; Krüger et al., Reference Krüger, Casado-Coy, Valle, Ramos, Sánchez-Jerez, Gago, Carretero, Beltran-Sanahuja and Sanz-Lazaro2020). In addition, biofilms formed on microplastic particles are sources of pathogenic bacteria that can negatively affect aquaculture (Cholewińska et al., Reference Cholewińska, Moniuszko, Wojnarowski, Pokorny, Szeligowska, Dobicki, Polechonski and Górniak2022).
Contamination in water sources for aquaculture production
Availability of clean water for aquaculture is an important consideration in site selection for aquaculture operation. In fact, suitable site selection for aquaculture activities is vital to alleviate potential problems associated with pollution and conflicting activities, and to ensure that the selected water body would be a conducive growing environment without jeopardizing the existing ecosystems (Table 1). Brigolin et al. (Reference Brigolin, Lourguioui, Taji, Venier, Mangin and Pastres2015) and Jayanthi et al. (Reference Jayanthi, Thirumurthy, Samynathan, Kumararaja, Muralidhar and Vijayan2021) used remote sensing, geospatial tools and mathematical models in combination with water quality factors, environmental characteristics and socioeconomic data to identify suitable areas for cage aquaculture in estuaries and coastal areas. Vaz et al. (Reference Vaz, Sousa, Gómez-Gesteira and Dias2021) and Arega et al. (Reference Arega, Lee and Choi2022) developed a habitat suitability model based on water quality, hydrodynamics and biogeochemistry for aquaculture site selection.
In aquaculture systems, pollutants can originate from both allochthonous sources (such as feeds, fertilizers and/or polluted water sources) and autochthonous sources (phytoplankton biomass, metabolites). Polluted water from rivers and coastal waters can seriously affect the health and growth of the culture species, resulting in high mortality and low yields. In closed culture systems such as ponds and tanks, the quality of the intake water can be controlled. Under limited circumstances, low-quality water can be first treated before use, although the production would still be lower compared to those with clean water intake. In aquaculture systems located in open waters such as lakes and coastal waters (Figure 1), yields are highly dependent on the in situ water quality. In these, natural waters where cage aquaculture or extractive aquaculture is common, pollutants are mainly associated with anthropogenic activities in the catchment and upstream areas. Kim et al. (Reference Kim, Kim, Lee and Lee2022a) used 15-N isotopic signatures to show that organic pollutants in estuaries and coastal areas were mainly contributed by sources related to anthropogenic activities, including organic fertilizers and aquaculture discharges exported through rivers.
To ensure the sustainability of aquaculture production through sound water quality management of open waters, Liu et al. (Reference Liu, Chen, Wang, Wang, Zhang, Li, Lin, Xiong, Zhu, Liu, Zhu and Shen2023a) proposed a watershed management framework using economic-based and water quality-based protection strategies to manage catchment areas for sustainable development. To prevent nonpoint source pollution, interactions between land cover, landscape pattern and design and pollution loading should be assessed and optimized (Ouyang et al., Reference Ouyang, Song, Wang and Hao2014; Falconer et al., Reference Falconer, Telfer and Ross2018; Rong et al., Reference Rong, Zeng, Su, Yue, Xu and Cai2021).
Factors affecting water quality in aquaculture production systems
Water quality in aquaculture systems is influenced by various physical, chemical and biological factors such as temperature, light, pH, dissolved oxygen, organic matter/nutrients, microorganisms and various biological interactions (Table 2). Climate change could exert drastic fluctuations in these physical chemical factors that would affect water quality, increase the incidence of fish diseases and cause high fish mortality and production (Lusiastuti et al., Reference Lusiastuti, Prayitno, Sugiani and Caruso2020). Alam et al. (Reference Alam, Sarkar, Miah and Rashid2021) reported that Nile tilapia, Oreochromis niloticus, produced fewer eggs under high temperatures associated with climate change, and suggested effective management strategies to overcome the low egg production in commercial fish hatcheries. Ocean acidification and decrease in pH caused problems in shellfish aquaculture, such as oysters (Abisha et al., Reference Abisha, Krishnani, Sukhdhane, Verma, Brahmane and Chadha2022; Mayrand and Benhafid, Reference Mayrand and Benhafid2023). Higher sea levels could cause positive consequences such as the creation of new habitats in the coastal waters or negative impacts like saltwater intrusion. Increased wind speed and waves caused sediment suspension and high turbidity that affected water quality and aquaculture activities (Shen et al., Reference Shen, Lin, Ye, Ren, Zhao and Duan2023). Mitigating measures to overcome impacts of physicochemical changes include adaptations in production systems, good culture strategies such as species diversification, and use of predictive models (Table 2). Abisha et al. (Reference Abisha, Krishnani, Sukhdhane, Verma, Brahmane and Chadha2022) suggested the development of climate-resilient aquaculture through adaptations to environmental factors that have negative impacts on organisms to minimize the impacts of climate change. Shen et al. (Reference Shen, Lin, Ye, Ren, Zhao and Duan2023) used satellite remote sensing to assess the impacts of the environment and improve the ecological and environmental regulations to support the sustainable development of the coastal area.
Table 2. Factors affecting water quality in aquaculture production systems and mitigation measures

High organic wastes in aquaculture systems, mainly from excess feeds and metabolites, caused water quality degradation characterized by high ammonia, nitrate and soluble reactive phosphorus, high biological oxygen demand (BOD), high chemical oxygen demand (COD) and low dissolved oxygen (Table 2). Phosphorus (P) can be a source of environmental contamination and eutrophication in aquaculture systems if not adequately removed from the wastewater. In terms of nitrogen, the proportion of toxic unionized ammonia (NH3) depends on the total ammonia concentration (ionized ammonium ion) and NH3 in the water column, which is in turn governed by water temperature and pH. Once ammonia concentrations in the water are high, fish are less able to excrete ammonia through gill diffusion resulting in the accumulation of ammonia in fish tissues, which would finally affect fish health and growth. Zhang et al. (Reference Zhang, Wang, Sun, Jiang, Qian, Wang and Li2022a) reported that toxic ammonia can reduce the quality and yield of Japanese sea perch (Lateolabrax japonicus). Due to its adverse effects on aquaculture species, ammonia concentrations in production systems should be closely monitored. Yu et al. (Reference Yu, Yang, Li and Chen2021) used a hybrid soft computing method to accurately predict ammonia concentrations in aquaculture water in real time. Temperature, dissolved organic carbon and redox potential are the primary drivers of chemical fluxes in freshwater aquaculture ponds (Yuan et al., Reference Yuan, Liu, Xiang, He, Kang and Ding2021).
Accumulation of organic matter in the pond bottom can be the main cause of hypoxic conditions in enriched aquaculture ponds (Yang et al., Reference Yang, Zhao, Tong, Tang, Lai, Li and Tang2021). Under anaerobic conditions, high organic matter accumulation can produce methane (CH4), hydrogen sulfide (H2S) and nitrous oxide (N2O), which could adversely affect water quality (Table 2). Toxic H2S, commonly found in production systems with low oxygen, could cause sudden fish/shrimp mass mortality. Wu et al. (Reference Wu, Hu, Hu, Chen, Yu, Zou and Liu2018b) reported that CH4 and N2O fluxes in inland aquaculture ponds were positively correlated to temperature and sediment organic carbon, and negatively correlated to dissolved oxygen concentration. Chen et al. (Reference Chen, Dong, Wang, Gao and Tian2016) and Yang et al. (Reference Yang, Zhang, Lai, Tan, Jin and Tong2018) noted that substantial amounts of CH4 and carbon dioxide were released from mariculture ponds. In freshwater aquaculture ponds, Zhao et al. (Reference Zhao, Zhang, Xiao, Jia, Zhang, Wang, Zhang, Xie, Pu, Liu, Feng and Lee2021) reported that high concentrations of CH4 were released and showed that dredging of the pond bottom as part of pond preparation was more effective in reducing CH4 compared to aeration. Thus, there is a need for immediate and continuous removal of toxic compounds such as ammonia, nitrite, H2S and CH4 in aquaculture systems.
Nutrient-rich waters are also associated with cyanobacterial blooms that could produce toxic–odor compounds such as geosmin and 2-MIB, causing an unpleasant taste to water and cultured organisms. Although a variety of bacteria and fungi produce geosmin, cyanobacteria including planktonic and benthic species belonging to Nostocales, Oscillatoriales and Synechococcales are major producers of geosmin (Watson et al., Reference Watson, Monis, Baker and Giglio2016; John et al., Reference John, Koehler, Ansell, Baker, Crosbie and Jex2018). Cyanobacterial toxins pose threats and risks to human and animal health. Cyanobacteria proliferate rapidly in eutrophic waters due to their ability to float and overcome light limitations (Table 2). Geosmin has been found to cause off-flavor in a wide range of environments including RAS (Azaria and van Rijn, Reference Azaria and van Rijn2018; Lukassen et al., Reference Lukassen, Podduturi, Rohaan, Jørgensen and Nielsen2019b). Lukassen et al. (Reference Lukassen, de Jonge, Bjerregaard, Podduturi, Jørgensen, Petersen, David, da Silva and Nielsen2019a) reported that higher densities of geosmin-producing bacteria were found in the intestinal mucous layer and digestive system of tilapia (O. niloticus) compared to the water column, indicating that probiotics can be used to manage intestinal microflora to improve fish quality. Due to the detrimental impacts of HABs on aquaculture production systems, environmental and human health, and socioeconomics, microalgal toxic species distribution and abundance should be closely monitored for early detection and preventive action. In fact, reduction of the external nutrient load is the most fundamental aspect of cyanobacterial control (Kibuye et al., Reference Kibuye, Zamyadi and Wert2021). Derot et al. (Reference Derot, Yajima and Jacquet2020) used two machine learning models with a long-term base to forecast HABs. Pal et al. (Reference Pal, Yesankar, Dwivedi and Qureshi2020) suggested biological options such as bacteria, viruses, fungi and zooplankton for controlling HABs. John et al. (Reference John, Koehler, Ansell, Baker, Crosbie and Jex2018) developed a novel polymerase chain reaction method targeting the geosmin synthase gene (geoA) to assess all important sources of geosmin, while Ma et al. (Reference Ma, Wang, Li, Peng and Yang2018) showed that chlorine aqueous solution under ultraviolet light could effectively remove geosmin and 2-MIB in acidic conditions.
In addition to nutrients, aquaculture systems can also be subjected to other pollutants such as antibiotics and heavy metals that could eventually affect the quality of the produce (Table 2). Le et al. (Reference Le, Hoang, Phung, Nguyen, Rochelle-Newall, Duong, Pham, Phung, Nguyen, Le, Pham, Nguyen and Le2022) noted heavy metal pollution in the aquaculture coastal area and emphasized the need for good management practices if sustainable aquaculture is to persist in the coastal area. The use of antibiotics and chemicals in aquaculture can also have far-reaching effects on ecological food pyramids. Fernanda et al. (Reference Fernanda, Liu, Yuan, Ramalingam, Lu and Sekar2022) showed that water quality parameters in aquaculture ponds were significantly correlated with the abundance of antibiotic-resistant (AR) genes which were brought down by a river polluted by various sources from the cultivated and industrial lands. In the environment, the partitioning and distribution of antibiotics are positively correlated to salinity, suspended solids, pH, ammonia and zinc, and negatively correlated to temperature, dissolved oxygen, phosphate, COD, oil, copper and cadmium (Li et al., Reference Li, Wen, Bao, Huang, Mu and Chen2022a). Ecological and biological risks of antibiotics are high and can be detrimental to aquaculture products. Chen et al. (Reference Chen, Wu, Li, Wang, Song, Wang and Yan2022) developed a biomarker using cyanobacterial carbonic anhydrase for monitoring antibiotics. Chemicals used in aquaculture should also be removed before discharging wastewater into the surrounding environment. Sulfonamides from aquaculture wastewater can be degraded using laccase-syringaldehyde mediator system through response surface optimization, degradation kinetics and degradation pathways (Lou et al., Reference Lou, Wu, Ding, Zhang, Zhang, Zhang, Han, Liu, He and Zhong2022). Pandey et al. (Reference Pandey, Daverey, Dutta and Arunachalam2022) suggested the removal of malachite green, which is commonly used for disease treatment in aquaculture ponds, using laccase immobilized biochar. Yanuhar et al. (Reference Yanuhar, Musa, Evanuarini, Wuragil and Permata2022) reported that water quality in concrete ponds can be improved by aeration, filtration and reduction of organic matter by optimizing the feed.
In addition to physical and chemical parameters, disease agents such as bacteria, fungi and other pathogenic organisms can also affect water quality and aquaculture performance (Table 2). Microbial communities in aquaculture systems are shaped by the environmental conditions which are in turn influenced by inland discharges, climate changes and anthropogenic pressures. Swathi et al. (Reference Swathi, Shekhar and Karthic2021) reported that water quality parameters were closely related to the outbreak of white spot disease in shrimp culture ponds. Thus, regular monitoring and estimating microbial diversity would allow farmers to link water quality parameters to subsequent fish performance and assess the environmental health of the aquaculture systems and the vicinity for early detection of microbial conditions that could lead to impaired fish health.
Water quality management in aquaculture production systems and methods to enhance it
Water quality in aquaculture production systems
Aquaculture production systems including RAS, IMTA, aquaponics (aquaculture and hydroponics) and ecosystem-based approaches were designed and constantly improved to enhance water quality and production (Table 3). These integrated production systems which have zero-water exchange and produce microorganisms as food sources, can be integrated with different types of biofiltration, biocoagulation, bioflocculation and biological interactions including bioflocs and bioremediation (Xu et al., Reference Xu, Du, Qiu, Zhou, Li, Chen and Sun2021; Igwegbe et al., Reference Igwegbe, Ovuoraye, Białowiec, Okpala, Onukwuli and Dehghani2022) to enhance their wastewater treatment performance (Table 4).
Table 3. Aquaculture production systems for improving water quality in aquaculture

Table 4. Technologies and processes for improving water quality in aquaculture systems

Aquaponics
Aquaponics, the integration of aquaculture and hydroponics, is conceptually based on the efficient use of water and recycling of accumulated organic nutrients using plants, as one of the effective approaches in addressing the problems of aquaculture wastewater treatment, pollution in public waters, improved water quality in culture systems and sustainable aquaculture development (Yep and Zheng, Reference Yep and Zheng2019; Chiquito-Contreras et al., Reference Chiquito-Contreras, Hernandez-Adame, Alvarado-Castillo, de J. Martínez-Hernández, Sánchez-Viveros, Chiquito-Contreras and Hernandez-Montiel2022); Okomoda et al., Reference Okomoda, Oladimeji, Solomon, Olufeagba, Ogah and Ikhwanuddin2023). Essentially, aquaponics uses bacterial processes and enhances plant nutrient uptake to recover and recycle nutrients from aquaculture systems (Kalayci Kara et al., Reference Kalayci Kara, Fakıoğlu, Kotan, Atamanal and Alak2021; Chen et al., Reference Chen, Kim, Thatcher, Hamilton, Alva, Zhou (George) and Brown2023). Sopawong et al. (Reference Sopawong, Yusoff, Zakaria, Khaw, Monir and Amalia2023) showed that integrating fish culture and plants in a bio-green floating system significantly improved water quality, fish health and aquaculture production. In addition, aquaponics overcomes the land scarcity for aquaculture as the system can be constructed and designed to fit any area available, such as in urban areas and water-scarce areas. Palm et al. (Reference Palm, Knaus, Appelbaum, Goddek, Strauch, Vermeulen, Jijakli and Kotzen2018) and Obirikorang et al. (Reference Obirikorang, Sekey, Gyampoh, Ashiagbor and Asante2021) demonstrated the increased efficiency of aquaculture production in aquaponics improvised for commercial aquaculture production and food security. To make the aquaponics more effective, Calone et al. (Reference Calone, Pennisi, Morgenstern, Sanyé-Mengual, Lorleberg, Dapprich, Winkler, Orsini and Gianquinto2019) and Ekawati et al. (Reference Ekawati, Ulfa, Dewi, Amin, Salamah, Yanuar and Kurniawan2021) combined it with RAS as aquaponic-RAS (A-RAS), which proved to be effective in improving water quality, survival rate, feed conversion ratio (FCR) and yield in catfish aquaculture (Table 3). Based on the same principle, Goddek and Körner (Reference Goddek and Körner2019) designed RAS-hydroponic multi-loop aquaponic system for better fish and plant production with flexible sizing. Liu et al. (Reference Liu, Hu, Song, Chen and Zhu2019) introduced crayfish integrated system for efficient use of waste for rice production. There are different combinations of fed and extractive species in different systems to improve water quality, such as catfish, plants and bacteria in hydroponic-biofilm and NFT systems (Mohapatra et al., Reference Mohapatra, Chandan, Panda, Majhi and Pillai2020; Li et al., Reference Li, Wang, Liu, Luo, Rauan, Zhang, Li, Yu, Dong and Gao2022b) to improve biofilter and ammonia removal efficiencies. Addy et al. (Reference Addy, Kabir, Zhang, Lu, Deng, Current, Griffith, Ma, Zhou, Chen and Ruan2017) showed that microalgae were more efficient in ammonia removal compared to plants in aquaponic co-cultivation. Other technologies such as biochar-supplemented planting panel system, polylactic acid addition and smart sensing systems have been integrated into the design of aquaponics to improve water quality (Table 3).
Integrated multi-trophic aquaculture
The concept of IMTA utilizes complementary aquaculture species along the food chain in the process of eating and being eaten such that wastes are fully recycled and minimal pollutants are released to the adjacent waters (Figure 3). In IMTA system, commercially important fed species (the main fish or invertebrates that consume given feeds) are cultured together with commercially important extractive species (aquatic species such as seaweeds or mollusks that feed/use the waste of other species) so that ecological balance and water quality in the system could be maintained (Figure 3). Since feeding is an important factor in an IMTA system, Flickinger et al. (Reference Flickinger, Costa, Dantas, Proença, David, Durborow, Moraes-Valenti and Valenti2020) showed that feed management is important to determine the water quality that translates into prawn and fish production in IMTA.

Figure 3. Integrated multitrophic aquaculture (IMTA) systems; in tanks (A), in ponds (B) and in coastal waters (C).
The selection of the species from various trophic is based on their physiological and ecology functions to ensure a complete recycling of organic matter in the system with minimal wastes and good water quality, which contributes to the sustainability of the aquaculture industry (Table 3). Largo et al. (Reference Largo, Diola and Marababol2016) reported the use of abalone (donkey’s ear, Haliotis asinina) as fed species and seaweeds (Gracilaria heteroclada and Eucheuma denticulatum) as the inorganic nutrient extractive species. Seaweeds functioned effectively in sequestering nutrients in various fish and shellfish cultures to minimize the impacts of pollution and improve water quality not only in aquaculture systems, but also in the related water bodies (Table 3). Kelp (Macrocystis pyifera) farms in a macroalgae-based IMTA were used to sequester nitrogenous compounds from salmon aquaculture effluents resulting in low chlorophyll concentrations and improved water quality (Hadley et al., Reference Hadley, Wild-Allen, Johnson and Macleod2018). In freshwater IMTA, Paolacci et al. (Reference Paolacci, Stejskal, Toner and Jansen2022) showed that duckweed, Lemna spp., could substantially remove total nitrogen and total phosphorus, maintain good water quality and increase aquaculture yields. In addition to macroalgae, microalgae can be introduced in IMTA in the form of periphyton and/or microalgae–bacterial consortia to reduce nutrients and other pollutants, improve water quality and produce algal biomass for enhancement of culture yields in the system (Milhazes-Cunha and Otero, Reference Milhazes-Cunha and Otero2017).
Recirculating aquaculture system
The RAS is a closed-circuit high-density aquatic animal farming where water from fish tanks is recirculated to remove solid and liquid wastes, and the purified water is returned to the aquaculture tanks (Figure 4). It is designed to provide a more controlled aquaculture system to reduce water usage and produce less wastes (both liquid and solid wastes), and thus it is more efficient and economical compared to the conventional flow-through and cage aquaculture systems (Table 3). In RAS, the relative water renewal rate can be optimized, the fish FCR decreased and the growth rate increased (Pulkkinen et al., Reference Pulkkinen, Kiuru, Aalto, Koskela and Vielma2018). As excess and poor-quality feeds can cause water quality problems in RAS, Kamali et al. (Reference Kamali, Ward and Ricardez-Sandoval2022) took into account the effects of feeding regimes on the accumulation of ammonia and dissolved oxygen in designing a new RAS to enhance the sustainability of aquaculture.

Figure 4. A recycling aquaculture system with an additional algae/plant culture compartment.
The efficiency of RAS in water quality management could be enhanced by combining the system with other functional components such as depuration system to eliminate off-flavor, microalgae system to enhance nutrient removal and bacterial communities as in simultaneous partial nitrification, anammox and denitrification system to enhance organic–inorganic matter recycling (Table 3). Biofiltration in RAS functions to convert ammonia to the less toxic form, nitrate. According to Santos et al. (Reference Santos, Ortiz-Gándara, Del Castillo, Arruti, Gómez, Ibáñez, Urtiaga and Ortiz2022), nitrate is about 100–200 folds less toxic.
Other alternative methods of nutrient removal such as direct or indirect oxidation, adsorption by zeolites and activated carbon, air stripping and reverse osmosis have their own drawbacks in terms of low efficiency and high energy costs (Díaz et al., Reference Díaz, Ibáñez, Gómez, Urtiaga and Ortiz2012; Gendel and Lahav, Reference Gendel and Lahav2013). Yogev et al. (Reference Yogev, Vogler, Nir, Londong and Gross2020) showed that P from RAS can be efficiently (>99%) removed through biomineralization in an anaerobic reactor and reused as fertilizer. For other toxic compounds, Bergstedt et al. (Reference Bergstedt, Skov and Letelier-Gordo2022) proposed the use of hydrogen peroxide to remove H2S from a saltwater RAS. RAS is advantageous in areas with limited land and water. In countries with severe water shortages, such as Gulf Cooperation Council countries, RAS is useful for recycling wastewater to overcome water scarcity for aquaculture (Qureshi, Reference Qureshi2022).
Integration of production systems using ecosystem-based approaches for water quality improvement
In most aquaculture systems, toxic compounds such as ammonia, nitrite and H2S can deteriorate water quality, increase mortality and reduce yields. Although Aquaponics, IMTA and RAS have been designed individually to improve water quality and increase yields, integration of these production system could increase the efficiencies and performances of aquaculture systems. Integration of A-RAS (Aquaponics and RAS), and I-RAS (IMTA and RAS), supported by a variety of functional biological components such as bacteria and microalgae can make aquaculture production systems more productive, cost-effective and efficient with less water consumption and lower disease risks (Figure 5).

Figure 5. Integrated recycling aquaculture system (I-RAS) combining different systems and technologies (integrated multitrophic aquaculture (IMTA), biofloc, bioremediation, bacteria-microalgae consortium, water quality monitoring, and artificial intelligence-internet-of-things (AI-IoT)), to make the I-RAS more efficient and effective in recycling the waste, while enhancing water quality and aquaculture production.
Essentially aquaponics, IMTA, RAS and their combinations (A-RAS, I-RAS) are conceptually based on ecosystem-based approaches, where holistic integration and management of different ecosystem components are essential to maintain its ecological resilience and stability to ensure optimum production in closed aquaculture systems. However, ecosystem-based aquaculture system can also be carried out in the open system such as the integration of aquaculture and mangrove forest management in eco-green approach (Racine et al., Reference Racine, Marley, Froehlich, Gaines, Ladner, MacAdam-Somer and Bradley2021; Musa et al., Reference Musa, Mahmudi, Arsad, Lusiana, Wardana, Ompusunggu and Damayanti2023). Ecosystem model with the co-culture of bivalves (as the grazers) and seaweeds (as nutrient consumers) would drive the nutrient-phytoplankton-zooplankton-detrital food web, increase the efficiency of waste recycling, improve water quality and enhance aquaculture yields (Cabral et al., Reference Cabral, Levrel, Viard, Frangoudes, Girard and Scemama2016; Park et al., Reference Park, Shin, Do, Yarish and Kim2018). Fan et al. (Reference Fan, Meirong, Hui, Jianguang, Lars and Zengjie2020) reported increased production of kelp (Saccharina japonica – seaweed) and oysters (Crassostrea gigas – a mollusk) with improved water quality, making the ecosystem resilient and stable (Table 3).
Methods for water quality enhancement
Different technologies (such as bioremediation, bio-floc and internet-of-things [IoT]) and processes (chemical reactions, filtrations, coagulations and flocculations) can be imbedded in closed aquaculture systems such as aquaponics and RAS, or open systems such as coastal waters to make the wastewater treatment and recycling more efficient, which in turns, improve water quality and enhance aquaculture yields (Table 4, Figure 5). Liu et al. (Reference Liu, Du, Tan, Xie, Luo and Sun2021b) integrated heterotrophic biofloc and nitrifying biofloc filters to simultaneously control ammonia, nitrite, nitrate, soluble reactive phosphorus and alkalinity with relevant functional microbes such as ammonia and nitrite-oxidizing bacteria, denitrifying bacteria, phosphorus accumulating organisms (PAOs), denitrifying PAOs and glycogen accumulating bacteria.
Bioremediation
Bioremediation involves the use of environmentally friendly microorganisms to mitigate pollution, improve water quality and maintain ecological health in aquaculture systems (Devaraja et al., Reference Devaraja, Yusoff and Shariff2002; Sun et al., Reference Sun, Li, Tang, Lin, Zhao and Chen2022). These bioremediation bacteria function to decompose organic wastes into useful inorganic compounds that are recycled to maintain a healthy nutrient cycle in various culture systems (Table 4). Bioremediation minimizes the use of antibiotics and drugs and thus, decreases the detrimental consequences of routinely used chemotherapeutic agents and produces safe aquatic products for human consumption (Sha et al., Reference Sha, Dong, Gao, Hashim, Lee and Li2022). In addition, these environmentally friendly bacteria help to improve the health conditions of cultured organisms by protecting them against infectious diseases, delivering antigens and providing several other health benefits in aquaculture.
Several bioremediation bacteria have been used in aquaculture and the most common and popular ones are Bacillus species. Geng et al. (Reference Geng, Li, Liu, Ye and Guo2022) used bacteria (Bacillus subtilis and Bacillus licheniformis) and microalgae (Chlorella vulgaris) to bioremediate aquaculture wastes, and these organisms, in turn, became foods for the filtering triangle sail mussel (Hyriopsis cumingii). In addition, Bacillus species enhanced the digestive enzymes activities of the mussel. Gao et al. (Reference Gao, Gao, Liu, Cai, Zhang and Qi2018) reported that an efficient aerobic denitrifier Bacillus megaterium has a high capacity to remove toxic nitrite and improve water quality. John et al. (Reference John, Krishnapriya and Sankar2020) reported that ammonia, nitrite and nitrate concentrations in tilapia culture wastewater microbial consortium were significantly reduced by using microbial consortium of Bacillus cereus, Bacillus amyloliquefaciens and Pseudomonas stutzeri as bioremediators.
Phytoremediation using plants such as macrophytes and microalgae, for sequestering nutrients, is another form of bioremediation that is useful treatment to improve water quality aquaculture systems (Table 4). Tejido-Nuñez et al. (Reference Tejido-Nuñez, Aymerich, Sancho and Refardt2019) showed improved water quality when the aquaculture effluent was treated with C. vulgaris and Tetraselmis obliquus, indicating that the microalgae were effective in nutrient removal. Nie et al. (Reference Nie, Mubashar, Zhang, Qin and Zhang2020) suggested a few options for the integration of microalgae culture with the aquaculture system such as permeable floating photobioreactors, bacteria–microalgae consortia, mixotrophic microalgae cultivation and biofilm production. Bioflocculation of microalgae and bacteria can enhance nutrient removal and facilitate microalgae harvesting (Nguyen et al., Reference Nguyen, Le, Show, Nguyen, Tran, TNT and Lee2019a). Kumar et al. (Reference Kumar, Santhanam, Park and Kim2016) showed that agar–alginate algal blocks, known as immobilized marine microalgae biofilter systems, were effective for nutrient removal from aquaculture wastewater. Microalgae can be introduced not only in the biofiltration system but also as a component to utilize inorganic N and P for their enhanced growth, and the resulting biomass can be valorized as feed for other aquatic organisms (Milhazes-Cunha and Otero, Reference Milhazes-Cunha and Otero2017). Li et al. (Reference Li, Zhang, Duan and Wang2019) and Nguyen et al. (Reference Nguyen, Tran, Le, Phan, Show and Chia2019b) reported that C. vulgaris produced higher biomass with a significant decrease in total N, total P, BOD and COD when recycled aquaculture wastewater was used as the culture medium. Wang et al. (Reference Wang, Qi, Bo, Zhou, Yan, Wang and Cheng2021) showed that microalgae produced higher biomass and nutritional contents when cultured in fishery wastes. When cultured with bioremediation bacteria (binary microalgae culture), microalgae exhibited a high growth rate, enhanced bio-flocculation, high-value metabolites and high removal efficiencies of total organic carbon, ammonium nitrogen and total phosphorus (Rashid et al., Reference Rashid, Park and Selvaratnam2018; Luo et al., Reference Luo, Wu, Jiang, Yu, Liu, Min, Li and Ruan2019). An increased number of degrading bacteria causes the integration of microalgae bacteria more effective in the degradation of organic pollutants in aquaculture wastewater, which promotes fish health (Zhang et al., Reference Zhang, Yang, Lai, Li, Zhan, Zhang, Jiang and Shu2022b).
Biofloc technology
Bioflocs are aggregates of mixed biological communities consisting of bacteria, algae, fungi and zooplankton that function not only to degrade the organic matter, reduce contaminants and improve water quality, but also to form an important source of food and immunostimulants to the cultured organisms (Table 4). The microbial community enhances the nutrient recycling of metabolites through in situ bioremediation, generating nutrients for the development of microalgae and zooplankton which serve as natural foods, and maintains the water quality in the system (Chen et al., Reference Chen, Kim, Thatcher, Hamilton, Alva, Zhou (George) and Brown2023). In the biofloc technology (BFT), bacterial communities dominated by heterotrophic bacteria can be developed in aquaculture systems using appropriate carbon sources in suitable C:N ratios (Gaona et al., Reference Gaona, da Paz Serra, Furtado, Poersch and Wasielesky2016). Ríos et al. (Reference Ríos, Monteagudo, Barrios, González, Vaillant, Bossier and Arenal2023) reported that C:N ratio of 10 significantly enhanced the immune stimulation in shrimp. Heterotrophic bacteria use organic carbon such as starch and sugar to generate energy and to grow into micro-biomass. Putra et al. (Reference Putra, Effendi, Lukistyowati, Tang, Fauzi, Suharman and Muchlisin2020) observed that molasses was the best biofloc starter for a tilapia culture system. Luo et al. (Reference Luo, Zhang, Cai, Tan and Liu2017) suggested the use of external carbohydrates (poly-β-hydroxybutyric and polycaprolactone) to improve the bacterial community, nitrogen dynamic and biofloc quality in tilapia (O. niloticus) culture system. Kim et al. (Reference Kim, Song, Rajeev, Kim, Kang, Jang and Cho2022b) reported that environmentally friendly microbial groups in a biofloc system of Pacific white shrimp, Litopenaeus vannamei, include Rhodobacteraceae, Flavobacteriaceae and Actinobacteria. In general, in BFT, heterotrophs were better compared to autotrophic bacteria for the treatment of the wastewater (Kim et al. Reference Kim, Hur, Kim, Jung and Han2020).
Physical–chemical methods
Physical and chemical methods such as filtrations, coagulation, flocculation and adsorption function to remove contaminants from the aquaculture wastewater, while electrochemical oxidation breakdown persistent organic compounds and aeration increased the dissolved oxygen in the water (Santos et al., Reference Santos, Ortiz-Gándara, Del Castillo, Arruti, Gómez, Ibáñez, Urtiaga and Ortiz2022). These methods can be applied singly or in combination in various aquaculture systems to further increase the efficiency of water quality improvement and enhance aquaculture production (Table 4). Biofilters (media with attached microorganisms such as bacteria, fungi, algae and protozoans) and membrane filters remove contaminants as the wastewater flows through them (Ng et al., Reference Ng, Ng, Mahmoudi, Ong and Mohammad2018; Hassan et al., Reference Hassan, Rashid, Muhaimeed, Madlul, Al-Katib and Sulaiman2022; Jin et al., Reference Jin, Sun, Ren and Huang2023). Coagulation (clumping of particles), flocculation (settling of coagulated materials) and adsorption (adhering of substances) can effectively remove suspended and dissolved solids from the aquaculture wastewater (Letelier-Gordo and Fernandes, Reference Letelier-Gordo and Fernandes2021; Igwegbe et al., Reference Igwegbe, Ovuoraye, Białowiec, Okpala, Onukwuli and Dehghani2022). Yanuhar et al. (Reference Yanuhar, Musa, Evanuarini, Wuragil and Permata2022) reported that water quality in concrete ponds can be improved by aeration, filtration and reduction of organic matter by optimizing the feed. Different types of biofiltration, biocoagulation, bioflocculation and biological interactions can be selected to enhance wastewater treatment and performance in aquaculture systems depending on their functionality and costs (Table 4).
Santos et al. (Reference Santos, Ortiz-Gándara, Del Castillo, Arruti, Gómez, Ibáñez, Urtiaga and Ortiz2022) introduced electrochemical oxidation as an alternative to biofiltration in RAS and reported several advantages including the decrease of toxic compounds and harmful by-products, water disinfection, reduced water use, easy adaptation to different production scales and an increase in fish health and yields. In addition, aquaculture effluents can be treated by coagulation of phosphorus and organic matter using FeCl3 and AlSO4 (Letelier-Gordo and Fernandes, Reference Letelier-Gordo and Fernandes2021). Kujala et al. (Reference Kujala, Pulkkinen and Vielma2020) and Lindholm-Lehto et al. (Reference Lindholm-Lehto, Pulkkinen, Kiuru, Koskela and Vielma2020) used a woodchip reactor, organic flocculants and slow sand filtration to efficiently remove nitrogen, phosphorus, geosmin and heavy metal, from rainbow trout (Oncorhynchus mykiss) culture.
IoT technologies and models
Traditionally, water quality monitoring in aquaculture systems needs manual sampling that requires a lot of time and cost. With the advent of technologies, real-time monitoring and early warning systems based on the IoT and intelligent monitoring system (IMS) can be designed and developed to make water quality monitoring and management more efficient and effective. IoT, consisting of collective network of communication devices integrated with artificial intelligence (AI) and modeling, can improve the monitoring and management of essential water quality parameters such as dissolved oxygen, pH values, turbidity and temperature in an aquaculture system (Figure 5). Wireless sensor network has been used widely for water quality monitoring (Shi et al., Reference Shi, Sreeram, Zhao, Duan and Jiang2018; Wei et al., Reference Wei, Tindik, Fui, Haviluddin and Hijazi2023). Rana et al. (Reference Rana, Rahman, Dabrowski, Arnold, McCulloch and Pais2021) used the machine learning approach to assess the influence of water quality parameters on the growth performance of freshwater aquaculture. Rahman et al. (Reference Rahman, Xi, Dabrowski, McCulloch, Arnold, Rana, George and Adcock2021) developed an integrated framework for aquaculture prawn farm management using sensors, machine learning and augmented reality-based visualization methods through real-time interactive interfaces. Thus, models for accurate predictions of water quality parameters, such as the hybrid prediction model (Eze et al., Reference Eze, Halse and Ajmal2021; Ranjan et al., Reference Ranjan, Tsukuda and Good2023) and fuzzy comprehensive evaluation method (You et al., Reference You, Xu, Su, Zhang, Pan, Hou, Li and Ding2021), can be developed for improved water quality management. Caballero and Navarro (Reference Caballero and Navarro2021) and Oiry and Barillé (Reference Oiry and Barillé2021) used the sentinel-2 satellite to monitor water quality, cyanoHAB and microphytobenthos. Xiang et al. (Reference Xiang, Cui, Li, Zhang, Mu, Liu and Zhao2023) used satellite remote sensing to monitor water color and water transparency, in relation to land-based activities that cause water turbidity and an increase of pollutants in aquatic ecosystems.
Precision feeding with minimal food waste is essential to maintain good water quality in aquaculture systems since excess feed is one of the major reasons for water quality deterioration in aquaculture systems. Fiordelmondo et al. (Reference Fiordelmondo, Magi, Mariotti, Bakiu and Roncarati2020) reported that feeding type and management could improve water quality in rainbow trout farming. Liu et al. (Reference Liu, Du, Zhang, Luo, Sha and Wang2023b) developed a precision feeding system on a software platform by integrating feeding management, a water quality monitoring system, a fish feeding activity sensor and an automatic feeding machine on a software platform. For convenience, efficiency and precision, Wu et al. (Reference Wu, Duan, Wei, An and Liu2022) applied intelligent and unmanned equipment for water quality management, underwater inspection, precision feeding and biomass estimation in deep-sea aquaculture. Ubina and Cheng (Reference Ubina and Cheng2022) noted unmanned systems are necessary for locations that are difficult to access due to risks associated with extreme climate and long distances from the shore.
The IoT can be used to develop automatic fish feeding with precise amounts and timing. Gao et al. (Reference Gao, Xiao and Chen2019) developed IoT-based intelligent fish farming system that includes a forecasting method for water quality management. The overall framework and constructs of the IoT and IMS-based aquaculture environment should integrate the control circuit, information collection, culture observation, data transmission and early warning system. IoT in aquaculture water quality monitoring involved the development of a cloud-based dashboard for data acquisition. Several cameras installed in the aquaculture farm are used to upload information wirelessly to the dashboard. Water quality parameters such as temperature, pH, conductivity, salinity, turbidity, dissolved oxygen and light intensity can be downloaded from a wireless sensing module. Islam et al. (Reference Islam, Pal, Chowdhuri, Salam, Islam, Rahman, Zahid and Idris2021) proposed a cost-effective long-range multistep predictor to improve the forecasting for water quality monitoring. Sampaio et al. (Reference Sampaio, Araújo, Dallago, Stech, Lorenzzetti, Alcântara, Losekann, Marin, Leao and Bueno2021) used low-to-high frequency data for water quality monitoring and fish production.
Bai et al. (Reference Bai, Fu, Li, Stankovski, Zhang and Li2021) proposed a risk assessment approach using bio-reaction kinetic models to evaluate pollutant accumulation in fish tissue as the index for environmental quality and safety in aquaculture. Various models for predicting and managing HABs have been established to reduce the impacts of algal toxins and water quality deterioration associated with eutrophication in aquaculture (Derot et al., Reference Derot, Yajima and Jacquet2020). Water quality modeling can also be based on disease agents. Jampani et al. (Reference Jampani, Gothwal, Mateo-Sagasta and Langan2022) suggested a water quality modeling framework to model and evaluate AR bacteria and AR genes in aquaculture systems.
AI techniques are useful and convenient for water quality management in aquaculture operations that are subjected to harsh environments and extreme climate such as offshore cage aquaculture. Chang et al. (Reference Chang, Wang, Wu, Hsieh, Wu, Cheng, Chang, Juang, Liou, Hsu, Huang, Huang, Lin, Peng, Huang, Jhang, Liou and Lin2021) developed an AI-IoT smart cage culture management system to solve problems related to physical inaccessibility to large coastal and off-shore aquaculture operations. In fact, intelligent and unmanned equipment provide convenient and efficient applications for water quality management, precision feeding and biomass estimation in aquaculture (Wu et al., Reference Wu, Duan, Wei, An and Liu2022). AI-IoT methods supported by sensors, wireless networks, automation and cloud data approaches are also applied for water quality monitoring in coastal waters, estuaries and land-based aquaculture systems (Danh et al., Reference Danh, Dung, Danh and Ngon2020; Huan et al., Reference Huan, Li, Wu and Cao2020; Pasika and Gandla, Reference Pasika and Gandla2020).
Policy and regulation
Policies and regulations are important in ensuring the implementation of aquaculture effluent management strategies as rapid expansion in the aquaculture industry not only provides economic opportunities but also presents risks to the environment and human society. In their assessment of sustainable global aquaculture Davies et al. (Reference Davies, Carranza, Froehlich, Gentry, Kareiva and Halpern2019) noted that many countries with active aquaculture sectors have some level of governance but lack clear frameworks for sustainable aquaculture development. Bohnes et al. (Reference Bohnes, Hauschild, Schlundt, Nielsen and Laurent2022) proposed a stepwise framework to assess the environmental impacts of aquaculture industries taking into account the existing national policy coupled with economic equilibrium models and life cycle assessment of aquaculture activities, especially those related to aquaculture feed production and usage.
Aquaculture farmers in many countries in Asia, where 90% of aquaculture activities are located, have difficulties in adopting environmental governance due to their small farms with limited physical and financial resources. For large farms, access to global markets via certification could be the major driver for adopting environmental governance. Quyen et al. (Reference Quyen, Hien, Khoi, Yagi and Karia Lerøy Riple2020) reported that Vietnamese shrimp farmers followed specific certification guidelines and conducted good aquaculture practices to produce quality and safe products as required by the importing countries, avoiding rejections and economic losses. However, most aquaculture smallholders are experiencing environmental and water quality problems that extend beyond the boundary of their farms. To mitigate environmental risk due to non-sustainable aquaculture practices, Bush et al. (Reference Bush, Oosterveer, Bottema, Meuwissen, de Mey, Chamsai, Ho and Chadag2019) suggested implementing environmental governance for water quality management such as certification, finance and insurance on a wider landscape instead of focusing on each farm. Bohnes et al. (Reference Bohnes, Hauschild, Schlundt, Nielsen and Laurent2022) proposed a stepwise framework to assess the environmental impacts of aquaculture industries taking into account the existing national policy coupled with economic equilibrium models and life cycle assessment of aquaculture activities, especially those related to aquaculture feed production and usage. Wood et al. (Reference Wood, Capuzzo, Kirby, Mooney-McAuley and Kerrison2017) also showed that a small farm on its own is unlikely to have a significant effect on water quality and environmental conservation compared to a very large farm or a conglomerate of small farms. Thus, environmental policies and regulations that consider all elements of farm-to-market operation including production systems (cost-effectiveness and sustainable supply); water quality (sources and effluents); ecosystem health (ecosystem services) and socioeconomics (human health, economy and livelihoods) are needed to make the aquaculture industry a viable food producer.
Conclusions
Water quality is one of the critical factors to be considered in aquaculture as it has significant effects on fish growth, health and yields. A lack of knowledge and practices in water quality management could severely impede the growth of the aquaculture sector and jeopardize the utilization of the available water resources for a sustainable aquaculture industry.
Aquaculture requires a significant understanding of the factors and problems affecting production systems, in addition to improvements of approaches and technologies in water quality management. Water quality enhancement in production systems such as RAS, IMTA and aquaponics through efficient integration with physical, chemical and biological factors would boost the FCR and improve the health of cultured animals. The recycling of nutrients using different organisms along the aquatic food chain, such as bacteria, microalgae, seaweeds and fish, can enhance the growth, survival and production of the cultured species as well as accumulate the biomass of the supporting organisms. In addition, microalgae-based technologies are a promising solution for aquaculture wastewater treatment and the resulting microalgal biomass can be valorized. The use of these technologies in the forms of biofloc, bioremediation, coagulation-flocculation-biofiltration technologies and various ecosystem-based approaches provide options for aquaculture best practices that could improve water quality, resulting in improved aquaculture production.
The application of AI and IoT in aquaculture production systems supported by sensors, wireless transmission systems, unmanned equipment, automation and big data would enable intelligent water quality monitoring, precision feeding systems, fish activity monitoring and early problem detection. The integration of smart production systems and advanced processes would result in precision feeding, improved water quality, increased survival rates and increased growth of the cultured species. Overall, the use of these technologies in water quality management supported by relevant policy and regulation would facilitate the approach to sustainable aquaculture production via effective management of the environment and fish health.
Open peer review
To view the open peer review materials for this article, please visit http://doi.org/10.1017/wat.2024.6.
Author contribution
F.M.Y.: Conceptualization, writing the original draft, graphics, reviewing and editing. U.W.A.D.: Reviewing, editing and graphics, N.M.R.: Reviewing, editing and graphics. R.H.: Reviewing and editing.
Competing interest
The authors declare no competing interest exists.












Comments
Dear Dr. Elaine Halls
Editorial Office, Cambridge Prisms: Water
Thank you very much for inviting me to contribute a manuscript to the `Water‘. Thanks also for giving me many months to complete this manuscript. My sincere apologies for the long delay. I have uploaded an article on `Water Quality Management in Aquaculture’. Hope it meets your requirements for the journal. Since I am behind time in submitting this manuscript, I haven’t uploaded the graphics abstract. yet. I will submit it upon acceptance of the manuscript. Please send me additional forms that I need to submit.
Best regards
Fatimah