INTRODUCTION
Toxoplasmosis caused by the protozoan Toxoplasma gondii (Apicomplexa) is one of the most common parasitic infections worldwide. Apart from humans, it afflicts various mammals and birds, with felids playing the major role as definitive hosts of the parasite. Humans are infected either by consumption of undercooked meat containing a tissue cyst or during ingestion of oocysts shed by the definitive host in faeces. In women, infection during pregnancy can result in vertical transmission of T. gondii infection [Reference Montoya and Liesenfeld1, Reference Bachmeyer2]. The primary infection in immunocompetent individuals is typically asymptomatic, or accompanied with only mild and non-specific symptoms, and is usually followed by a lifelong latent infection. However, from this state of latency T. gondii infection may be reactivated as a result of immune disorders [Reference Israelski and Remington3, Reference Meers4]. This makes toxoplasmosis one of the major opportunistic infections afflicting patients with advanced HIV infection. The most typical form of this infection in these patients is a potentially life-threatening toxoplasmic encephalitis (TE), whereas extracerebral localization – in eyes, lung, bone marrow, heart or other organs is much less frequent [Reference Israelski5, Reference Rabaud6]. Cases of reactivated T. gondii infection in non-HIV individuals are rarely reported, with the possible exception of Toxoplasma-seropositive patients following allogenic stem cell transplantations, as in the reported incidence of 9·3% reactivation [Reference Meers4, Reference Sugane7]. The critical level of immunity is based on the number of CD4+ T lymphocytes (CD4 < 100/μl). With the introduction of effective prophylaxis for toxoplasmosis (trimethoprim-sulfamethoxazole 160/800 mg p.o. q.i.d. as primary prophylaxis in patients with CD4 < 100/μl) in the early 1990s and combination antiretroviral therapy (cART) the number of cases sharply decreased thereafter, some studies actually quoting zero cases, and it appeared that toxoplasmosis would no longer pose a threat to patients with HIV infection [Reference Girard8–Reference Mateen10]. In reality, TE still occurs and this has prompted our current research into Toxoplasma reactivation.
METHODS
The cohort consisted of all HIV-infected patients attending the AIDS Centre at Bulovka Hospital in Prague, Czech Republic, between November 1988 and February 2013. Blood samples were collected from all patients at 3- to 6-month intervals for T. gondii serology as well as immunological, haematological and biochemical tests.
Throughout this study complement-fixation test (CFT), IgG ELISA and IgM ELISA were used for detection of toxoplasmic antibodies. Positive results for CFT were defined as a titre of ⩾1:8, for IgG ELISA at absorbance of ⩾0·25, and IgM ELISA was positive at a positivity index (absorbance of tested sample/absorbance of cut-off control) of >1·1 [Reference Montoya11]. The Toxoplasma status of patients whose test results fluctuated during follow-up was considered negative when the initial sample was negative and no more than one positive result was detected thereafter. A single isolated seropositivity with very low antibody levels was interpreted as a diagnostic error or non-specific or false-positive reaction. Toxoplasma status was considered positive when the initial sample was positive and repeated (two or more) positive samples were detected.
The other monitored laboratory tests included the HIV RNA viral load measured by polymerase chain reaction (PCR; limit of detection 20 copies/ml), parameters of both humoral immunity – serum immunoglobulins IgG (normal range 7·51–15·6 g/l), IgM (normal range 0·46–3·04 g/l) and IgA (normal range 0·82–4·53 g/l) and β-2 microglobulin (normal range 0·7–1·8 mg/l) levels, and cellular immunity such as number of CD4+ T lymphocytes (CD4; normal range 700–1100/μl), CD8+ T lymphocytes (CD8; normal range 500–900/μl) and natural killer cells (NK; normal range 300–700/μl) tested by flow cytometry.
Diagnosis of reactivation of T. gondii infection was assumed in patients with focal symptoms of CNS damage and typical CNS imaging (multiple ring enhancing lesions on CT or MRI). The diagnosis was then confirmed either by successful therapeutic intervention or on the basis of positive autopsy findings in patients who did not survive the episode of TE. Evaluation of liquor T. gondii PCR was not included into the study because lumbar puncture was not performed in the majority of patients with TE.
In all patients with suspected reactivation of T. gondii infection therapy commenced with sulfadiazine 1·5 g p.o. q6 h + pyrimethamine 75–100 mg p.o. q.i.d. (with an initial loading dose of 200 mg pyrimethamine) and folinic acid for a minimum of 6 weeks [Reference Bertschy12].
Ethical statement
The study was approved by the local Ethics Committee of Bulovka Hospital and was conducted in accordance with the ethical standards laid down in the 1975 Declaration of Helsinki, as revised in 2008. All patients agreed to participate in the study and signed an informed consent.
Statistical analysis
Geometric means, together with corresponding 95% confidence intervals (CI) were calculated to characterize the location and variability of the analysed variables in the groups. Patients with reactivated T. gondii infection were compared to the reference group consisting of all Toxoplasma-seropositive individuals without reactivation. The comparison of means between groups was based on the Student's two-sample t test applied to logarithmically transformed data. The incidence rate of Toxoplasma reactivation in HIV-infected patients was calculated from the total follow-up time of Toxoplasma-seropositive persons and the number of reactivation cases. Tests of categorical variables were based on Fisher's exact test. For CD4, CD8 and NK cell markers, quartiles were calculated for Toxoplasma-seropositive patients for whom data on all three factors was available, and according to these quartiles the patients were divided into four groups. All statistical tests were evaluated as two-sided at a significance level of 0·05. Stata, release 9·2 (StataCorp LP, USA) statistical software was used for statistical analysis.
RESULTS
During the whole period of the study 1235 HIV-infected individuals were enrolled. Serological tests revealed that 502 patients were infected with T. gondii prior to diagnosis of HIV infection (425 males, 77 females). The prevalence of positive Toxoplasma serology was 40·2% in HIV-infected men and 43·5% in HIV-infected women. The remaining 623 males and 96 females showed no serological evidence of T. gondii infection. In 14 originally Toxoplasma-seronegative patients (ten men, four women) seroconversion showing recent T. gondii infection was observed during follow-up. Clinical signs of primary T. gondii infection were observed only in one patient – a pregnant woman with transient cervical lymphadenopathy. The median follow-up period of repeatedly tested patients with HIV/T. gondii co-infection was 3·4 years (based on 2–47 samples), maximum follow-up was 23·5 years.
Total person-time of follow-up of HIV/T. gondii co-infected patients was 2909·3 person-years. A total of 298·5 person-years were recorded in the period before 1988–1996 (before the introduction of cART in the Czech Republic) and 2610·8 person-years in the era of cART (1997–2013).
Reactivation of T. gondii infection in the form of TE was diagnosed in 21 (4·2%) patients (19 men; i.e. 4·5% of all men in the cohort) and two women (2·6% of all women). In the first period TE was diagnosed in 12 cases and in the period after 1996 in nine patients. The age range of patients was 24·7–61·2 years (mean 40·2 years). Regarding HIV transmission, 13 patients were homosexual/bisexual men, four heterosexual individuals (two men, two women), three intravenous drug users and one blood recipient. Subsequent Toxoplasma reactivation ranged from 0 to 9·2 years (mean 1·8 years) after diagnosis of HIV infection.
A total of 12 (57·1%) cases (five in the first and seven in the second period) were so-called ‘late presenters’ in whom HIV testing was initiated by symptoms of TE, and thus both diagnoses were made simultaneously. In 18 (85·7%) patients TE was the first opportunistic infection indicating progression to the clinical category C (AIDS). Only in three cases was TE not the first AIDS-defining illness and it occurred 5–16 months after progression to AIDS. In these three patients the first indicative opportunistic infections comprised either oesophageal candidiasis, Salmonella septicaemia or pneumocystis pneumonia. None of the patients with TE was taking anti-Toxoplasma prophylaxis. Out of the entire cohort, there were 16·7% patients on trimethoprim-sulfamethoxazole prophylaxis. The median duration of prophylaxis was 19 months.
All cases of TE involved clinical signs of focal neurological lesions and bore a typical finding under CT and MRI examination. Following preliminary diagnosis of TE all patients underwent therapy involving a combination of sulfadiazine + pyrimethamine and folinic acid for a minimum of 6 weeks, as described above.
Seven out of 12 patients who developed TE in the first period died 8–34 days after initiation of therapy. The definitive diagnosis of TE was confirmed in all deceased patients by histological findings of necrotic lesions in brain tissue and the presence of T. gondii tachyzoites. A further four patients who survived TE died later of other causes and the autopsies failed to detect any signs of active T. gondii infection. One patient from this subgroup is currently alive. Likewise, autopsies of a further 146 deceased HIV-positive patients from our cohort showed no signs of active T. gondii infection whatsoever. All nine patients who suffered from TE in the second period were successfully treated with no fatal outcomes.
The resulting incidence rate of Toxoplasma reactivation in our cohort was 40·2/1000 person-years (95% CI 22·8–70·8) for the first period and 3·4/1000 person-years (95% CI 1·8–6·6) for the second period. Thus the incidence of reactivation during the second monitored period was 11·8 times lower.
Regarding the immune status of Toxoplasma-seropositive patients the available CD4, CD8 and NK cell values in reactivated patients were in the lowest quartile category, i.e. below the 25th percentile (CD4 < 33·8/μl, CD8 < 819·7/μl, NK < 189·0/μl), with the exception of three patients with higher CD8 counts. Out of 21 cases with reactivation of T. gondii infection, seven patients had all three, and 12 had two lymphocyte subsets in the lowest quartile. For the remaining two reactivated patients only CD4 counts are available, these are also in the lowest category.
In the whole cohort, 26 patients had all three values of lymphocyte subsets under the 25th percentile, approximately one quarter of whom were reactivated. A characteristic feature was an extremely low CD4 and NK cell count at the time of reactivation. Although there were relatively great differences in individual patients, the cell counts were always lower than the normal values. The latent T. gondii infection reactivated only in patients with CD4 counts < 300/μl. Moreover in 13 (61·9%) cases CD4 counts were < 50/μl. As shown in Table 1, low nadir CD4 counts correlate with risk of Toxoplasma reactivation. The highest proportion of reactivations was found in the group with < 50 CD4/μl. In patients who had T. gondii reactivation, the mean CD4 count was 12·6 times lower than in the group without reactivation. Similarly NK cell counts were 4·3 times lower in patients with reactivation than in the reference group without reactivation (see Table 2).
Table 1. Risk of Toxoplasma reactivation related to the nadir of CD4+ T lymphocyte counts in HIV/T. gondii co-infected patients
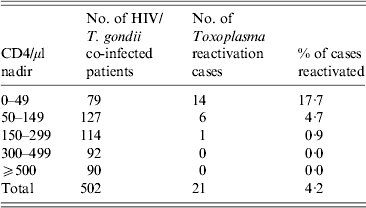
Table 2. Immunological and virological parameters of HIV-infected patients with reactivation of T. gondii infection compared to background values of the reference group of HIV-infected patients with non-reactivated T. gondii infection
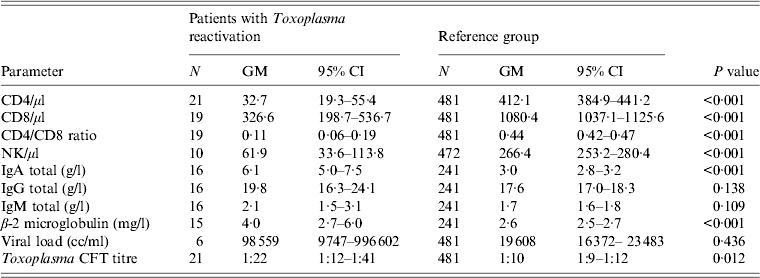
GM, Geometric mean; CI, confidence interval.
Similarly, mean CD8 counts were significantly lower (3·3 times) than in the reference group although in 19 cases of reactivation for which data are available these counts were within the normal range or even higher than normal values in seven cases (36·8%).
Conversely, mean levels of total IgA and β-2 microglobulin were found to be significantly higher (IgA twofold, β-2 microglobulin 1·5 times) during reactivation than in the control group. For IgG and IgM the difference between groups was not statistically significant.
The humoral response during Toxoplasma reactivation revealed no characteristic features. Low CFT titres and IgG levels prevailed; nevertheless, in seven (33·3%) cases, titres of ⩾1:64 accompanied with medium IgG levels were recorded. The geometric mean of titres was 1:22, which is, however, significantly higher than the mean value in HIV-infected patients with latent T. gondii infection (P = 0·012). IgM antibodies were tested in 17 patients, with a positive result in only one case (5·9%). A single patient out of 21 (4·8%) TE cases showed completely negative results in six consecutive examinations for anti-Toxoplasma antibodies. In this patient the diagnosis of TE was verified post-mortem histologically.
DISCUSSION
Literature sources reveal a certain amount of data pertaining to the incidence of reactivated T. gondii infection, particularly TE in HIV-infected patients. The bulk of this data was collected prior to the introduction of cART, and quoted incidence rates are often very high: for instance, in France (1983–1994) an overall incidence of 205/1000 patient-years [Reference Bossi13], and in the USA 15 cases/1000 HIV-infected person-years [Reference Jones14]. In Denmark an overall cumulative incidence of 8% in AIDS patients was reported [Reference Smith15], while in France Derouin et al. [Reference Derouin, Thulliez and Garin16] registered an annual reactivation rate of 12% in HIV/T. gondii co-infected patients, and according to Israelski et al. 38% of Toxoplasma-seropositive AIDS patients developed TE [Reference Israelski5]. However, comparison of these results is problematic because of the diverse methodology in these studies. Some studies lack information on important parameters, such as Toxoplasma status in persons involved or at least seroprevalence in the observed cohorts.
The calculated incidence of 40 cases of reactivation per 1000 Toxoplasma-seropositive HIV-infected patients per year, as quoted in this Czech study in the period prior to 1996, evidently reflects the lower limits of this broad spectrum. During the monitored period reactivation of T. gondii infection was observed in 4·5% of male and 2·6% of female patients. On the other hand, if the figures are applied specifically to the category of patients with most severe immunodeficiency (CD4 < 50/μl), the reactivation rate is 17·7%. Conversely, application of the same data to all patients with AIDS would be less marked because immunological parameters in some cases improved in the course of treatment with no subsequent reactivation.
After 1996, the efficacy of antiretroviral therapy improved with the introduction of cART. In HIV-infected patients cART restores cell-mediated immunity including recovery of anti-Toxoplasma-specific CD4 and CD8 responses [Reference Lejeune17]. Hence, with the introduction of cART a number of studies registered a decline in the incidence of reactivated T. gondii infection. At the Johns Hopkins HIV Clinic, Baltimore, USA, in predominantly intravenous drug users, the incidence rate for toxoplasmosis decreased from 12/1000 person-years in 1994 to five in 2000 [Reference Moore and Chaisson18, Reference Sacktor19]. Analogically, in the Multicenter AIDS Cohort Study of gay/bisexual men in the USA, there was a trend for a decreased mean incidence rates from 5·4/1000 person-years in 1990–1992 to 2·2 in 1996–1998 and no cases of TE were observed in this study after 1998 [Reference Mateen10]. In a German study on HIV-infected patients, the rates dropped from 5·7% in 1995–1996 to 2·2% in 1997–8 [Reference Maschke20]. In Italy, patients who had undergone cART were 5% less likely to develop TE [Reference Antinori21]. The results of these studies are, however, limited by the fact that they include Toxoplasma-seronegative subjects who are at no risk of reactivation whatsoever; additionally, even the prevalence of T. gondii infection in the monitored groups is not quoted. Due to the fact that the Toxoplasma status of all subjects is evident in our study we can safely eliminate any bias in this respect.
Our results, based on long-term monitoring of HIV/T. gondii co-infected patients, show that the incidence of reactivated T. gondii infection in the Czech Republic has declined twelvefold since the introduction of cART. Nonetheless, in certain regions a high prevalence continues to be reported. In a recent Brazilian cross-sectional study with 80% T. gondii seroprevalence TE was observed in 5·6% of HIV-infected patients [Reference Xavier22].
None of the patients in our cohort were treated by cART prior to T. gondii reactivation or after 1996 either because they were diagnosed with HIV after the first symptoms of TE or because they refused to undergo treatment. Thus, our results do not support the conclusions of the Italian study that failure of antiretroviral treatment is the main determinant when TE occurs in patients receiving cART [Reference Antinori21].
It is well known that T. gondii infection reactivates only in patients with significantly depleted CD4 counts, which are frequently presented as major (and often the only) parameter of host cell immunity in HIV-infected patients [Reference Israelski5]. In this study, a mean CD4 of 33/μl (95% CI 19–55) was recorded, similarly to other studies that quote a mean CD4 of 32/μl (95% CI 22–42) [Reference Happe23], 44/μl [Reference Bossi13] or 69/μl (95% CI 58–80) [Reference Antinori21].
Compared to HIV/T. gondii co-infected subjects, patients with reactivated T. gondii infection had a mean CD4 count 14 times lower. Nevertheless, even a major depletion of CD4 did not necessarily cause reactivation: in the category of patients with CD4 <50 reactivation occurred in only 17·7% of cases. Evidently, although CD4 plays a key role in immunity against T. gondii as an important producer of interferon gamma (IFN-γ), it is not the sole determinant for reactivation [Reference Denkers and Gazzinelli24]. Our results demonstrate that Toxoplasma reactivation occurs in HIV/T. gondii co-infected patients with not only the lowest CD4 but also CD8 and NK cell levels. This is in accordance with recent studies documenting a crucial role of CD8 in the control of parasite reactivation during HIV infection [Reference Bhadra and Khan25].
In contrast to CD4 investigations, studies of reactivated T. gondii infections in immunocompromised patients do not, as a rule, present CD8 counts, which are of crucial importance in this subset in terms of long-term immunity against T. gondii infection. The role of CD8-derived IFN-γ during the acute phase of infection is to reduce parasite numbers and then, during the chronic phase, to exert sufficient immune pressure to maintain the parasite encysted as a bradyzoite [Reference Gigley, Bhadra and Khan26]. In a mouse model, CD8 have a protective effect in the initial stages of reactivation of T. gondii infection [Reference Khan27]. Specific CD8 play a synergistic role with CD4 in IFN-γ production and protective immunity to T. gondii infection [Reference Gazzinelli28].
NK cells also play an important role in the innate immune response to T. gondii infection through cytotoxicity and production of cytokines, particularly IFN-γ [Reference Vivier29]. In immunocompromised laboratory mice, a potent NK cell response could compensate for the lack of T cells during the immune response against T. gondii [Reference Denkers30, Reference Gazzinelli31].
Similarly to CD8, NK cells are often not investigated in studies of reactivated T. gondii infection. The results in our study showing significantly reduced NK count in patients with Toxoplasma reactivation emphasize their significance in the course of AIDS.
Elevated immunoglobulin levels, in particular IgA, are fairly frequently observed in patients with severe immunodeficiency. However, it is not clear if there is a causal relationship with T. gondii infection or if it is only a result of the dysregulation of the immune system caused by HIV. An analogous interpretation could be ascribed to the 1·5-fold elevation of β-2 microglobulin values in reactivated patients, although in some studies increased serum β-2 microglobulin is mentioned as a reliable marker of fetal T. gondii infection [Reference Dreux32, Reference Nesovic-Ostojic33]. In contrast, our findings did not confirm elevated levels in HIV-infected adults recently infected by T. gondii [Reference Machala34].
Whereas HIV-infected individuals with latent T. gondii infection are at risk of reactivation, seronegative patients may become infected, which would manifest as seroconversion. The incidence rate of reactivation in HIV/T. gondii co-infected patients detected in the period prior to the introduction of cART (40·2/1000 person-years) was approximately nine times higher compared to the incidence rate of new T. gondii infections in HIV-infected and Toxoplasma-seronegative persons (4·6/1000 person-years).
In contrast to reactivation, primary T. gondii infection is not associated with severe immunosuppression; the mean count of CD4 (501/μl, 95% CI 363–691) was in fact higher than in controls. It is not, as a rule, associated with severe clinical symptoms [Reference Machala34].
In patients with reactivated T. gondii infection, this study detected neither significantly elevated CFT titres compared to the reference group, nor (with a single exception) positive IgM antibodies by ELISA test. If these data are available prior to, and after, reactivation then the lack of a rising trend in antibody levels is evident.
As previously shown, the predictive value of elevated CFT titres and/or IgG ELISA absorbance is as low as 14% for reactivation of T. gondii infection. Due to gradual impairment of both cell-mediated and humoral responses as a consequence of progressing HIV infection, toxoplasmic antibody production generally tends to be weak in immunocompromised patients [Reference Machala35]. This explains why in various studies including the present study, 2–6% of patients with confirmed reactivated T. gondii infection were seronegative [Reference Bossi13, Reference Happe23]. We can assume that these patients were infected a very long time ago and belonged to a group of nearly one third of patients with a ‘declining’ antibody dynamic trend [Reference Machala35].
The presence of specific IgA in the absence of IgM is considered an important criterion for reactivation; however, IgA are not detected in all patients with reactivation [Reference Goncharov36]. On the contrary, IgM antibodies are found in isolated cases because they can persist in the serum of certain HIV-infected patients for long periods of time. Indeed, diagnostic practice has few cases where increasing titres of toxoplasmic antibodies in IgM negative patients, or IgA positivity with concurrent negative IgM, are interpreted as ‘reactivation of T. gondii infection’. As a rule, there are no means of confirming this diagnosis and reactivation has achieved an almost ‘mythical’ status which is discussed in various contexts and is subject to speculation. For instance, according to Brown et al. the anamnestic response resulting from reactivation of previous infection produces elevated IgG antibody titres which are associated with increased risk of schizophrenia [Reference Brown37]. However, observation of confirmed cases of reactivation of T. gondii infection in HIV-infected patients suggests that latent T. gondii infection may reactivate only under conditions of severe immunodeficiency and the incidence of reactivation in the general population is highly improbable; and because the elevation of toxoplasmic antibody levels is not a characteristic sign of reactivation, it would be impossible to diagnose by serology even in the event that it had, in fact, occurred.
In conclusion, we state that in view of the fact that Toxoplasma status is continually monitored in our cohort of HIV positive patients, it is possible to evaluate the incidence of reactivation of this opportunistic infection prior to, and post-cART therapy. Reactivation of this form of encephalitis still occurs, albeit at far lower counts than previously. The circumstances of such reactivation have likewise changed: at present, it is observed almost exclusively in so-called ‘late-presenters’ or patients who refuse treatment.
ACKNOWLEDGEMENTS
This study was supported by the Grant Agency of the Ministry of Health of the Czech Republic (IGA MZ CR, No. NT/11429-5) and grants from Charles University in Prague (PRVOUK/P24/LF1/3 and SVV-2014-260026). The authors are grateful to Blanka Sirocka and Jarmila Sedlakova for excellent technical assistance.
DECLARATION OF INTEREST
None.





