Crossref Citations
This article has been cited by the following publications. This list is generated based on data provided by
Crossref.
Kawamura, Satoru
1995.
Unsolved issues in S-modulin/recoverin study.
Behavioral and Brain Sciences,
Vol. 18,
Issue. 3,
p.
479.
Brown, R. Lane
and
Karpen, Jeffrey W.
1995.
Molecular insights gained from covalently tethering cGMP to the ligand-binding sites of retinal rod cGMP-gated channels.
Behavioral and Brain Sciences,
Vol. 18,
Issue. 3,
p.
471.
McGinnis, James F.
1995.
Gene therapy, regulatory mechanisms, and protein function in vision.
Behavioral and Brain Sciences,
Vol. 18,
Issue. 3,
p.
481.
Smith, Steven O.
1995.
Nuclear magnetic resonance studies on the structure and function of rhodopsin.
Behavioral and Brain Sciences,
Vol. 18,
Issue. 3,
p.
488.
Roberson, Erik D.
and
Sweatt, J. David
1995.
Regulation of adenylyl cyclase in LTP.
Behavioral and Brain Sciences,
Vol. 18,
Issue. 3,
p.
485.
Kaplan, Michael W.
1995.
Linking genotypes with phenotypes in human retinal degenerations: Implications for future research and treatment.
Behavioral and Brain Sciences,
Vol. 18,
Issue. 3,
p.
478.
Crouch, Rosalie K.
and
Corson, D. Wesley
1995.
The structure of rhodopsin and mechanisms of visual adaptation.
Behavioral and Brain Sciences,
Vol. 18,
Issue. 3,
p.
472.
Molday, Robert S.
and
Hsu, Yi-Te
1995.
Further insight into the structural and regulatory properties of the cGMP-gated channel.
Behavioral and Brain Sciences,
Vol. 18,
Issue. 3,
p.
500.
Pepperberg, David R.
Hood, Donald C.
and
Birch, David G.
1995.
Light Adaptation and Post-Flash Recovery in Human Rods.
p.
MA3.
Schnetkamp, Paul P. M.
1995.
Na-Ca + K exchanger and Ca2+homeostasis in retinal rod outer segments: Inactivation of the Ca2+efflux mode and possible involvement of intracellular Ca2+stores in Ca2+homeostasis.
Behavioral and Brain Sciences,
Vol. 18,
Issue. 3,
p.
488.
Tamai, Makoto
1995.
Glutamate accumulation in the photoreceptor-presumed final common path of photoreceptor cell death.
Behavioral and Brain Sciences,
Vol. 18,
Issue. 3,
p.
490.
Wahlsten, Douglas
1995.
The genetic kaleidoscope of vision.
Behavioral and Brain Sciences,
Vol. 18,
Issue. 3,
p.
490.
Hurley, James B.
1995.
Recoverin, a calcium-binding protein in photoreceptors.
Behavioral and Brain Sciences,
Vol. 18,
Issue. 3,
p.
497.
Bownds, M. Deric
and
Arshavsky, Vadim Y.
1995.
How many light adaptation mechanisms are there?.
Behavioral and Brain Sciences,
Vol. 18,
Issue. 3,
p.
496.
Polans, Arthur S.
and
Adamus, Grazyna
1995.
Recoverin is the tumor antigen in cancerassociated retinopathy.
Behavioral and Brain Sciences,
Vol. 18,
Issue. 3,
p.
483.
Barnstable, Colin J.
1995.
Mechanisms of photoreceptor degenerations.
Behavioral and Brain Sciences,
Vol. 18,
Issue. 3,
p.
470.
Willardson, Barry M.
Yoshida, Tatsuro
and
Bitensky, Mark W.
1995.
Cyclic nucleotides as regulators of light-adaptation in photoreceptors.
Behavioral and Brain Sciences,
Vol. 18,
Issue. 3,
p.
493.
Sagoo, Mandeep S.
and
Lagnado, Leon
1995.
Modulation of the cGMP-gated channel by calcium.
Behavioral and Brain Sciences,
Vol. 18,
Issue. 3,
p.
486.
Matthews, H. R.
and
Fain, G. L.
1995.
Reduced cytoplasmic calcium concentration may be both necessary and sufficient for photoreceptor light adaptation.
Behavioral and Brain Sciences,
Vol. 18,
Issue. 3,
p.
481.
Garavito, R. Michael
1995.
The atomic structure of visual rhodopsin: How and when?.
Behavioral and Brain Sciences,
Vol. 18,
Issue. 3,
p.
474.
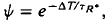 where ΔT is the decrease in saturation period, and where TR* is the slope of the line that relates T and ln If in a given state of adaptation. Dark- and light-adapted responses to flash intensities
where ΔT is the decrease in saturation period, and where TR* is the slope of the line that relates T and ln If in a given state of adaptation. Dark- and light-adapted responses to flash intensities 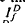 and
and 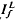 , respectively, exhibited similar absolute peak photocurrent and falling-phase kinetics when
, respectively, exhibited similar absolute peak photocurrent and falling-phase kinetics when 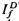 and
and 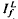 satisfied the relation,
satisfied the relation, 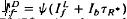 , where Ib is the background intensity. It is argued that ψ approximates the relative PDE*/R* gain of transduction, i.e. the relative peak level of activated cGMP phosphodiesterase (PDE*) produced by a given, small amount of photoactivated visual pigment (R*). Interpreted on this view, the results imply that light adaptation derives largely from a decrease in PDE*/R* gain, rather than from the stimulation of guanylate cyclase activity. The data are consistent with the possibility that modulation of the lifetime of PDE* underlies the background dependence of ψ.
, where Ib is the background intensity. It is argued that ψ approximates the relative PDE*/R* gain of transduction, i.e. the relative peak level of activated cGMP phosphodiesterase (PDE*) produced by a given, small amount of photoactivated visual pigment (R*). Interpreted on this view, the results imply that light adaptation derives largely from a decrease in PDE*/R* gain, rather than from the stimulation of guanylate cyclase activity. The data are consistent with the possibility that modulation of the lifetime of PDE* underlies the background dependence of ψ.