Introduction
In conservation biology, species restoration is playing an increasingly important role in counteracting ongoing biodiversity decline, assisting in the establishment of once-widespread species and restoring populations in areas where they have become extinct (IUCN/SSC, 2013; Destro et al., Reference Destro, De Marco and Terribile2018; Pettorelli et al., Reference Pettorelli, Barlow, Stephens, Durant, Connor and Schulte to Bühne2018). Reintroduction is a key restoration method defined by the IUCN as ‘the intentional movement and release of an organism inside its indigenous range from which it has disappeared’ (IUCN/SSC, 2013). Reintroductions often aim to minimize interventions to create viable populations with the least necessary human support (Corlett, Reference Corlett2016).
Reintroduction efforts have been undertaken for a wide and increasing range of species (Bennett et al., Reference Bennett, Doerr, Doerr, Manning, Lindenmayer and Yoon2013; Gray et al., Reference Gray, Crouthers, Ramesh, Vattakaven, Borah and Pasha2017; Soorae, Reference Soorae2018), with varying degrees of success. Failures are most often because of factors such as predation, human–wildlife interactions and habitat mismatch, all of which affect the viability and reproduction of the target species (Wimberger et al., Reference Wimberger, Downs and Perrin2009; Bennett et al., Reference Bennett, Doerr, Doerr, Manning, Lindenmayer and Yoon2013). There is no general definition of reintroduction success but calculating the population growth rate, population size and reproduction probability has been suggested as a useful approach to determining reintroduction success (Robert et al., Reference Robert, Colas, Guigon, Kerbiriou, Mihoub, Saint-Jalme and Sarrazin2015). This should be accompanied by determining the frequency of stochastic events (i.e. random catastrophic events such as adverse weather conditions that occur with a given probability and potentially affect demographic rates, and hence reduce the population size at the reintroduction site; Robert et al., Reference Robert, Colas, Guigon, Kerbiriou, Mihoub, Saint-Jalme and Sarrazin2015). Moreover, it is crucial not only to assess the long-term viability of reintroduced individuals under given constraints but also to evaluate and rank the impacts of management interventions retrospectively to help adjust management actions and compare alternative future interventions (Pereira & Navarro, Reference Pereira and Navarro2015; Robert et al., Reference Robert, Colas, Guigon, Kerbiriou, Mihoub, Saint-Jalme and Sarrazin2015).
Here we analyse long-term demographic data (2008–2019) of a reintroduced migratory European population of the northern bald ibis Geronticus eremita, named the Waldrappteam population. We also assess the long-term probability of persistence of the reintroduced population using an individual-based model in which quantification of the effects of the main management measures and of stochastic events on demographic rates during the study period provides the framework for the model. We assess the impacts of future alternative management measures and stochastic events as functions of an individual-based model. We hypothesize that at the present time the northern bald ibis population can survive without further management and release. We predict that the observed demographic rates will ensure population growth and do not differ between the breeding colonies.
Species and study area
The northern bald ibis was categorized as Critically Endangered on the IUCN Red List from 1994 and recategorized as Endangered in 2018 (BirdLife International, 2018). The species is migratory, mainly insectivorous and can live to an age of up to 30 years in captivity. Northern bald ibises are mainly seasonally monogamous and breed in colonies of up to hundreds of birds (Boehm et al., Reference Boehm, Bowden, Seddon, Hatipoglu, Oubrou and el Bekkay2020). Juveniles learn the migration route by following conspecifics to the wintering ground, where they usually remain until sexual maturity (Fritz et al., Reference Fritz, Unsoeld, Voelkl, Kaufman, Bashaw and Maple2019). The last remaining wild population of northern bald ibises has changed to a sedentary lifestyle and inhabits the Moroccan coast (Bowden et al., Reference Bowden, Smith, El Bekkay, Oubrou, Aghnaj and Jimenez-Armesto2008).
In 2002 European scientists started a research project on the species, which in 2014 changed into a European Union LIFE+ funded translocation and conservation project. By late 2021 this reintroduced Waldrappteam population comprised c. 200 individuals, in two breeding colonies in southern Germany and two in Austria (Fritz, Reference Fritz2021). The common wintering area is the nature reserve WWF Oasi Laguna di Orbetello in Tuscany, Italy (Fig. 1). All four breeding colonies are considered to be one population because they share the same wintering ground (Wirtz et al., Reference Wirtz, Boehm, Fritz, Kotrschal, Veith and Hochkirch2018).

Fig. 1 The established breeding grounds and the common wintering ground of the Waldrappteam population of northern bald ibis Geronticus eremita. Black lines indicate migration routes.
In Andalusia, Spain, another reintroduction project with northern bald ibises, Proyecto Eremita, is ongoing, (Boehm et al., Reference Boehm, Bowden, Seddon, Hatipoglu, Oubrou and el Bekkay2020). This population was also founded with hand-reared birds from various zoo breeding colonies, with the aim of establishing a self-sustaining wild population. In contrast to the migratory Waldrappteam population, these birds have a largely sedentary lifestyle. Since 2008 chicks from this Spanish population have been hatching in the wild and by 2020 this population consisted of c. 115 birds (M. Quevedo, pers. comm., 2020). Remote monitoring indicates that to date there has been no overlap of the activity areas of these two populations (for more information on the study species and the reintroduction projects see Supplementary Material 1).
Methods
We included 384 northern bald ibises from the 2008–2019 generations in the study. They had either hatched in zoo breeding colonies (mainly Zoo Rosegg, Austria; Cumberland Wildpark Gruenau, Austria; and Zoo Zurich, Switzerland) and had been raised by human foster parents for release, or they had hatched in the wild as offspring of released birds and had hence been raised by their biological parents. All foster parent-raised individuals originated from the European Endangered Species Programme. Birds for release were trained to follow a microlight plane that led them to the common wintering ground, where they were integrated into the wild population (Fritz et al., Reference Fritz, Kramer, Hoffmann, Trobe and Unsoeld2017a,Reference Fritz, Wirtz and Unsoeldb). Since 2011, an increasing number of chicks have been raised in the wild by their biological parents and, at their first migration, they follow their conspecifics to the common wintering ground. At the time of the present analysis some of the foster parent-raised individuals had not yet been assigned to a breeding colony because they had not reached sexual maturity and thus had not returned to a breeding area.
Intensive monitoring has yielded comprehensive life history data for each individual. In 2012, 68% of the birds carried GPS tags, and from 2013 onwards the whole population was monitored remotely (Sperger et al., Reference Sperger, Heller, Voelkl and Fritz2017). Initially, only battery-powered devices were used (either specially manufactured devices for our needs or commercial devices made by Fleetronics, Brussels, Belgium; company no longer exists). From 2016 onwards, solar-powered devices became available (custom-made devices from the University of Konstanz, Konstanz, Germany, and commercial devices from Ornitela, Vilnius, Lithuania).
We assigned the 384 individuals to four demographic stages representing age classes with characteristic life-history events (Fig. 2, Supplementary Fig. 1). Stage 1 represents juveniles from fledging until the end of their first year; after the first autumn migration these birds usually remain at the wintering grounds. Stage 2 represents juveniles in their second year; they usually stay year-round at the wintering grounds. Stage 3 represents subadults in their third year; although not mature, most of these birds perform a partial or even full migration. Stage 4 represents adults older than 3 years; these birds usually migrate and reproduce annually although some individuals could remain at the wintering grounds.

Fig. 2 Life cycle of the northern bald ibis. Life stages are in circles (for definitions, see text); s1–s4 represent the survival probabilities of the respective stages. Light grey section: the part of the population raised by biological parents; dark grey section: the part of the population raised by human foster parents. ASP = adult females added temporarily. Only the female part of the population was considered for the simulations. (Readers of the printed journal are referred to the online article for a colour version of this figure.)
We calculated the survival probabilities per stage, sex, colony and raising type (biological or foster parent) using the Kaplan–Meier estimator (Kaplan & Meier, Reference Kaplan and Meier1958) with the survival package (Therneau, Reference Therneau2015) in R 4.0.3 (R Core Team, Reference Core Team2020). We estimated the survival probabilities for fledglings that were c. 45 days of age. We omitted adult birds that were added temporarily during the breeding season from the calculations of the survival probabilities. These birds do not migrate with the population but are released temporarily and added to the population during the breeding season, to improve breeding success. They are therefore not a permanent part of the population. We used a likelihood ratio test to test for significance between different classes in Cox proportional hazard models (Kleinbaum & Klein, Reference Kleinbaum and Klein2012). We also calculated hatching and fledging rates.
We calculated fecundity as the reproductive rate per nest (i.e. the mean number of fledglings per nest). At the start-up of newly founded colonies, adult males and females were temporarily released at the breeding site and re-caught after the breeding season to compensate for missing mates and to enable the migrant birds to reproduce. The rate of adults added temporarily was successively reduced, and this supplementation was completely terminated at the end of the data period (see also Supplementary Material 2).
Management of the Waldrappteam population
During the course of the reintroduction project a range of management measures were implemented (Table 1). The efficiency of the human-led migration as the main translocation method was improved substantially. The number of translocated and released juveniles per season was increased from c. 10 per season initially, up to 30 per season (Fritz et al., Reference Fritz, Kramer, Hoffmann, Trobe and Unsoeld2017a,Reference Fritz, Wirtz and Unsoeldb; Fritz, Reference Fritz2021).
Table 1 Quantification of main management measures and stochastic events affecting the Waldrappteam population of the northern bald ibis Geronticus eremita during the study period (2008–2019). These data provide the framework for modelling the respective outcomes of different future management interventions and stochastic events.

Adding adult birds temporarily at the breeding sites to provide mating partners for arriving migrants has proven to be an efficient method to increase the reproduction rate, particularly in small founder colonies. During the study period 38% of the breeding pairs included adults added temporarily and 39% of the fledglings came from such couples.
During 2008–2013 the mean annual loss rate was 25% (44 lost birds in total). Illegal hunting in Italy caused an annual mean of 40% of these losses (14 hunted birds in total). During the LIFE+ period (2014–2019), a campaign against this environmental crime was implemented based on remote GPS monitoring of the whole population, enabling the identification of accidents and the implementation of effective post-poaching measures in the field and for complaints to be sent to the responsible public prosecutors’ offices. One perpetrator was charged and convicted, which as a precedent produced a lasting effect (Fritz, Reference Fritz2015; Fritz et al., Reference Fritz, Kramer, Hoffmann, Trobe and Unsoeld2017a,Reference Fritz, Wirtz and Unsoeldb). With these measures, the mean loss rate from this crime dropped to 17% of the overall annual losses, corresponding to a reduction of 59% from the pre-LIFE+ rate.
During the whole study period (2008–2019), the mean annual loss rate was 31% (224 lost birds in total). Electrocution of birds roosting on unsecured power poles caused an annual mean of 45% of these losses (45 birds in total). In Germany, most unsecured power poles were retrofitted by 2016 because of a legal regulation. Before these measures were introduced, 41% of all electrocution losses (seven cases during 2008–2015) occurred in Germany, whereas since these measures were introduced (from 2016 onwards) only one further case has been documented in Germany (Fritz et al., Reference Fritz, Unsoeld, Voelkl, Kaufman, Bashaw and Maple2019). This corresponds to a 91% reduction in the loss rate in Germany because of these retrofitting measures, indicating the possibility of almost complete elimination of this cause of death. Population development during the study period was also impaired occasionally by stochastic events, in particular the early onset of winter, brood failure because of predation, and infections (Table 1).
Population viability analysis
For the population viability analysis and population projections, we focused on the female half of the population (the sex ratio is 1:1). Amongst the fledglings with unknown sex (five individuals in the whole population) we included 50% in the analysis (see also Supplementary Material 2).
We analysed the population trajectories for different scenarios as outlined below. We calculated extinction probability as the proportion of 100 repetitions in which the population went extinct within 50 years (PEXT_50). We calculated the intrinsic growth rate λ as the mean annual finite rate of change of the population size, where λ > 1 implies population growth and λ < 1 implies a declining population size. We calculated the effect of input parameters on demographic rates for scenarios where λ > 1 and PEXT_50 ≤ 5%. In addition, we ran generalized linear models (GLMs) and used an ANOVA to rank the contributions of the demographic rates in the different life stages to λ as the response variable using a gamma error structure with inverse link. We also included stochastic events (catastrophic events that could reduce the survival of the population) in the model, with frequency and severity in the given range of stochastic events observed during the study period (Table 1). We calculated how often each combination of scenarios (see below) resulted in λ > 1 and PEXT_50 ≤ 5%.
The model documentation follows the TRACE documentation framework (TRAnsparent and Comprehensive model Evaluation; Grimm et al., Reference Grimm, Augusiak, Focks, Frank, Gabsi and Johnston2014; Supplementary Material 3). The model description follows the ODD protocol (Overview Design concepts and Details; Grimm et al., Reference Grimm, Berger, Bastiansen, Eliassen, Ginot and Giske2006, Reference Grimm, Berger, DeAngelis, Polhill, Giske and Railsback2010). An individual-based model was implemented in NetLogo 6.0.3 (Wilensky, Reference Wilensky1999). The corresponding NetLogo and R scripts are available online (Supplementary Material 4).
For the population viability analysis, we defined three different reproductive rates depending on the included female fledglings and fledglings of unknown sex (Supplementary Material 2). The ‘baseline’ rate included female fledglings raised by adult wild females (stage 4). The ‘status quo’ rate also included female fledglings raised by females added temporarily. The ‘all chicks’ rate also included female fledglings raised by human foster parents. To calculate the reproductive rate, we divided the number of fledglings by all adult females (stage 4) of the population, irrespective of whether they were breeding or not, plus adult females that were added temporarily at the breeding site to compensate for missing mates. Thus, the reproductive rate used in the context of the population viability analysis is an expression of breeding probability, indicating how likely it is that a female will breed.
We also calculated the baseline rate separately for the two breeding colonies in Burghausen and Kuchl, and separately for females raised by biological parents and human foster parents. We implemented generalized linear mixed models (GLMMs) using a Poisson error structure and maximum likelihood to investigate whether any differences in reproduction rate were significant.
We analysed the model for various future scenarios that are based on realistic alternative management measures from which we derived the survival and reproduction values (Table 1). The management scenarios compare situations in which no management took place to scenarios in which baseline demographic rates were improved by management interventions of 10, 25 and 100% (Table 1). We combined three levels of survival rate at each stage and four levels of reproductive rate in a full factorial design. Additionally, we combined the ‘status quo’ rate and ‘all chick’ rate with baseline survival probabilities, which resulted in 326 management scenarios (Table 2, Supplementary Material 5). Only for the simulation of the baseline scenario did we draw the demographic rates from the probability distributions defined by the mean and standard deviation of the demographic rates. For all other scenarios we used the calculated mean values of the demographic rates. Of these 326 management scenarios, we chose 14 management scenarios of special interest for closer examination (Table 2), where parameter combinations were within the scope of realistic management interventions that aimed to improve survival and reproduction. If these simulations resulted in λ > 1 and PEXT_50 ≤ 5%, they were chosen for testing the robustness of the population to stochastic events such as periods of inclement weather affecting demographic rates and the supplemental release of foster parent-raised juveniles. This happened in nine cases (Table 2). For comparison, we also tested the baseline scenario even if λ < 1 and PEXT_50 ≥ 5%, and the ‘all chicks scenario without supplements’ under stochastic events (Supplementary Material 5).
Table 2 Empirical values for survival per stage and for the three reproductive rates (RRs) of the Waldrappteam population of northern bald ibises as defined in the text (Fig. 4). We used these values and their improvements by 10, 25 or 100% in various combinations for the population simulation of 14 management improvement scenarios, which we designed based on management measures implemented during the study period (Table 1). Improvements of the empirical values are indicated with asterisks (*). The resulting population growth rate (λ) and extinction probability (PEXT_50) are shown in the two right-hand columns. Note that the reproductive rates comprise not just the reproducing females but all adult females in adult stage 4 and take into account only female offspring (i.e. per nest number of offspring); s1–s4 are the survival probabilities of the respective stages.
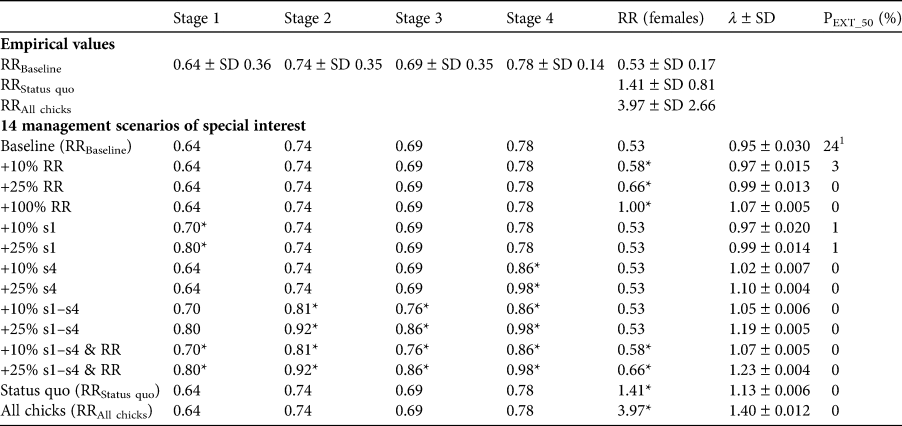
1 For a simulation across 100 years the extinction probability would be 87%.
The number of stochastic event and juvenile supplementation scenarios is given by equation 5.1.2 in Supplementary Material 5. These 11 scenarios were crossed with four levels of stochastic event frequency (5, 10, 15 or 20%), five levels of severity (5, 10, 15, 20 or 25 additional mortalities per stage), two levels of the number of juvenile supplements (15 or 30) and two levels of the timespan for supplementing individuals (4 or 7 years; i.e. number of years in which juveniles are added to the population in addition to natural reproduction; supplemented juveniles remain in the population until their death, injury or disappearance), which resulted in 820 parameter combinations for the stochastic event and juvenile supplementation scenarios.
Results
Of the 384 individuals, 195 could be confirmed as males and 184 as females, resulting in a sex ratio of 1.00:0.94 (male:female). For five juveniles sex was not determined at the time of data analysis. With respect to the raising type, 162 birds were raised in the wild by biological parents (42%), 213 by human foster parents (55%) and nine birds were supplemented as juveniles (2%). With respect to the breeding colonies, 127 individuals belonged to Burghausen (33%), 89 to Kuchl (23%), 89 to Überlingen (23%) and 22 to Rosegg (6%); 57 juveniles were not yet assigned at the time of data collection. A total of 232 birds died during the data collection period; of these, 123 died during stage 1 (53%), 45 during stage 2 (19%), 33 during stage 3 (14%) and 31 during stage 4 (13%; Supplementary Fig. 1).
At the end of 2019, 152 individuals were alive, including nine that were caught and placed into captivity mainly because of various kinds of severe and permanent injury. We counted these individuals as censored and so they did not affect the survival estimates. Thus, the release colony at the end of the data collection period comprised 143 individuals (69 males and 74 females). With respect to the life stages, 71 individuals belonged to juvenile stage 1 (50%; 37 females, 34 males), 28 to juvenile stage 2 (20%; 11 females, 17 males), 13 to subadult stage 3 (9%; 8 females, 5 males) and 31 to adult stage 4 (22%; 18 females, 13 males). With respect to the breeding colonies, 33 birds belonged to Burghausen (23%), 33 to Kuchl (23%), 51 to Überlingen (36%) and 15 to Rosegg (10%); 11 individuals (8%) were not yet allocated to a colony at the end of the data collection period. With respect to the raising types, 68 birds (48%) were raised by their biological parents, 73 birds (51%) by human foster parents and two birds (1%) were supplemented.
We calculated survival rates for all individuals from the fledging stage onwards. According to the Cox proportional hazard model, survival rates did not differ significantly between stages across the whole population (likelihood ratio test (N = 736) = 3.05, P = 0.4) or between sexes (likelihood ratio test (N = 379) = 0.80, P = 0.4). According to the Kaplan–Meier estimator, mean survival probability was 0.64 ± SD 0.36 for juvenile stage 1, 0.74 ± SD 0.35 for juvenile stage 2, 0.69 ± SD 0.35 for subadult stage 3 and 0.78 ± SD 0.14 for adult stage 4. The cumulative survival probability until sexual maturity (end of stage 3) was 0.33 ± SD 0.23. Concerning pre-fledging survival, 72% of the eggs hatched and 83% of the hatched chicks fledged (Table 3).
Table 3 Breeding statistics of the Waldrappteam population of northern bald ibises during 2011–2019.

1 Per cent eggs hatched.
2 Per cent hatchlings fledged.
The survival rates for biological parent- and foster parent-raised individuals differed significantly (likelihood ratio test (N = 375) = 8.26, P = 0.004; Fig. 3), as well as per stage and per raising type (likelihood ratio test (N = 715) = 10.17, P = 0.04). In juvenile stage 1, foster parent-raised individuals showed a higher survival rate (0.73 ± SD 0.37) than their biological parent-raised counterparts (0.52 ± SD 0.30; Table 2). The survival rates in the two breeding colonies at Burghausen and Kuchl did not differ significantly (likelihood ratio test (N = 216) = 1.56, P = 0.2), nor per stage and per colony, respectively (likelihood ratio test (N = 453) = 5.36, P = 0.3; Table 2).

Fig. 3 Survival plot of the raising types of the Waldrappteam population of northern bald ibises; p is the result of the likelihood ratio test. Shading surrounding the lines represents the standard deviation. Because of the young age of this newly founded population, only a small number of individuals reached an age older than 3 years (stage 4) during the study period, which causes a relatively large standard deviation.
The number of nests increased over time, with one nest in 2011 and 14 nests in 2019 and a mean of 3.43 ± SD 0.79 eggs per nest (Table 3). Overall fecundity was 2.15 ± SD 0.70 fledglings per nest. Fecundity rates amongst foster parent- and biological parent-raised females did not differ significantly (z = 0.68, P = 0.50), nor did the fecundity rates amongst females of the breeding colonies at Burghausen and Kuchl (z = 0.60, P = 0.55).
During the study period, 31 females reached sexual maturity (stage 4). Of these, 45% reproduced by the end of the data collection period. Of these 14 reproducing females, three (21%) reproduced once, five (36%) reproduced twice, five (36%) reproduced three times and one (7%) reproduced six times.
Population viability analysis
Over the data collection period 62 individuals reached adult stage 4, of which 31 were sexually mature females. Analysing these females as potential mothers yielded a baseline reproductive rate of 0.53 ± SD 0.17 (biological parent- and foster parent-raised females and their female fledglings; Table 2), a status quo reproductive rate of 1.41 ± SD 0.81 (female fledglings from females added temporarily also included) and an all chick reproductive rate of 3.97 ± SD 2.66 (human-raised and released female fledglings also included). We used these reproductive rates for the population viability analysis.
Without further implementation of management measures (Table 1), as assumed for the baseline management scenario, the population viability analysis indicated a λ of 0.95 ± SD 0.03, which is below the 1.00 threshold, with a 24% extinction probability within 50 years (Table 2). The full factorial design resulted in 326 management scenarios (Supplementary Material 5) with λ being 0.95–1.40. λ was > 1 and PEXT_50 was ≤ 5% in 308 of the 326 management scenarios (94%); in none of these 308 scenarios did the population go extinct in the 50-year projections. In a first analysis we investigated the frequencies of management scenarios with different demographic rates of the 308 scenarios with positive population development. This enabled us to understand how demographic rates affected population viability (Supplementary Material 5). The ‘status quo’ and ‘all chicks’ management scenarios were not considered here (therefore leaving 306 of 308 scenarios) because they were only simulated once with baseline survival values and the respective reproductive rate. In the 306 management scenarios with positive population development, only an increase in adult survival (stage 4) values improved the population viability: a 10 or 25% increase of adult survival increased the viability disproportionately. For example, a 10% increase of adult survival increased viability by 17%, thereby increasing the likelihood of avoiding population extinction (108 of 108 parameter combinations with adult survival increased by 10% led to positive population growth) compared to the baseline value (90 of 108 parameter combinations (83%) with baseline adult survival led to positive population growth). These results indicate the importance of survival of the adult reproductive stage for population viability. This is supported by the GLMs and the ANOVA ranking the effects of the survival rates (deviance (d) of ANOVA: d stage 1 = 0.05, d stage 2 = 0.05, d stage 3 = 0.05, d stage 4 = 0.68). Here, adult survival (stage 4) had the largest effect (Supplementary Material 6).
All management scenarios in which the reproductive rate was increased by 100% showed positive population development (81 of 81 parameter combinations). Only one management scenario in which the reproductive rate was increased by 25% did not show positive population development: the one in which no survival rate was increased (i.e. baseline values for stages 1–4, +25% reproductive rate management scenario; Table 2). Nevertheless, 88% of the management scenarios with the baseline reproductive rate showed positive population development (71 of 81 parameter combinations). The reproductive rate also had a strong effect on λ (d reproductive rate = 0.58) in the GLMs.
In the second analysis, 14 management scenarios of special management interest were analysed (Table 2, Fig. 4). For nine of these, λ was > 1 and PEXT_50 was ≤ 5%. If only the reproductive rate was increased, positive population growth occurred only after an increase of at least 100% (+100% reproductive rate, status quo, all chicks; i.e. a minimum of one female fledgling per female). Increasing only the survival of juveniles (stage 1) did not lead to positive population growth, but increasing adult survival (stage 4) did. An increase in all survival values for stages 1–4 also led to positive population growth; these effects were reinforced by an additional increase in the reproductive rate.

Fig. 4 Numbers of females per year in the 14 management scenarios of special interest for the Waldrappteam population of northern bald ibises (Table 2). Each line corresponds to a run of the respective scenario, and the bold black line is the mean of all 100 runs per scenario. λ ± SD is specified for each scenario. The description of each scenario is indicated in the heading of the graph. s1–s4, stage-specific survival probabilities; RR, reproductive rate. The starting point is 74 females (i.e. the number of females present in 2019).
For the stochastic event and juvenile supplementation scenarios, λ was 0.91–1.40. In 714 of 820 scenarios (87%), λ was > 1 and PEXT_50 was ≤ 5%. Baseline survival values led to positive population growth in 74–79% of the scenarios. Increasing adult survival (stage 4) by 10% led to positive population growth in 89% of the scenarios, whereas a 10% increase in survival in other age categories led to population growth in 100% of the scenarios. Increasing baseline survival by 25% led to positive population growth in 100% of the stochastic event and juvenile supplementation scenarios.
Each combination of stochastic event frequencies and severities occurred 41 times in all 820 stochastic event and juvenile supplementation scenarios. Ninety per cent of the scenarios with a low frequency (5%) and severity (5%) of stochastic events and 80% of the scenarios with a high frequency (20%) and severity (25%) of stochastic events resulted in positive population development. Eighty-eight per cent of the stochastic event and juvenile supplement scenarios with 30 supplemented juveniles per year over 7 years and 86% of the scenarios with 15 supplemented juveniles per year over 4 years resulted in positive population development.
Discussion
The comprehensive 12-year dataset of the reintroduced Waldrappteam northern bald ibis population facilitated a population viability analysis of its future population development under different scenarios. This approach is a powerful tool to evaluate the effectiveness of reintroduction and management scenarios. Such models, based on the best possible evidence, are recommended in the IUCN reintroduction guidelines (IUCN/SSC, 2013) for deciding on future management practices.
The outcome of the population viability analysis baseline scenario, with λ at 0.95 and a 24% extinction probability within 50 years, supports the rejection of the hypothesis that the reintroduced northern bald ibis population can survive without continuation of the management measures implemented during the study period (Table 1). Nonetheless, the population is close to a transition to sustainability, reflected in the comparatively good survival and fecundity values.
The baseline reproductive rate relates the actual number of fledged female offspring to the entirety of the adult females in the population, including the 39% non-reproducing females. It is an expression of the reproductive potential in the population. The given baseline rate of 0.53 could be increased significantly through translocation measures up to a rate of 3.97 (‘all chicks' reproductive rate). As indicated by the management improvement scenarios, continuing and improving the given translocation measures would have the most significant immediate effect on population development. Furthermore, continuation of measures reducing the causes of primary mortality could further improve survival and population growth.
In agreement with the outcome of the population viability analysis, the sustainable improvement of long-term survival is the primary objective of the reintroduction project, although the intraspecific comparison indicates that the survival values in the Waldrappteam population are already promising for all age classes. Improving the survival rate of adults should be a priority as this has the greatest impact on λ. This has also been confirmed for other long-lived species (Lebreton & Clobert, Reference Lebreton, Clobert, Perrins, Lebreton and Hirons1991; Lampila et al., Reference Lampila, Orell, Belda and Koivula2006; Pistorius et al., Reference Pistorius, Follestad and Taylor2006; Schaub et al., Reference Schaub, Zink, Beissmann, Sarrazin and Arlettaz2009; O'Shea et al., Reference O'Shea, Ellison and Stanley2011).
Demographic analysis
The demographic dataset covers a period of 12 years, including the later phase of a feasibility study (2008–2013) in which a small number of birds were released and the following 6-year period of the reintroduction project funded by LIFE+ (2014–2019). The survival probability in our study ranged from 0.64 for juveniles (stage 1) to 0.78 for adults (stage 4). A similar adult survival rate of 81% has been observed for a small relict migratory northern bald ibis population that was discovered in Syria in 2002 (Serra et al., Reference Serra, Lindsell, Peske, Fritz, Bowden and Bruschini2014). For the Moroccan population, an annual survival rate estimate of 77% has been reported across all age classes (Bowden et al., Reference Bowden, Smith, El Bekkay, Oubrou, Aghnaj and Jimenez-Armesto2008), which is within the range of the rates calculated for the Waldrappteam population. However, the Moroccan birds are not marked; their survival rate can only be estimated indirectly based on the annual counts of the pre-breeding population and the number of fledglings (Bowden et al., Reference Bowden, Smith, El Bekkay, Oubrou, Aghnaj and Jimenez-Armesto2008).
The cumulative survival probability until sexual maturity (stages 1–3) was 0.33. In the relict Syrian population the count of departing fledglings and arriving new breeders at the breeding site in Syria indicates a 14% survival rate until sexual maturity for this declining population (Boehm et al., Reference Boehm, Bowden, Seddon, Hatipoglu, Oubrou and el Bekkay2020), which is less than half that of the Waldrappteam population. Satellite tracking of the Syrian juveniles revealed they did not arrive at the common wintering site in Ethiopia because they lost the company of experienced conspecifics and died on the Arabian Peninsula (Serra et al., Reference Serra, Lindsell, Peske, Fritz, Bowden and Bruschini2014). This low survival rate of juveniles was a major cause of the extinction of this population in 2015 despite international conservation and translocation efforts (Bowden et al., Reference Bowden, Hamoud, Jbour, Fritz, Peske, Riedler and Soorae2011).
In our population, the first-year survival rate of foster parent-raised juveniles (0.73) was significantly higher compared to that of their biological parent-raised counterparts (0.52). This is in contrast with the widespread experience that released individuals have comparably low survival rates, particularly in the first period after release, because of translocation- and competition-induced stress, dispersal, disease or predation (Parker et al., Reference Parker, Ewen, Seddon and Armstrong2013). This is true particularly for captive-bred individuals, probably because they lack individual experience (Mathews et al., Reference Mathews, Orros, McLaren, Gelling and Foster2005). Our birds underwent extensive pre-release training, which provided them with essential experience regarding navigation, flight techniques, weather conditions, aerodynamics and predator avoidance (Portugal et al., Reference Portugal, Hubel, Fritz, Heese, Trobe and Voelkl2014; Voelkl & Fritz, Reference Voelkl and Fritz2017; Fritz et al., Reference Fritz, Unsoeld, Voelkl, Kaufman, Bashaw and Maple2019). This is assumed to improve post-release survival (Alonso et al., Reference Alonso, Orejas, Lopes and Sanz2011; Houser et al., Reference Houser, Gusset, Bragg, Boast and Somers2011; Zhang et al., Reference Zhang, Huang, Hong, Zhou, Huang and Li2017). In the Spanish northern bald ibis reintroduction project (Proyecto Eremita), the mean first-year survival rate for released juveniles of 31% (data for 2004–2018; Boehm et al., Reference Boehm, Bowden, Seddon, Hatipoglu, Oubrou and el Bekkay2020) was substantially lower than that of the Waldrappteam population. We assume that minimal pre-release training and unguided autumn dispersal (Muñoz & Ramírez, Reference Muñoz and Ramírez2017) account for the lower survival of the juveniles released in Spain (Fritz et al., Reference Fritz, Unsoeld, Voelkl, Kaufman, Bashaw and Maple2019).
The survival probability rate of the Waldrappteam population from hatching to fledging was 83%, which is substantially higher than the rate of 47% reported for the Moroccan wild population (Bowden et al., Reference Bowden, Aghnaj, Smith and Ribi2003). We assume that this difference mainly reflects the quality of the feeding habitats. For the Waldrappteam population, studies indicate high feeding efficiency and a high abundance of food invertebrates (mainly worms and larvae) in the soil of the preferred feeding habitats of meadows and pastures (Zoufal et al., Reference Zoufal, Fritz, Bichler, Kirbauer, Markut, Meran, Boehm, Bowden, Jordan and King2007; Fritz et al., Reference Fritz, Kramer, Hoffmann, Trobe and Unsoeld2017a,Reference Fritz, Wirtz and Unsoeldb). In contrast, the feeding habitats on the Atlantic coast of Morocco consist of semi-natural steppes with sparse cover of perennial and annual vegetation and low availability of freshwater bodies (Bowden et al., Reference Bowden, Smith, El Bekkay, Oubrou, Aghnaj and Jimenez-Armesto2008). Accordingly, positioning data indicate a small activity radius of c. 2 km around the nesting sites for the Waldrappteam population during the breeding season (Fritz et al., Reference Fritz, Kramer, Hoffmann, Trobe and Unsoeld2017a,Reference Fritz, Wirtz and Unsoeldb), whereas the foraging area of the two Moroccan colonies covers a strip of c. 4 km inland along a 50-km coastline. The fledgling survival rate in Morocco was even lower in the 1990s before supplementary freshwater was provided near the breeding grounds from 1998 onwards (Smith et al., Reference Smith, Aghnaj, El Bekkay, Oubrou, Ribi, Armesto and Bowden2008).
In the Waldrappteam population 61% of the mature females reproduced. Most of the non-reproducing females remain at the wintering site. In comparison, 63% of the full-grown (mature) birds in the Moroccan population reproduced (Bowden et al., Reference Bowden, Smith, El Bekkay, Oubrou, Aghnaj and Jimenez-Armesto2008). These rates are not directly comparable because the Moroccan data are based on counts of unmarked birds. Nonetheless, the data indicate that in this sedentary population in Morocco (with all birds on site) a similar proportion of adults do not reproduce.
The mean annual fecundity of the Waldrappteam population was 2.15 fledged chicks per nest. Fecundity values of northern bald ibis populations vary considerably, with 1.23 fledged chicks per nest in the wild sedentary Moroccan population, 0.97 fledged chicks per nest in the Spanish Proyecto Eremita release population (Table 4; Boehm et al., Reference Boehm, Bowden, Seddon, Hatipoglu, Oubrou and el Bekkay2020) and 2.24 fledged chicks per nest in the managed, temporarily wild-living population at Zoo Rosegg in Austria. The fecundity in the Waldrappteam population is therefore near the upper end of this range. This is consistent with the high survival probability from hatching to fledging of the Waldrappteam population (83%) and indicates a high quality of breeding habitats.
Table 4 Comparative statistics on fecundity (mean ± SD number of fledglings per nest) of the Waldrappteam population of northern bald ibises and the crested ibis Nipponia nippon. Two populations are managed zoo colonies, which are self-sustaining and free-flying from the start of the breeding season onwards but dependent on human care during winter.

1 Data from 2012 and 2013 were excluded because of brood control.
2 Core facility of the University of Vienna.
Conclusion
Despite comparatively good survival and fecundity data, the population viability analysis indicated that the Waldrappteam population needs further management and translocation measures. It is important at this stage to plan the type and duration of further management and translocation based on a quantitative, systematic analysis.
Based on the modelling outcomes, a major focus for the future management of this population should be further improvement of survival rates, with a particular focus on adult survival. This will be achieved mainly by implementing measures reducing the major mortality causes, namely illegal hunting in Italy and electrocution on unsecured power poles at the breeding sites (Fritz et al., Reference Fritz, Unsoeld, Voelkl, Kaufman, Bashaw and Maple2019). In addition, the proportion of reproducing females should be increased by establishing a breeding site south of the Alps that can be reached by the migrating birds in spring without crossing the Alpine chain.
The population viability analysis could be improved by including data on the movement of the northern bald ibises. This would help build a spatially explicit population simulation model accounting for differences in the migration routes to and from the colonies. Moreover, a thorough assessment of the factors influencing the viability of the northern bald ibises in the colonies would be an asset. These factors include the availability of food and nesting sites, and the weather in the breeding areas during the migration and in the wintering area (Schaub et al., Reference Schaub, Kania and Köppen2005).
The comparison of the demographic data with populations exhibiting a sedentary lifestyle did not indicate adverse effects of the migratory lifestyle in the Waldrappteam population. The survival rate of adults is similar to those of sedentary populations and the survival rate of released juveniles is higher in the migratory population (73%) compared to the sedentary population in Spain (31%). The high fecundity rate in the Waldrappteam population (2.15 fledged chicks per nest) presumably reflects the migratory behaviour that enables the adults to exploit rich northern feeding grounds during the reproductive period.
The current International Single Species Action Plan for the northern bald ibis (Bowden, Reference Bowden2015) does not include modelling either as an objective or as a recommendation. Systematic, quantitative approaches such as population viability analyses are essential strategic tools that are being used to an increasing extent in animal conservation and translocation efforts. It is essential that these tools are also implemented increasingly in further species conservation measures for the northern bald ibis. Regarding the Waldrappteam population, the results of this study will be incorporated into further conservation and reintroduction measures. This population should become self-sustaining as part of a second European LIFE project (2022–2028).
Acknowledgements
The northern bald ibis reintroduction project was co-financed with a 50% contribution from the LIFE financial instrument of the European Union (LIFE+ 12-BIO_AT_000143, LIFE Northern Bald Ibis) during 2014–2019. SD, VR, CS and SKS are associated with the Deutsche Forschungsgemeinschaft-funded research training group BioMove (RTG 2118-1). We thank Laura Stefani and Michael Stachowitsch for commenting on the text.
Author contribution
Study design and conception: SD, JF, SK-S; data analysis: SD, VR; population viability analysis design: SD, VR; population viability analysis programming: SD; programming support: CS; field data collection: CE; writing lead: SD; contribution to writing: VR, IK, JF, SK-S.
Conflicts of interest
None.
Ethical standards
This research abided by the Oryx guidelines on ethical standards.










