Introduction
Considerable gth of human populations in the last few decades has had a significant negative impact on biodiversity (Hanski, Reference Hanski2005). The degradation of wildlife habitats has resulted in declines of species, many of which are threatened with extinction (Ginsberg & Macdonald, Reference Ginsberg and Macdonald1990; Nowell & Jackson, Reference Nowell and Jackson1996; Mills & Hofer, Reference Mills and Hofer1998; Woodroffe, Reference Woodroffe2000). One of the key factors causing the decline of most large carnivore species is conflict with humans because of predation of livestock (Cozza et al., Reference Cozza, Fico, Battistini and Rogers1996; Woodroffe, Reference Woodroffe2000; Treves & Karanth, Reference Treves and Karanth2003) and attacks on humans (Kerbis Peterhans & Gnoske, Reference Kerbis Peterhans and Gnoske2002; Packer et al., Reference Packer, Ikanda, Kissui and Kushnir2005). Human–predator conflicts cause significant economic losses (Mishra, Reference Mishra1997; Butler, Reference Butler2000; Patterson et al., Reference Patterson, Kasiki, Selempo and Kays2004; Van Bommel et al., Reference Van Bommel, Bij de Vaate, Boer and Iongh2007; Palmeira et al., Reference Palmeira, Crawshaw, Haddad, Ferraz and Verdade2008) and can lead to retaliatory killing of predators (Ogada et al., Reference Ogada, Woodroffe, Oguge and Frank2003; Holmern et al., Reference Holmern, Nyahongo and Røskaft2007), and thus constitute a threat to both wild species and human livelihoods (Woodroffe & Ginsberg, Reference Woodroffe and Ginsberg1998; Hussain, Reference Hussain2003).
Human–wildlife conflicts have intensified in most African countries in recent decades because of exponential human population growth and economic activities (Woodroffe, Reference Woodroffe2000; Conover, Reference Conover2002). The highest intensity conflicts tend to occur where humans live adjacent to protected areas (Mishra, Reference Mishra1997; Conforti & de Azevedo, Reference Conforti and de Azevedo2003). In Africa there are a number of larger predator species, including the lion Panthera leo, leopard Panthera pardus, spotted hyaena Crocuta crocuta, baboons Papio sp., cheetah Acinonyx jubatus, African wild dog Lycaon pictus, caracal Caracal caracal and black-backed jackal Canis mesomelas (Butler, Reference Butler2000; Patterson et al., Reference Patterson, Kasiki, Selempo and Kays2004; Kolowski & Holekamp, Reference Kolowski and Holekamp2006; Holmern et al., Reference Holmern, Nyahongo and Røskaft2007; Van Bommel et al., Reference Van Bommel, Bij de Vaate, Boer and Iongh2007).
Livestock predation often follows a seasonal pattern (Butler, Reference Butler2000; Patterson et al., Reference Patterson, Kasiki, Selempo and Kays2004; Kolowski & Holekamp, Reference Kolowski and Holekamp2006) and is influenced by environmental conditions and husbandry practices (Ogada et al., Reference Ogada, Woodroffe, Oguge and Frank2003; Kolowski & Holekamp, Reference Kolowski and Holekamp2006). Most studies of predation on livestock in Africa have focused on East and Southern Africa, with few studies from West and Central Africa (Boy, Reference Boy1962; Sogbohossou, Reference Sogbohossou2004; Bauer & de Iongh, Reference Bauer and De Iongh2005; Van Bommel et al., Reference Van Bommel, Bij de Vaate, Boer and Iongh2007; Garba & Di Silvestre, Reference Garba, Di Silvestre, Croes, Buij, de Iongh and Bauer2008).
In contrast to East and Southern Africa, West Africa is characterized by low herbivore biomass (East, Reference East1984; Fritz, Reference Fritz1997) and fragmented wildlife populations mostly confined to small, unfenced protected areas that are surrounded by human settlements. The size of many of these reserves doesn’t guarantee the long-term conservation of their wildlife species (Woodroffe & Ginsberg, Reference Woodroffe and Ginsberg1998; Brashares et al., Reference Brashares, Arcese and Sam2001). Thus predation of livestock is inevitable (Binot et al., Reference Binot, Castel and Caron2006) and creates a negative attitude to conservation that can lead to the retaliatory killing of carnivores (Kolowski & Holekamp, Reference Kolowski and Holekamp2006; Holmern et al., Reference Holmern, Nyahongo and Røskaft2007).
The Pendjari Biosphere Reserve in the Republic of Benin is one of the best managed protected areas in the region, with one of the highest wildlife densities in West Africa (Delvingt et al., Reference Delvingt, Heymans and Sinsin1989; Lamarque, Reference Lamarque2004). However, the Reserve is located in an important livestock area in one of the poorest parts of the country. Livestock losses thus potentially affect the livelihood of local people. The Reserve is surrounded by a buffer and a hunting zone, intended to minimize human–wildlife conflict. The objectives of this study were to assess: (1) which species are responsible for livestock depredation, (2) any trends and seasonality of predation, (3) patterns of predation, and (4) any other factors that influence the occurrence of predation. We hypothesized that disturbance variables such as presence of safari hunting, poaching and illegal grazing will affect the intensity of livestock depredation.
Study area
The study was carried out around the Pendjari Biosphere Reserve in north-west Benin (Fig. 1). The Reserve is part of a complex of four adjoining protected areas (W, Pendjari, Arly and Oti-Mandouri) in four adjacent countries (Benin, Burkina Faso, Niger and Togo). Pendjari Biosphere Reserve was established in 1954, upgraded to National Park status in 1961 and to a UNESCO Man and Biosphere Reserve in 1986. It comprises Pendjari National Park (2,660 km2), Pendjari and Konkombri Hunting Zones (c. 1,600 and 251 km2, respectively) and a buffer zone with controlled land-use access for local people (c. 340 km2).
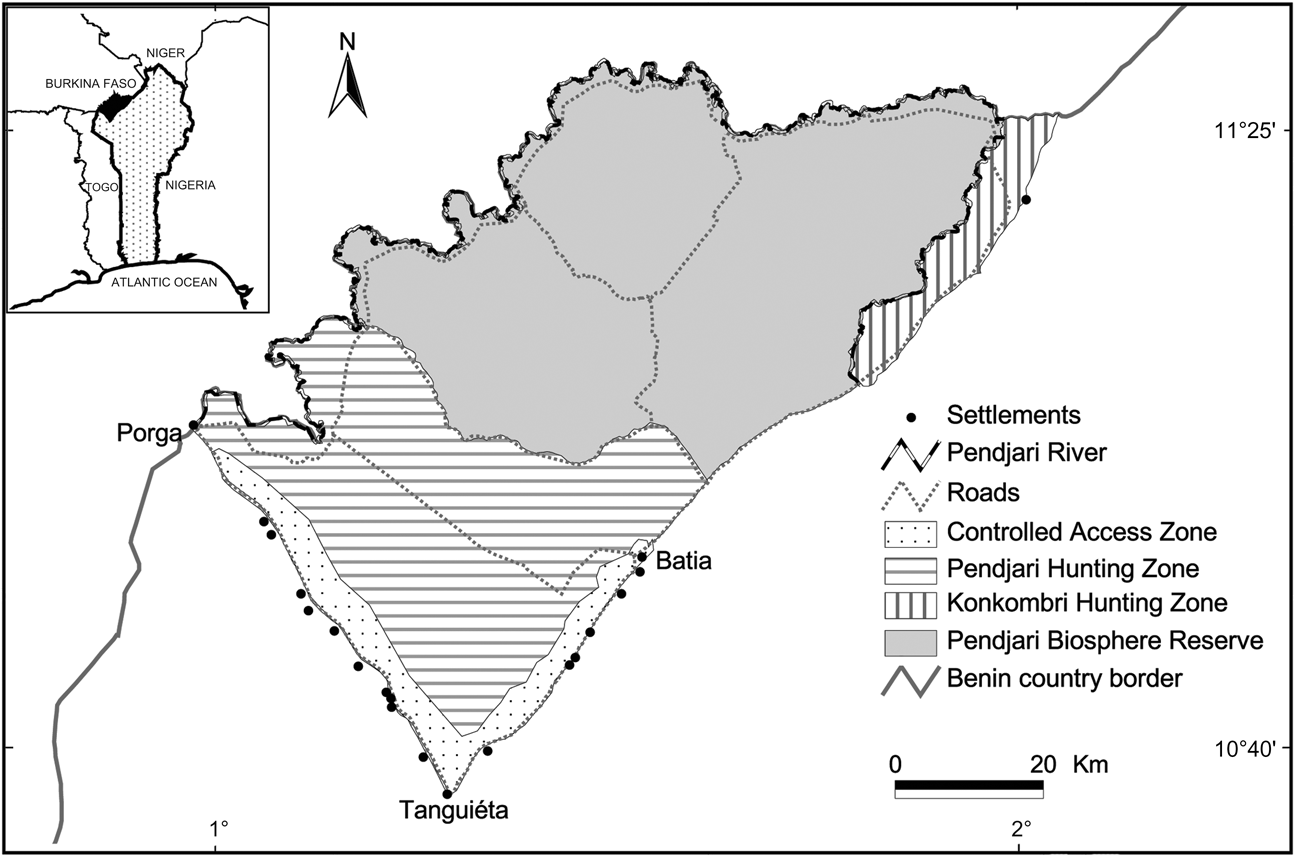
Fig. 1 Pendjari Biosphere Reserve and the surrounding controlled access and hunting zones. The black-shaded area on the inset indicates the position of the Reserve in north-west Benin, West Africa.
The Reserve is bordered to the north and west by the Pendjari River and to the east by the Atacora mountain range. In this Sudanian ecosystem the climate is characterized by a dry season from October to May and a wet season with a total annual rainfall of 800–1,000 mm. Vegetation is a mixture of open grass and tree savannahs interspersed with dry and gallery forests. These habitats harbour a variety of wildlife species including large carnivores (Delvingt et al., Reference Delvingt, Heymans and Sinsin1989). The density of lions in the Reserve is estimated to be between 0.67 (Di Silvestre, Reference Di Silvestre2002) and 1.5 lions per 100 km2 (Sogbohossou, Reference Sogbohossou2009) and the spotted hyaena occurs at a minimum density of 1.5 per 100 km2 (Sogbohossou, Reference Sogbohossou2009). The cheetah and wild dog populations, which almost disappeared, seem to be recovering, although numbers remain low, and there is no estimate of leopard abundance.
The Reserve is bordered by two main roads, Tanguiéta–Porga and Tanguiéta–Batia, along which there are 24 villages (Fig. 1). In addition to native farmers most villages are also inhabited by Fulani (with one to eight camps in each village), who are pastoralists. During the dry season migrating herds of cattle led by Fulani herdsmen from neighbouring countries reside within or close to the border of the Park in search of water and fodder.
The Reserve has been financed discontinuously through several programmes, with funding gaps almost abandoning the park to poachers during 1982–1985, 1991–1993 and 1998–2000. Since 2000 the Pendjari Project has managed the Reserve more intensively and illegal activities within the Reserve have largely been curtailed.
Methods
Data on the characteristics of human–wildlife conflict were collected from June to December 2007. All 24 villages surrounding Pendjari Biosphere Reserve were surveyed. We firstly discussed the history and characteristics of predation in group interviews. We then visited farmers’ households and Fulani camps, randomly, to ask more detailed questions about the characteristics of livestock depredation. In each household or camp we interviewed the head and if he was absent his elder son or the head’s wife. Other people present in a house usually helped in the recall of depredation cases. A total of 387 farmers’ households and 78 Fulani camps participated in the study. All predation cases from 2000 to 2007 were recorded. Group interviews allowed crosschecking of the data. Interviews were conducted by EAS with the help of a local guide. Colour plates of predator species were used during the interviews to ensure correct identification of species and their spoor. Respondents were also asked to describe the characteristics of the species to verify identification.
Data were analysed using SAS v. 9.1 (SAS Institute, Cary, USA). We considered predation by lions, spotted hyaenas and baboons. There were too few records of predation by leopards, cheetahs and wild dogs (< 3 per species) for analysis. Other predators (such as jackals, raptors and snakes), which mainly attack poultry, were not considered. The dependent variable is the intensity of depredation expressed as number of livestock killed. The independent variables used are presented in Table 1.
Table 1 The independent variables considered, by category, that could potentially influence predation occurrence and intensity around Pendjari Biosphere Reserve, and the method of analysis used for each variable.

* GLM, general linear modelling; PCA, principal component analysis
The distance to the closest protected area border (hunting zone or national park) from each village was determined from coordinates obtained with a global positioning system and ArcView v. 3.2 (ESRI, Redlands, USA). χ 2 tests were used to compare the intensity of depredation between predator and livestock species. We checked that the variables were not correlated. For variables with a continuous distribution we used a principal component analysis (PCA) to examine which variables significantly influenced the number of predation events (Table 1). We then tested these relationships using the Pearson non-parametric correlation.
For variables without the problem of co-linearity we used general linear modelling (GLM) to assess the relationship between predation intensity/frequency and the independent variables. The dependent variable was normalized using a log transformation. The minimum level of significance considered was P < 0.05. The GLM results are provided as F statistics.
Results
Livestock husbandry
Agriculture is the main source of rural livelihoods in the villages surveyed, with small-stock (sheep, goats and pigs) husbandry being of secondary importance. Cattle ranching, however, is the principal livelihood of the Fulani herders. Livestock represent savings for both local farmers and Fulani: the sale of small stock provides cash income to compensate for food shortages or to cover other expenses. Other sources of cash income include cotton cultivation, ecotourism and trade of natural resources (wood, straw, fruits) harvested in the Reserve.
Herding characteristics depend on the species and season. At night small stock are usually kept inside compounds or tied to trees. During the rainy season small stock are kept in enclosures, usually made of clay, or tied to trees to prevent them foraging in cultivated fields. In the dry season small stock roam freely in the village.
In the rainy season cattle are left to graze around the villages. During the dry season water and forage close to the villages become scarce and many Fulani herders allow their cattle to graze in the hunting zone. Some (1.2%) herders move their cattle to more humid areas in a rainy season migration. In this season 3.8% of herders leave the vicinity of the protected area to avoid conflicts with farmers caused by the grazing of farms by cattle.
Fulani camps comprise a circle of several huts or tents. Cattle are usually kept inside the circle of huts but sometimes a whole herd or a group of calves is kept in an enclosure made from thorny branches (Acacia spp., Dichrostachys cinerea and Balanites aegyptiaca). Thirteen percent of Fulani herders had received financial support from a project initiated by the Network of West and Central Africa for Lion Conservation to construct clay-brick enclosures to keep calves in at night.
Species involved in livestock predation
Lions (18.0%), spotted hyaenas (53.6%) and baboons (24.8%) were responsible for most livestock mortalities recorded (n = 752). Lions and hyaenas mainly attacked livestock during the night, whereas baboon attacks occurred during the day. The mean annual livestock loss per household was 1.8 head. The majority of livestock killed were sheep and goats (shoats, 52.1%), followed by pigs (42.3%), with cattle (3.7%) and dogs (1.9%) being infrequently taken. Predation intensity varied between predators (χ 2 = 66.28; P < 0.0001) and between livestock species (χ 2 = 47.04; P < 0.009; Fig. 2). Cattle were mainly killed by lions, and shoats by baboons and hyaenas. Pigs and dogs were mainly taken by hyaenas and lions.
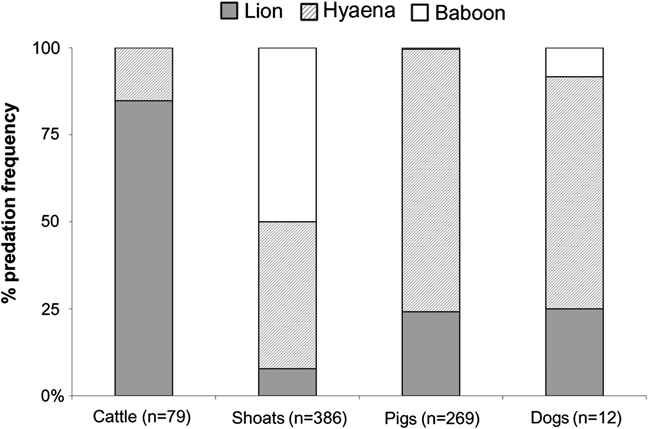
Fig. 2 Overall percentage frequency of attacks by lions Panthera leo, hyaenas Crocuta crocuta and baboons Papio anubis on cattle, shoats (sheep and goats), pigs and domestic dogs from 2000 to 2007 around Pendjari Biosphere Reserve (Fig. 1), based on information from the questionnaire survey.
Trends and seasonal distribution of predation
Predation intensity seemingly increased from five cases in 2000 to 222 cases in 2005 (Fig. 3), followed by a decline. Predation intensity varied by month (F = 4.43, df = 11, P < 0.0001) but not by season (F = 2.40, df = 1, P = 0.12). There was a peak at the end of the dry season in June–July and another at the end of the wet season in December (Fig. 4). This peak was particularly noticeable in the villages bordering the Atacora mountain range along the Tanguiéta–Batia road. On the Tanguiéta–Porga road a less pronounced peak is evident in the middle of the wet season to the beginning of dry season (Fig. 4). Lions and hyaenas mainly predated livestock from the end of the wet season to the beginning of the dry season, with predation by baboons being most intense at the end of the dry season and from the end of the wet season to the beginning of the dry season.

Fig. 3 (a) Total number of attacks by lions, hyaenas and baboons, and the three species combined, on livestock, irrespective of species, and (b) the number of cattle, shoats (sheep and goats), pigs and domestic dogs, killed by all predators combined, around Pendjari Biosphere Reserve (Fig. 1) from 2000 to 2007, based on the questionnaire survey.

Fig. 4 (a) Total number of monthly predation events along the Tanguiéta-Batia road, which is bordered by the Atacora mountain range, and the Tanguiéta-Porga road, and mean total monthly rainfall, and (b) total number of monthly attacks by lions, hyaenas and baboons, for 2000–2007.
The intensity of predation decreased when the rainfall of the previous month increased (r = -0.14, P = 0.007). However the rainfall of the current and previous years, and the month of predation, were not significantly correlated with the intensity of predation. The number of dry months in the year was significantly negatively correlated with the intensity of predation (r = -0.13; P = 0.011).
Geographical distribution of predation
The number of predation events was significantly different between the villages around the hunting zone (F = 4.26, df = 25, P < 0.001; Fig. 5). There was a significant difference between the two road axes in the number of livestock killed (F = 68.18, df = 1, P < 0.0001): 41.3% of the interviewees along the Tanguiéta–Porga road axis and 14.2% of interviewees along the Tanguiéta–Batia road axis had lost at least one animal to predation. Livestock predation intensity increased towards the National Park (r = -0.31; P < 0.0001) but not relative to the distance from a hunting zone (P > 0.05).
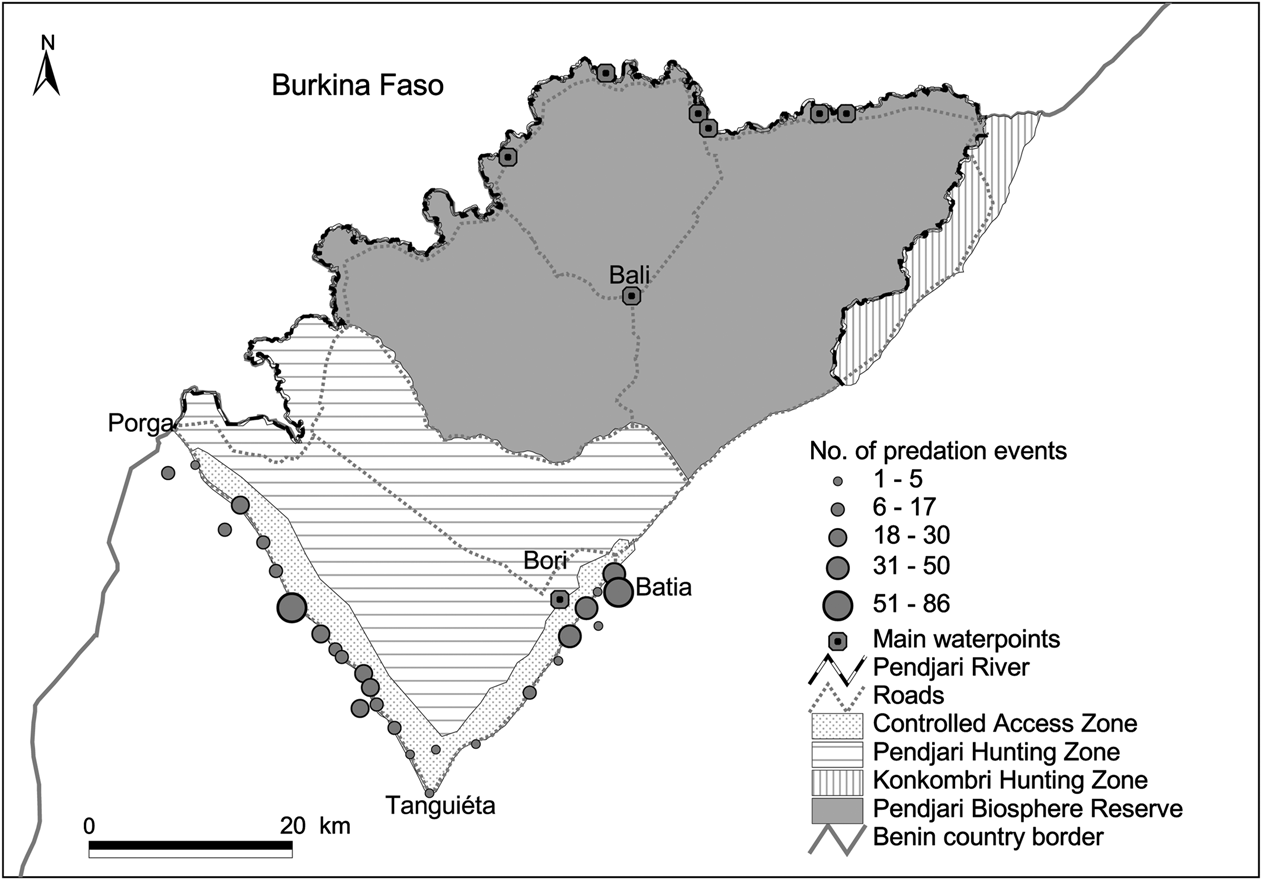
Fig. 5 Predation rates around Pendjari Biosphere Reserve. The size of the circles for each village around the hunting zone indicates the total number of predator attacks during 2000–2007.
Illegal herding and hunting
Only the annual number of herbivores killed by safari hunting had a significant impact on predation rate (r = -0.11; P = 0.03). The PCA and correlation indicated that the number of poachers and the number of herders arrested in the National Park in a year may have affected the intensity of predation in that year but the impact on predation intensity was not significant (P > 0.05).
Discussion
A generic problem with using questionnaires to assess depredation is that people invariably attribute livestock mortality to predators (Wagner, Reference Wagner1988; Hoogesteijn et al., Reference Hoogesteijn, Hoogesteijn and Mondolfi1993; Rasmussen, Reference Rasmussen1999). We believe that potential bias was minimal in this study as each interviewee generally reported few cases of depredation, and the low numbers involved may have helped the respondents remember specific cases. In many instances we obtained confirmation of predation events from neighbours.
There are several possible explanations for the recorded increase in predation intensity following the more intensive management of the Pendjari Biosphere Reserve that commenced in 2000. Firstly, the implementation of a management plan may have enhanced predator populations in the Reserve (Oli et al., Reference Oli, Taylor and Rogers1994; Saberwal et al., Reference Saberwal, Gibbs, Chellam and Johnsingh1994; Mishra, Reference Mishra1997; Wang & Macdonald, Reference Wang and Macdonald2006). Survey data suggest that the number of lions in the Reserve increased between 2002 (Di Silvestre, Reference Di Silvestre2002) and 2009 (Sogbohossou, Reference Sogbohossou2009). Alternatively, depredation could have increased because of growth of the human population, with increased encroachment, reduced natural prey populations and unfavourable habitat changes (Thouless & Sakwa, Reference Thouless and Sakwa1995; Cozza et al., Reference Cozza, Fico, Battistini and Rogers1996; Meriggi & Lovari, Reference Meriggi and Lovari1996; Mladenoff et al., Reference Mladenoff, Haight, Sickley and Wydeven1997; Dahle et al., Reference Dahle, Sørensen, Wedul, Swenson and Sandegren1998; Mizutani, Reference Mizutani1999; Woodroffe, Reference Woodroffe2000; Liu et al., Reference Liu, Linderman, Ouyang, An, Yang and Zhang2001; Naughton-Treves et al., Reference Naughton-Treves, Grossberg and Treves2003; Treves & Karanth, Reference Treves and Karanth2003; Kolowski & Holekamp, Reference Kolowski and Holekamp2006). We believe it most likely that the increase in the number of lions explains the increases in predation intensity (Di Silvestre, Reference Di Silvestre2002; Sogbohossou, Reference Sogbohossou2009).
Prey selection
Spotted hyaenas, followed by baboons and then lions, were the predominant predators of livestock. As lions are hunted in the hunting zones it is possible that they occur at lower densities than spotted hyaenas. This, along with the high plasticity of hyaenas, may explain the predominance of hyenas, compared to lions, as livestock raiders (Boydston et al., Reference Boydston, Kapheim, , Watts, Szykman and Holekamp2003). In other areas where lions are at high densities they tend to be important livestock predators (Karani, Reference Karani1994; Kerbis Peterhans & Gnoske, Reference Kerbis Peterhans and Gnoske2002; Patterson et al., Reference Patterson, Kasiki, Selempo and Kays2004). Similarly, when at high densities spotted hyaenas are responsible for a high proportion of attacks (Holmern et al., Reference Holmern, Nyahongo and Røskaft2007). Depredation by baboons, which was relatively important around the Reserve, has rarely been reported to be a significant problem elsewhere (Butler, Reference Butler2000).
Although lions preyed on small livestock they were principally predators of cattle, whereas hyaenas and baboons mainly attacked small stock. This supports the hypothesis that selection of livestock species corresponds to the size of the predator (Caro, Reference Caro1994; Patterson et al., Reference Patterson, Kasiki, Selempo and Kays2004) in accordance with the size of their natural prey (Bodendorfer et al., Reference Bodendorfer, Hoppe-Dominik, Fischer and Linsenmair2006; Hayward, Reference Hayward2006; Bauer et al., Reference Bauer, Vanherle, Di Silvestre and De Iongh2008). Lions nevertheless killed a higher proportion of small stock in Pendjari compared to Waza National Park area in Cameroon (Van Bommel et al., Reference Van Bommel, Bij de Vaate, Boer and Iongh2007), Tsavo ranches in Kenya (Patterson et al., Reference Patterson, Kasiki, Selempo and Kays2004) and around the Serengeti National Park in Tanzania (Holmern et al., Reference Holmern, Nyahongo and Røskaft2007).
Seasonality of predation
Livestock predation usually follows seasonal patterns (Oli et al., Reference Oli, Taylor and Rogers1994; Michalski et al., Reference Michalski, Boulhosa, Faria and Peres2006) although there are some exceptions (Holmern et al., Reference Holmern, Nyahongo and Røskaft2007). We recorded a peak in predation by lions and hyaenas in the late wet season, similar to what has been observed in Tsavo (Patterson et al., Reference Patterson, Kasiki, Selempo and Kays2004). This is presumably explained by the variation in prey dispersal with season. During the dry season wild herbivores tend to concentrate near water sources within the Reserve, where it is probably easier for lions and hyaenas to prey on them (Kays & Patterson, Reference Kays and Patterson2002). As the wet season progresses and water is more readily available, prey populations disperse widely. In areas with low mean prey density it may be easier for predators to prey upon livestock at these times (Hunter, Reference Hunter1952; Ayeni, Reference Ayeni1975; Eltringham et al., Reference Eltringham, Cooksey, Dixon, Raine, Sheldrick, McWilliam, Packer, Coe, McWilliam, Stone and Packer.1999). This also probably explains why attacks on livestock were less important in drier years around Pendjari.
However, the pattern of prey movement in relation to the seasonal availability of water may vary from area to area. For example, around Waza National Park lion attacks were only recorded at villages far away from the Park during the wet season, whereas they occurred in all seasons around villages close to the Park (Van Bommel et al., Reference Van Bommel, Bij de Vaate, Boer and Iongh2007). Thus the season of peak depredation on livestock is seemingly related to prey distribution and availability and distances of villages from a protected area. In regions where attacks peak in the dry season this may be because, subsequent to migration of prey after the rains, livestock become an easy alternative for resident carnivores (Rudnai, Reference Rudnai1979; Karani, Reference Karani1994). Sometimes predation increases during calving as calves are easier to attack than adult cattle (Polisar et al., Reference Polisar, Matix, Scognamillo, Farrell, Sunquist and Eisenberg2003; Michalski et al., Reference Michalski, Boulhosa, Faria and Peres2006).
Seasonal predation patterns were different, however, for baboons, which predated livestock mostly during the dry season. This probably explains the difference between the two road axes in the seasonal distribution of predation; baboon attacks are concentrated along the Tanguiéta–Batia road because of its proximity of the Atacora mountains. The dry season begins in November–December, the period when local people set fires to burn the bush. At this time even the hills, a prime baboon habitat, are burned. Thus it probably becomes increasingly difficult for baboons to feed in the wild. Livestock in villages bordering the Atacora mountains thus become an alternate source of food. New forage after the fires draws the baboons away from the villages. Towards the end of the dry season food becomes scarce again, resulting in baboons again predating livestock. Increased predation by baboons in periods of wild food shortage has also been reported in Uganda (Naughton-Treves et al., Reference Naughton-Treves, Treves, Chapman and Wrangham1998) and in Zimbabwe (Butler, Reference Butler2000).
Husbandry techniques
Husbandry techniques may have a great impact on livestock predation (Robel et al., Reference Robel, Dayton, Henderson, Meduna and Spaeth1981; Oli et al., Reference Oli, Taylor and Rogers1994; Cozza et al., Reference Cozza, Fico, Battistini and Rogers1996; Mishra, Reference Mishra1997; Ogada et al., Reference Ogada, Woodroffe, Oguge and Frank2003; Patterson et al., Reference Patterson, Kasiki, Selempo and Kays2004; Wang & Macdonald, Reference Wang and Macdonald2006; Van Bommel et al., Reference Van Bommel, Bij de Vaate, Boer and Iongh2007; for a different opinion see Graham et al., Reference Graham, Beckerman and Thirgood2005).
In the Pendjari area traditional enclosures, which are low, with sparse branches, and the absence of enclosures in most cases, probably encourage livestock predation (Butler, Reference Butler2000; Mazzolli et al., Reference Mazzolli, Graipel and Dunstone2002; Wang & Macdonald, Reference Wang and Macdonald2006). Improved fences and walls are inexpensive and are sustainable methods of deterring predators (Jackson & Wangehuk, Reference Jackson and Wangehuk2001; Ogada et al., Reference Ogada, Woodroffe, Oguge and Frank2003; Treves & Karanth, Reference Treves and Karanth2003); it would be of value to test them around Pendjari. Dogs are relatively inefficient against predators and also served as prey. Similar cases were reported from around Waza (Van Bommel et al., Reference Van Bommel, Bij de Vaate, Boer and Iongh2007) and Serengeti National Parks, where hyaenas kill dogs (Holmern et al., Reference Holmern, Nyahongo and Røskaft2007). However, guarding dogs and other guarding animals have proved to be successful elsewhere (Marker-Kraus et al., Reference Marker-Kraus, Kraus, Barnett and Hurlbut1996; Bangs & Shivik, Reference Bangs and Shivik2001; Marker, Reference Marker2002; Ogada et al., Reference Ogada, Woodroffe, Oguge and Frank2003; Rigg et al., Reference Rigg, Finďo, Wechselberger, Gorman, Sillero-Zubiri and Macdonald2011). The efficiency of guarding animals probably depends on the size and character of the breed and on the size of the predator to be deterred. In Pendjari dogs were reported to be efficient against jackal and baboon attacks but not against lions or hyaenas.
Other factors
Our results suggest that distance to the Reserve was strongly correlated with predation risk. This is similar to results from Waza National Park (Van Bommel et al., Reference Van Bommel, Bij de Vaate, Boer and Iongh2007), the Serengeti (Holmern et al., Reference Holmern, Nyahongo and Røskaft2007) and Brazil (Michalski et al., Reference Michalski, Boulhosa, Faria and Peres2006; Palmeira et al., Reference Palmeira, Crawshaw, Haddad, Ferraz and Verdade2008). The effect of distance could be related to species. Lions usually stay close to their natural habitat whereas hyaenas often move far from protected areas (Kruuk, Reference Kruuk1972; Hofer & East, Reference Hofer and East1993; Mills & Hofer, Reference Mills and Hofer1998; Holmern et al., Reference Holmern, Nyahongo and Røskaft2007). In Pendjari it was the distance to the Reserve more than the distance to the hunting zone that influenced predation patterns. Thus the Reserve is the main source of wildlife utilized in the hunting zones, which largely function as a sink and thus as a buffer. This pattern has been found in many other areas (Doak, Reference Doak1995; Noss et al., Reference Noss, Dinerstein, Gilbert, Gilpin, Miller, Terborgh, Trombulak, Soulé and Terborgh1999). However, the low density of competitors in hunting zones may attract wildlife and predators, which may then further disperse into villages, creating conflicts. Thus it is debatable whether hunting zones successfully act as buffers.
We expected that factors affecting the integrity of the vegetation and of natural prey populations, such as poaching, illegal grazing and safari hunting, would influence conflicts. However, only the number of herbivores shot annually significantly affected the predation rate. The non-significant impact of illegal grazing and poaching could be related to the relatively low numbers of herders and poachers arrested every year in the Reserve because of the increase in patrolling by the rangers.
Implications for conservation
Our findings suggest that conflicts could be significantly reduced by improving husbandry practices. This includes the construction of predator-proof enclosures and a change in herding practices. The park staff, the Wildlife Office and NGOs working in the area should focus on education. As benefits from wildlife can positively affect attitudes (Oli et al., Reference Oli, Taylor and Rogers1994; de Boer & Baquete, Reference Boer and de & Baquete1998; Conforti & de Azevedo, Reference Conforti and de Azevedo2003; Mishra et al., Reference Mishra, Allen, McCarthy, Madhusudan, Bayarjargal and Prins2003), decision makers and conservationists need to ensure that people receive benefits from the Biosphere Reserve. Local people are already involved in reserve management, and receive 30% of the safari hunting revenues. Further studies, however, would facilitate a better assessment of the impact of these revenues and the determinants of people’s perceptions and attitudes in this area. Although direct financial compensation is an alternative to the augmentation mitigation measures (Michelle & Smirnov, Reference Michelle, Smirnov, Seidensticker, Christie and Jackson1999; Stahl et al., Reference Stahl, Vandel, Herrenschmidt and Migot2001; Wang & Macdonald, Reference Wang and Macdonald2006) this may not be an appropriate approach for a relatively poor country such as Benin where it is already difficult to secure funds for conservation. Any measures applied need to be based on the knowledge of factors that influence local attitudes (Zimmermann et al., Reference Zimmermann, Walpole and Leader-Williams2005) and not just a replication of what is applied elsewhere (Treves & Karanth, Reference Treves and Karanth2003).
Mitigation measures need to be underpinned by a thorough understanding of the socio-ecology and use of space by large predators, which could influence mitigation measures (Stahl et al., Reference Stahl, Vandel, Herrenschmidt and Migot2001). Previous studies, particularly of lions, have shown that conflicts are mostly with problem individuals (Stander, Reference Stander1990; Woodroffe & Ginsberg, Reference Woodroffe and Ginsberg1998; Patterson et al., Reference Patterson, Kasiki, Selempo and Kays2004; Bauer & de Iongh, Reference Bauer and De Iongh2005). To limit the territorial expansion of predators into human settlements around Pendjari Biosphere Reserve investigations are required to identify management actions that need to be conducted in the Biosphere Reserve by the Wildlife Office.
Acknowledgements
Financial support for this work was provided to EAS by the Netherlands Organization for International Cooperation in Higher Education and Research and a Kaplan Graduate Award from Panthera Foundation. We thank the Wildlife Office (Centre National de Gestion des Réserves de Faune) and the Pendjari Project for allowing us to work in Pendjari Biosphere Reserve. We are grateful to A. Gbangboche, R. Glele Kakai and P. Vos for their help with statistical analyses, and to P. Neuenschwander and two anonymous reviewers for their helpful comments.
Biographical sketches
Etotépé A. works to conserve carnivores in West and Central Africa and is interested in lion ecology and behaviour and human–lion conflict in West Africa. Hans H. de Iongh is involved in research on the conservation of large carnivores in West and Central Africa. Brice Sinsin has led several programmes on natural resources and conflicts related to wildlife conservation in West African protected areas for more than 20 years. Geert R. de Snoo works in conservation biology with a focus on the impact of human activities on biodiversity. Paul J. Funston has studied the conservation of large carnivores and behavioural ecology in Africa for the last 20 years.








