No CrossRef data available.
Published online by Cambridge University Press: 15 April 2024
Mineralogical features of two orebodies, lenses (C-3 and C-4), at the central part of the Oktyabr'sky deposit have been investigated. Multidirectional mineralogical zoning in the northern and southern orebodies is shown, confirming the hypothesis of their formation from various magmatic flows, which have individual features and their own modes of formation. The southern C-3 and northern C-4 orebodies differ in their mineralogical associations: C-3 is characterised by a high-sulfur association of sulfides; and C-4 contains minerals with a sulfur deficit (talnakhite, sugakiite). Variations in Fe and Ni ratios in pentlandite are controlled by the volatility of sulfur during ore crystallisation. Direct crystallisation zoning is observed in the disseminated ores of the C-4 orebody (borehole RT-107), where the fugacity of sulfur ($f_{S_2}$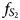 ) increases from bottom to top. In contrast in orebody C-3 (borehole RT-30) $f_{S_2}$
) increases from bottom to top. In contrast in orebody C-3 (borehole RT-30) $f_{S_2}$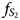 decreases in the same direction. This reverse zoning coincides with the vectors of the evolution of ore systems in various blocks of the Main Ore Body of the Oktyabr'sky deposit. The difference in typomorphic features of disseminated ores of the southern and northern orebodies is confirmed by differences in the associations of platinum-group element minerals (PGMs). Disseminated ores in picritic gabbro-dolerites and massive pyrrhotite ores in the exocontact of the intrusion within the southern orebody differ in the specialisation of PGMs: the former is characterised by minerals of the Pd–Bi–Sb–Te system, the latter by only Pt minerals. The similarity of these types of ores lies in the similar reverse mineralogical and geochemical zoning from top to bottom along the sections, caused by the evolution of the sulfide melt in this direction. The formation of reverse zoning of disseminated ores (zones of ‘marginal reversion’) is probably due to the action of a mechanism of repeated influx of a melt of an increasingly primitive composition into the upper parts of the crystallising flow. Unidirectional trends in massive and disseminated ores are more likely to be due to the action of the same type of mechanism.
decreases in the same direction. This reverse zoning coincides with the vectors of the evolution of ore systems in various blocks of the Main Ore Body of the Oktyabr'sky deposit. The difference in typomorphic features of disseminated ores of the southern and northern orebodies is confirmed by differences in the associations of platinum-group element minerals (PGMs). Disseminated ores in picritic gabbro-dolerites and massive pyrrhotite ores in the exocontact of the intrusion within the southern orebody differ in the specialisation of PGMs: the former is characterised by minerals of the Pd–Bi–Sb–Te system, the latter by only Pt minerals. The similarity of these types of ores lies in the similar reverse mineralogical and geochemical zoning from top to bottom along the sections, caused by the evolution of the sulfide melt in this direction. The formation of reverse zoning of disseminated ores (zones of ‘marginal reversion’) is probably due to the action of a mechanism of repeated influx of a melt of an increasingly primitive composition into the upper parts of the crystallising flow. Unidirectional trends in massive and disseminated ores are more likely to be due to the action of the same type of mechanism.
Associate Editor: Daniel Atencio