INTRODUCTION
More than 100 serotypes of Shiga toxin-producing Escherichia coli (STEC), have been associated with human disease [Reference Johnson, Thorpe and Sears1]. The most well known is E. coli O157:H7, or O157 STEC, known to cause sporadic cases and outbreaks of potentially life-threatening illness [Reference Riley2]. STEC of serogroups other than O157, or non-O157 STEC, are also associated with haemolytic uraemic syndrome (HUS), as well as disease outbreaks in the USA and abroad [Reference Johnson, Thorpe and Sears1, Reference Viljanen3–Reference Brooks10]. Retrospective reports have estimated that 37–50% of STEC infections per year are caused by non-O157 STEC organisms [Reference Brooks11, 12]; additionally, the rate of non-O157 STEC detection increased more than threefold during 2000–2006, probably due to wider use of Shiga toxin (Stx) testing [Reference Gould13]. All suspected and confirmed STEC-related illness, regardless of serotype, is nationally notifiable and reportable in New York State (NYS) [14–16].
Recently, national guidelines for foodborne disease outbreak response have been distributed by the Council to Improve Foodborne Outbreak Response (CIFOR). These guidelines provide a thorough framework for successful preparation for, and investigation and control of most foodborne outbreaks. Specifically, outbreak investigation relies on rapid confirmation of an aetiological agent, often identified through routine laboratory methods [17]. Isolation of an organism is most helpful in epidemiological investigation since it enables further characterization that can be used to detect outbreak strains and ultimately identify a source [17]. While routine methods, such as selective media, which can identify and lead to isolation of O157 STEC have long been available [Reference March and Ratnam18], no selective media has been developed to identify non-O157 STEC routinely. Thus, laboratory surveillance for detection and confirmation of non-O157 STEC disease provides a unique challenge.
Methods such as enzyme immunoassay (EIA) and polymerase chain reaction (PCR) have been developed to detect Stx in patients' specimens [Reference Kehl19, Reference Pulz20], providing clinical diagnostic laboratories (CDLs) with a means to detect non-O157 STEC. However, because of the inability to isolate an organism, and, in the case of EIA, false-positive and false-negative test results [Reference Beutin, Zimmermann and Gleier21, 22], either method alone is inadequate to guide clinical, as well as public health, action [Reference Manning23–Reference Cohen27]. CDC recommends performing, in parallel, testing (i.e. EIA and/or PCR) to identify all STEC and selective-media culture to detect O157 STEC [24, 25]. Nonetheless, recent laboratory surveys have noted infrequent use of any Stx-specific method, and where Stx testing is performed, it is rarely performed together with O157 STEC culture-based methods [Reference Cohen28, Reference Hoefer29]. As a result, non-O157 STEC are probably under-reported, and the role of non-O157 STEC in clinical disease is probably not fully understood.
To optimize detection of non-O157 STEC, the New York State Department of Health (NYSDOH) has developed a systematic approach to identify, confirm, and type cases and clusters of STEC-associated disease in NYS. The system involves coordination of epidemiological and environmental health investigation, CDL testing, and confirmatory testing and identification at the NYSDOH Wadsworth Center Bacteriology Laboratory (WC). Use of this system enables rapid detection, successful identification, and control of disease. We describe two outbreaks of non-O157 STEC disease reported to NYSDOH, one associated with unpasteurized cider consumption, and the other an outbreak of bloody diarrhoea in a prison. In both instances, our enhanced public health approach was implemented to contribute to identification, control, and understanding of non-O157 STEC disease.
METHODS
Epidemiological and environmental health investigations
O157 STEC infections, Stx-positive test results (as presumptive evidence of O157 STEC disease), HUS, and clusters of suspected communicable disease (e.g. bloody diarrhoea) are all reportable in NYS [15, 16]. Upon notification of illness or positive STEC laboratory testing, epidemiology and environmental health personnel convene to initiate an investigation. In general, investigations are initiated for all clusters, and at times for individual cases depending on the setting. For example, a single case of STEC in a vulnerable population, such as in a nursing home or a pre-school, might cause an investigation to be launched. Requests to forward specimens to WC for confirmatory testing were made. Working case definitions were developed by using existing national surveillance case definitions [14]. Enhanced surveillance was conducted to compare disease incidence with baseline rates and identify additional illness. Food preparation reviews and onsite water-supply evaluation were performed where food was prepared or processed. Environmental samples were collected, as available and appropriate. Questionnaires designed by NYSDOH were used to identify risk factors associated with illness. Statistical analysis was conducted with Epi-Info™ 2002 software (CDC, USA) and SAS® version 8.02 (SAS Institute Inc., USA). Appropriate measures were implemented to control the spread of disease.
Outbreak 1
On 28 September 2004, infection-control personnel at a local community hospital notified local health officials of four patients hospitalized with diarrhoeal illness. All four had stool samples with visible blood that were negative for O157 STEC by latex agglutination. The local health department began an investigation and identified through routine surveillance interviews that all affected persons had consumed products processed at a local orchard. NYSDOH environmental health staff were notified immediately and a coordinated investigation was launched.
A confirmed STEC case was defined as (1) diarrhoea (⩾3 loose stools/24 h) in a person with a history of visiting or consuming foods produced at a specific orchard during 1 September–15 October 2004, and (2) a positive laboratory test for Stx only in stool or for Stx and O111 STEC. A confirmed case of cryptosporidiosis met criterion (1) and had a positive laboratory test for Cryptosporidium parvum. An unmatched case-control study of both STEC and cryptosporidiosis cases was conducted to identify exposures and behaviours associated with disease. Control subjects were well persons identified by random-digit dialling in case-patients' home area codes, who had visited or consumed products from the same orchard during the same period.
Outbreak 2
On 8 September 2005, the New York State Department of Correctional Services (NYSDOCS) notified NYSDOH of an outbreak of bloody diarrhoea involving inmates at a NYSDOCS correctional facility. According to the facility's infection control nurse, stool from at least three inmates had tested Stx-positive. The facility was a large maximum-security prison that included a special care unit for inmates with chronic medical conditions, such as end-stage renal disease. Given the sensitive nature of the population, a coordinated investigation was launched.
A confirmed STEC case was defined as (1) diarrhoea (⩾3 loose stools/24 h) in an inmate during 29 August–15 September 2005, and (2) laboratory evidence of Stx only in stool or for Stx and O45 STEC. A suspected case met criterion (1) only. A cohort study was conducted among prisoners of affected units to identify exposures and behaviours associated with disease.
Laboratory investigations
Stool specimens were initially screened by commercial or hospital clinical microbiology laboratories, according to their routine protocol. Screen positive broth specimens or stool specimens submitted to WC were tested for the presence of Shiga toxin genes (stx1 and/or stx2) through real-time PCR (Stx-PCR) and plated directly onto sorbitol MacConkey agar (SMAC) and cefixime-tellurite SMAC agar and incubated overnight at 37°C. Sorbitol-fermenting organisms isolated were subcultured and tested by Stx-PCR until a single Stx-positive organism was isolated. Stx-positive isolates were identified as E. coli biochemically and tested to determine the serogroup using either latex agglutination (Oxoid, UK) or antisera tube agglutination (Statens Serum Institut, Denmark). Testing of food, environmental, and bovine stool samples was accomplished by screening samples with Stx-PCR, followed by immunomagnetic separation with anti-O-antigen antibody-coated particles (Invitrogen Dynal AS, USA) and culturing as described previously [Reference Bopp30]. Isolates that were unable to be serogrouped at WC were forwarded to CDC for analysis. Media composition, biochemical and serological identification, and DNA extraction and Stx-PCR procedures have been described previously [Reference Bopp30]. Isolates were characterized by pulsed-field gel electrophoresis (PFGE) by using standardized protocols [Reference Ribot31].
RESULTS
Outbreak 1
The epidemiological investigation identified a total of 213 persons who became ill. Of the 27 confirmed STEC cases, 26 (96%) had specimens that yielded stx1-positive O111 STEC with an identical PFGE pattern. The remaining specimen was Stx-PCR positive for stx1, but no STEC was isolated by culture (see Table 1). Twelve of the STEC cases also had laboratory evidence of C. parvum infection; 31 persons had C. parvum infection only [Reference Coronado32]. The orchard and its cider were a popular public attraction; proprietors estimated thousands of persons might have visited the orchard or consumed the unpasteurized cider. An unmatched case-control study implicated consumption of unpasteurized apple cider as the source of the outbreak. Those who had consumed cider produced at the orchard had 13 times greater odds of illness than those not exposed to the cider (odds ratio 12·9, 95% confidence interval 3·4–49·9). No other food item or contact with animals at the orchard was significantly associated with illness.
Table 1. Epidemiological characteristics of STEC outbreaks
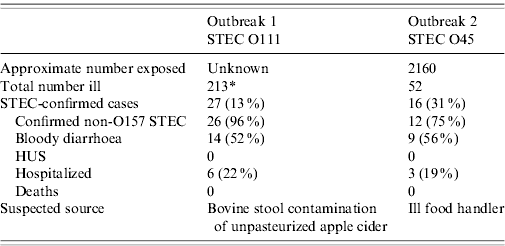
* Includes suspected and confirmed cases of STEC and cryptosporidiosis.
Environmental evaluation of the orchard and cider-production facility was conducted. The cider press was located within 60 ft of a pen containing three calves that served as an attraction for cider customers. One employee responsible for feeding the calves was also a cider press operator; gloves or other personal protective equipment were not used for either task. E. coli was cultured from the cider press, but no STEC was identified. Cider samples collected from case-patients' homes and the orchard were negative for STEC by culture and PCR. Two out of three stool samples collected from the calves and analysed at WC were PCR-positive for stx1, but no STEC colonies were isolated. Public health interventions included ceasing production of and discarding cider present at the orchard. Additionally, press releases and NYSDOH alerts were distributed to notify the public and providers of the outbreak, to request reporting of any illness associated with cider consumption, and to recommend discarding any cider purchased at the orchard.
Outbreak 2
Initial reports included stools from 18 patients tested at two separate CDLs; stools from three (17%) patients were EIA-positive for Stx, and 15 (83%) were culture-negative for O157 STEC. Stools from both CDLs, as well as primary specimens from patients, were forwarded to WC for Stx-PCR and further characterization [24]. Stools from 12 (66%) of the 18 patients grew O45 STEC that was indistinguishable by PFGE; four (22%) stools were Stx-PCR positive for stx1, but yielded no STEC isolates, and two were negative. In addition to these 16 confirmed cases, the ensuing epidemiological investigation identified 36 suspect cases (total ill: 52/2160 inmates, attack rate: 2·4%) (see Table 1). A cohort study failed to identify any food items or common exposures significantly associated with illness. Thirteen (81%) confirmed case-patients had received meals that were delivered by inmate food handlers to their unit or cell rather than dining in the prison cafeteria. The same food items had also been served in the cafeteria. An inmate food handler involved in preparation of the delivered meals reported working while ill immediately before the outbreak, but refused to provide a specimen for testing. Although the correctional facility operated a small dairy farm immediately outside the facility, no illness was identified among prisoners who had direct contact with cows. A subset of prisoners had worked with cows and had no contact with the general prisoner population, living in a dormitory located outside the wall-enclosed prison. Food and environmental samples from the prison cafeteria, dairy farm, and dormitory were negative for STEC. Stool samples from dairy cows were PCR positive for both stx1 and stx2, but negative for O45 and O157 STEC. No serious deficiencies were identified after review of food-preparation methods. Public health interventions included enhanced surveillance for bloody diarrhoea and STEC within the correctional system statewide, and reinforcement of correct food-preparation procedures at the correctional facility.
DISCUSSION
These two outbreaks demonstrate successful coordination of public health notification and response in identifying instances of non-O157 STEC disease. The NYSDOH system of early, close collaboration between CDL and public health laboratorians, epidemiologists, and environmental health staff is efficient in identifying both O157 STEC and non-O157 STEC. This allows detection, appropriate control-measures implementation, cessation of disease transmission, and characterization of non-O157 STEC disease and organisms.
In outbreak 1, consumption of unpasteurized apple cider contaminated by cows' faeces was the probable source of illness. This hypothesis was supported by the identification of both O111 STEC and C. parvum, both known colonizers of the bovine intestine [33], along with the investigative environmental findings. Occurrence of new cases decreased after closure of the cider press. Bovine faecal contamination of unpasteurized apple cider has been reported, particularly with O157 STEC [33]. Additionally, O111 STEC has been associated with outbreaks of bloody diarrhoea and HUS [Reference Johnson, Thorpe and Sears1, 5, Reference Banatvala6, Reference Brooks10]. The outbreak's source was not confirmed, but environmental samples might have been negative because of low-level or intermittent contamination.
In outbreak 2, a source of the STEC could not be identified. The ill food handler might have contaminated the delivered food. Contamination could not be verified because the ill food handler was not tested, and the food he had helped to prepare was not available for testing. The dairy cows located immediately outside the correctional facility were an unlikely source of illness because no illness occurred among those having direct contact with cows. O45 STEC has been identified as a cause of sporadic cases of bloody diarrhoea [Reference Johnson, Thorpe and Sears1]. This is the first known outbreak of O45 STEC; outbreak propagation was most likely related to the confined nature of the population and the common source of food rather than the relative pathogenicity of the organism.
The potential of non-O157 STEC to cause outbreaks as well as severe complications of infections (e.g. HUS) is well documented [Reference Johnson, Thorpe and Sears1, Reference Viljanen3–Reference Brooks10]. However, reliable straightforward methods to isolate non-O157 STEC have lagged in availability. PCR targeting Stx genes is an effective means of differentiating non-O157 STEC from commensal flora [Reference Pulz20], but is not widely used by CDLs. Rather, CDLs that screen for Stx have adopted EIAs or continue to use selective-media culture for O157 STEC exclusively [Reference Johnson, Thorpe and Sears1, Reference Cohen28, Reference Hoefer29]. CDC has recommended that CDLs screen all stool samples for Stx and O157 STEC and forward all Stx EIA-positive, O157 STEC-negative (along with O157 STEC) specimens to a public health laboratory for confirmation and organism isolation [25]; since 2009, this procedure has been mandatory in NYS [34].
The responsibility for identification of non-O157 STEC disease does not rest with laboratories alone; collaboration is key to successful surveillance, detection, and response. The CIFOR guidelines support, upon identification of an outbreak, the establishment of an outbreak investigation team with clear leadership, and define specific roles for all team members [17]. In NYS, the default leader is an epidemiologist who is responsible for overseeing all aspects of an outbreak investigation. Epidemiologists and sanitarians at all levels are instrumental in coordination of efforts, and WC uses information provided by epidemiologist and sanitarian colleagues to prioritize testing and best utilize limited resources.
An important distinction must be made between singular, seemingly sporadic, cases and clusters and outbreaks. The CIFOR guidelines are designed to be implemented once an outbreak is identified. This is a rational approach to most foodborne disease, especially that which is well understood and readily confirmed in the laboratory. However, non-O157 STEC is not as well understood or easily identified given current CDL practice [Reference Cohen28, Reference Hoefer29]. In NYS, a coordinated investigation is more likely when a cluster of illness is identified. While not every positive STEC result may lead to instigation of a coordinated investigation, every case does receive some level of inquiry at the local and/or state level in order to triage for an appropriate response. Timely response is instrumental in successful outbreak control. We believe that thorough investigation of all STEC-positive reports probably enables not only early outbreak detection, but a more informed response. By having the information and possibly samples on hand at the outset, aetiological confirmation or source identification is not delayed as it may otherwise be [17]. Such careful surveillance and coordinated response need not be limited to STEC, and are applicable to cases and outbreaks of other foodborne illness, although different laboratory and epidemiological strategies are likely to be applied.
Healthcare providers have an essential role in detection and study of non-O157 STEC disease. CDC recommends that clinical providers request both Stx testing and culture for O157 STEC, especially with a history of bloody diarrhoea, and that CDLs strongly consider including O157 STEC in their routine bacterial enteric panel and be able to test simultaneously for Stx by screening method and O157 STEC by culture [25]. CDLs may not test for STEC except under certain circumstances (e.g. only if specifically requested to do so) [Reference Voetsch26, Reference Hoefer29]. In both NYS outbreaks, notification and response were initiated only when laboratory-testing results of clinical specimens were available and not when suspicious illness was identified. Had clinicians reported the suspected STEC disease when they had ordered tests, the public health response might have been initiated sooner. This would have lead to faster confirmation, more timely removal of the contaminated food products, and possible prevention of disease in outbreak 1. Furthermore, reporting delay caused environmental sample collection after the suspected contaminated products were no longer available. This probably prevented definitive determination of causation that might help guide future public health policy and clinical recommendations.
Clinical providers should not discount their responsibility to participate in the public health process. NYSDOH encourages providers to report all cases of acute bloody diarrhoea, including suspected cases of STEC infection, given their likely aetiology of reportable, disease-causing agents. Timely identification of STEC disease is crucial to appropriate clinical management [Reference Johnson, Thorpe and Sears1]. Specifically, the decision to prescribe antibiotics or anti-motility agents, although appropriate in certain enteric infections, might cause an adverse outcome in STEC disease [Reference Wong35, Reference Bell36]. Furthermore, progression of STEC infection to HUS might be avoided by aggressive fluid resuscitation, which might not be appropriate for other enteric infections [Reference Ake37]. We report that close collaboration between a variety of public health professionals has enabled identification of non-O157 STEC in settings not described previously. These professionals include, but are not limited to, local and state epidemiologists, environmental health staff, state public health laboratorians, and local clinical and commercial laboratorians. The addition of healthcare providers to this collaboration will better serve individual patients and ultimately enhance protection of the public's health.
ACKNOWLEDGEMENTS
The authors thank members of the Dutchess County Department of Health, Clinton County Health Department, New York State Department of Agriculture and Markets, New York State Department of Correctional Services, Wadsworth Center and Metropolitan Area and Capital District Regional Offices, New York State Department of Health, and Enteric Diseases Laboratory Branch, Centers for Disease Control and Prevention who helped to investigate these two outbreaks.
The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention.
DECLARATION OF INTEREST
None.



