INTRODUCTION
Non-typhoidal Salmonella causes an estimated 1·2 million infections, 19000 hospitalizations and 370 deaths annually in the United States [Reference Scallan1]. Although the majority of human Salmonella infections are acquired by the foodborne route, people may also acquire salmonellosis from contact with infected animals. Eleven percent of domestically acquired non-typhoidal salmonellosis is attributable to animal contact [Reference Hale2]. Non-typhoidal Salmonella is responsible for more hospitalizations (48%) and deaths (62%) than any other enteric zoonoses, and children are disproportionately more affected [Reference Hale2].
Poultry, a natural reservoir for Salmonella, can become infected or colonized through both vertical transmission from infected hens [Reference Liljebjelke3] and horizontal (environmental) transmission [Reference Loharikar4, Reference Roy5]. Horizontal transmission may arise from other birds, the nearby environment, feed, rodents, insects, and other sources. Human salmonellosis linked to direct and indirect contact with poultry – including chickens, ducks, turkeys, and geese – is a rising public health concern [Reference Loharikar4, Reference Barton6–Reference Wilkins11], probably because of the increasing popularity of backyard flocks [Reference Barton6]. Between 1996 and 2012, 45 outbreaks of human salmonellosis linked to poultry exposure were reported to the Centers for Disease Control and Prevention (CDC), resulting in >1581 illnesses, 221 hospitalizations, and five deaths [Reference Barton6]. In 2012, eight outbreaks of human salmonellosis linked to live poultry contact were investigated resulting in 517 laboratory-confirmed illnesses, 90 hospitalizations, and three deaths (CDC, unpublished data). By contrast, only two outbreaks of human Salmonella infections linked to live poultry occurred in each year from 2003 to 2005 [Reference Barton6]. Because poultry infected with Salmonella may appear healthy but still shed bacteria that may lead to human illness [Reference Barton6], heightened awareness of this emerging infection source is necessary.
To serve a growing demand, 20 mail-order hatcheries (MOHs) produce and sell ~50 million baby poultry each year in the United States [Reference Gaffa7]. Within 24 h of hatching, baby poultry (e.g. chicks, goslings, ducklings) are shipped from hatcheries through the United States Postal Service in cardboard boxes containing up to 100 birds each. Birds can typically be purchased in agricultural feed stores or via the internet.
The MOH industry is encouraged to maintain environmental hygiene standards based on guidance from the United States Department of Agriculture (USDA), Animal and Plant Health Inspection Service (APHIS), Veterinary Services (VS), National Poultry Improvement Plan (NPIP). The NPIP is a voluntary partnership between industry, state and federal government, with the goal of eliminating specific poultry diseases from breeder flocks to prevent egg-transmitted and hatchery-disseminated diseases [12]. The NPIP does not certify chicks shipped from hatcheries as free from all Salmonella strains that cause human illness [Reference Barton6].
We report the findings of a multistate outbreak of human Salmonella enterica serotype Braenderup (S. Braenderup) infections linked to poultry in backyard flocks sourced from one MOH in the southern United States. Our results illustrate how partnerships with the MOH industry provided epidemiological and environmental data that ultimately led to the MOH source that supplied poultry linked with this outbreak. Furthermore, we characterize poultry distribution and business practices in the MOH industry. Finally, findings from this investigation demonstrate and emphasize the need for a ‘One Health’ approach that leverages expertise in human, animal, and environmental health to address this complex problem at the human–animal–environment interface, to prevent additional human illnesses and outbreaks [13].
METHODS
Detection of the outbreak
As part of routine national surveillance, isolates of strains of Salmonella species obtained from clinical specimens of ill persons are forwarded to state public health laboratories for serotyping and subtyping analysis by pulsed-field gel electrophoresis (PFGE). The PFGE patterns are then electronically submitted to PulseNet, the national molecular subtyping network for foodborne disease surveillance, and catalogued in a database. PulseNet database managers monitor the database for clusters of illnesses and report them to foodborne disease epidemiologists at the CDC.
On 1 October 2012, PulseNet identified a cluster of 14 cases of S. Braenderup infections with identical PFGE patterns (XbaI pattern JBPX01·0767) submitted from 10 states. This particular pattern of S. Braenderup is rare, and was only seen 13 times prior to this cluster identification. CDC and state and local public health officials initiated a multistate investigation to determine the source of this outbreak.
Case definition and finding
A case was defined as a diarrhoeal illness in a person residing in the United States, with onset from 25 July 2012 to 27 February 2013 with culture-confirmed S. Braenderup infection yielding PFGE pattern XbaI pattern JBPX01·0767 (the outbreak strain). Throughout the investigation, PulseNet notified epidemiologists of S. Braenderup isolates matching the outbreak strain newly uploaded to the database.
Patient interviews
Ill persons were interviewed by state and local health officials using routine state enteric disease questionnaires to identify hypotheses pertaining to potential sources of infection, including food, animal, and other exposures commonly associated with enteric illness outbreaks. CDC epidemiologists compiled data on ill persons from all states and analysed them for common exposures. Once contact with live poultry became the primary hypothesis, ill persons were interviewed using a questionnaire developed by CDC and APHIS (CDC-APHIS-VS live poultry questionnaire). This questionnaire asked about poultry-related exposures in the 7 days before illness onset, including types and breeds of poultry contacted, types of exposures, and poultry-handling practices. Study investigators entered data from questionnaires into electronic databases (Microsoft Excel 2010, Microsoft Access 2010), and descriptive statistics and frequencies were calculated. The binomial test was used to compare expected frequencies (based on interviews conducted in a national population-based survey [14]) with observed frequencies. Results with P < 0·05 were considered statistically significant.
Traceback investigation
To trace live birds back to hatcheries of origin, state and local public health officials and CDC collected information on sources of poultry in households of ill persons. Nine MOH owners were interviewed by state public health and NPIP partners using a specific MOH industry questionnaire developed to facilitate the traceback investigation, identify the original poultry source, and better characterize MOH industry practices such as drop-shipping, comingling, and multiplying that may complicate outbreak investigations. Drop-shipping describes the situation in which one hatchery is not able to complete a customer order itself, and requests a second hatchery to fill the order on its behalf. In this situation the customer receives a shipment of birds directly from the second hatchery under the first hatchery's name; birds from the second hatchery never actually enter the first hatchery's facilities. Comingling is the situation in which a hatchery will receive eggs for hatching or birds from a second hatchery and add them to its own flock, so that a bird's original source is obscured. With the business practice known as multiplying, a hatchery receives from another facility hatching eggs to produce breeder birds, or receives breeder birds themselves. The breeder birds can then be used to produce more eggs for hatching and distribution.
Environmental investigation
Periodic environmental swabbing to test for Salmonella species by serogroup is conducted by MOHs on a self-determined basis. Specimens were collected monthly from poultry, poultry trays, and the environment of the suspect hatchery from June 2012 to January 2013 and were sent to a state veterinary diagnostic laboratory for culture and serotyping. As part of this investigation, Salmonella isolates identified from the suspect hatchery at the state veterinary diagnostic laboratory were forwarded to the USDA-APHIS-VS National Veterinary Services Laboratories (NVSL) for repeat serotyping and PFGE analysis.
Human subjects considerations
As part of the emergency public health response to this outbreak, this investigation did not require human subjects review. All interviewees provided verbal informed consent.
Ethical standards
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008.
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional guides on the care and use of laboratory animals.
RESULTS
Case finding
Forty-eight laboratory-confirmed cases from 24 states met the case definition (Fig. 1). Illness onset dates ranged from 25 July 2012 to 27 February 2013 (Fig. 2). The median age of patients (n = 48) was 27·5 years (range <1–88 years), 29 (60%) were female, and 15 (31%) were children aged <10 years. Of all patients for whom clinical data was reported, seven (22%) were hospitalized; no deaths were reported.
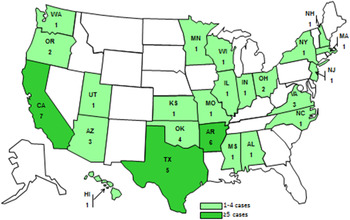
Fig. 1. Location and number of case-patients infected with the outbreak strain of Salmonella Braenderup, by state, July 2012–February 2013
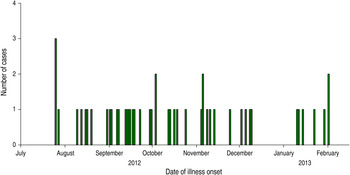
Fig. 2. Number of cases of human Salmonella Braenderup infections with the outbreak strain, by date of illness onset, United States, July 2012–February 2013
Interviews
Thirty-three ill persons were initially interviewed by state and local health departments using standard enteric disease questionnaires. Twenty-six of 32 (81%) interviewed ill persons reported contact with live poultry in the week before illness (one person could not recall), which is significantly higher than the ~2·5% of healthy persons that reported live chicken exposure in the week prior to interview in a national population-based survey (P < 0·001) [14]. This was the only statistically significant result.
Twenty ill persons (or their parents/guardians) were interviewed on details of their live poultry exposure using the CDC-APHIS-VS live poultry questionnaire. Table 1 summarizes the results of the interviews. Thirteen feed stores and 12 MOHs of the 20 core MOHs nationwide in eight states were named as sources of poultry. While one hatchery, which we refer to as hatchery D, was linked through interviews to more cases (n = 12, 63%) than any other mentioned hatchery, the remaining ill persons reported purchasing their poultry from feed stores or hatcheries apart from hatchery D.
Table 1. Exposures in ill persons with Salmonella Braenderup infection interviewed with the CDC-APHIS-VS live poultry questionnaire (n = 20), United States, July 2012–February 2013
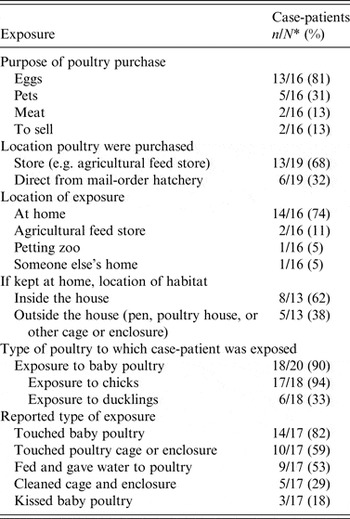
* Denominators differ as a result of missing data.
Traceback investigation
The results of the MOH industry questionnaire provided information on hatchery D distribution chains. We combined this information with the results of interviews of ill persons to determine the lines of sale pertinent to this investigation, as illustrated in Figure 3. We discovered that all interviewed cases could ultimately be linked to hatchery D, based on complex lines of sale and distribution between hatcheries and feed stores, ultimately to consumers (Fig. 3).
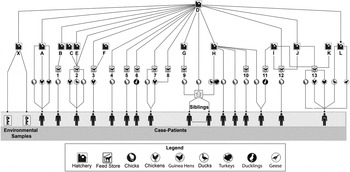
Fig. 3. Traceback investigation: distribution from ultimate source hatchery to interviewed ill persons, United States, July 2012–February 2013. (Distribution chain flows from top to bottom. Hatchery D supplied poultry to or drop-shipped poultry for eight hatcheries: X, A, B, E, G, H, J, and L, and supplied poultry directly to eight feed stores: 2, 3, 4, 5, 6, 7, 8, and 12. Other hatcheries in turn supplied poultry to feed stores, and ill persons received poultry from hatcheries or feed stores, respectively. For example, hatchery B supplied poultry to feed store 1; hatcheries C, D, and E all supplied poultry to feed store 2, etc. Data were available for only 16/48 case-patients.)
The questionnaire administered to the MOH business owners captured poultry distribution and business practices in the industry, including the proportion of hatcheries participating in drop-shipping and comingling, current Salmonella prevention measures, and staff and consumer knowledge and education, as summarized in Table 2.
Table 2. Mail-order hatchery characteristics, from the mail-order hatchery industry questionnaire (n = 9), United States, July 2012–February 2013

* A few commercial Salmonella vaccines for poultry are available; they may vary based on which serogroups or serotypes are included. Although two of six hatcheries reported vaccinating birds against Salmonella, the likelihood that their vaccines contained strains of S. Braenderup is small.
Environmental investigation
Periodic environmental sampling at hatchery D conducted by the hatchery owner revealed S. Braenderup matching the outbreak strain in samples taken from hatchlings and hatchling trays in August, September, and October 2012; from a sample of crickets found outside of the hatchery in August 2012 after a coincident increase in the cricket population; and from the floor of a poultry house in December 2012, thus providing additional evidence that hatchery D was involved in the outbreak.
Routine environmental sampling by a hatchery in the northeastern United States, which we refer to as hatchery X, identified the outbreak strain. Although hatchery X was not named by any of the interviewed ill persons, hatchery interviews revealed that hatchery X received breeder birds directly from hatchery D for the purposes of multiplying, and conversely did not sell eggs or birds to hatchery D.
Public health action
Prior to discovery of hatchery D's connection to this outbreak, the owner had begun implementing improvements in sanitation procedures and insect control in August 2012 based on routine environmental testing results having identified Salmonella. The improvements included insecticide spraying, new steps in egg disinfection, and the addition of another spray vaccination to the existing vaccination programme. After the samples were confirmed to be S. Braenderup matching the outbreak strain, APHIS-VS, including NPIP, CDC, state health departments, state agriculture departments, and MOH staff collaborated to develop additional appropriate hatchery-level interventions. Hatchery D further enhanced sanitation and disinfection measures and prepared for the usage of an autogenous vaccine specific to the outbreak strain of Salmonella and other Salmonella serotypes found in the hatchery's flock. Hatchery D continued to work with state public health and agriculture partners and conduct monthly sampling and monitoring for Salmonella. Thirty-two samples conducted from December 2013 to March 2014 yielded no S. Braenderup.
DISCUSSION
We investigated a multistate outbreak of S. Braenderup infections with onsets occurring between July 2012 and February 2013. The origin of the implicated poultry was traced back to one MOH, hatchery D, through a combined epidemiological, laboratory, and traceback investigation.
Because of an unexpected increase in the cricket population in and around hatchery D, suspected to be secondary to an unusual summer weather pattern (Michael Merchant, Texas A&M AgriLife Extension Service, written communication), the owner of hatchery D took samples of crickets found just outside the hatchery in August 2012. These tests revealed the outbreak strain. This population increase may have triggered the outbreak if the crickets were vectors to introduce the outbreak strain of Salmonella into the hatchery. Alternatively, the crickets may simply have tracked through areas of the hatchery contaminated by the S. Braenderup-infected poultry, picking up the outbreak strain en route.
In contrast to past live poultry-associated Salmonella outbreaks, this outbreak was unique in that 12 (60%) of the 20 core MOHs nationwide were named as sources of infected poultry, emphasizing a new complexity in the investigation of outbreaks of human illness linked to backyard flocks. Complicated business practices such as drop-shipping and comingling obscure identification of the hatchery from which a particular shipment of birds originated. This investigation highlights the importance of understanding such business practices in discovering the source of an outbreak, and the results of the MOH industry questionnaire suggest how prevalent these practices are in the industry. Through tracing back poultry distribution chains, with the knowledge of drop-shipping as a potential practice, we discovered hatchery D to be the ultimate source, directly or indirectly, of birds bought by all interviewed ill persons. Although hatchery X was not named by any ill person as a source of birds, its environmental sampling revealed the outbreak strain, and it was subsequently found to have received breeder birds from hatchery D. This hatchery culled their infected birds after the outbreak strain was detected. The distribution of eggs and birds through multiple MOHs and the interdependence of the industry emphasize the importance of comprehensive Salmonella prevention and control programmes being implemented and maintained at all MOHs and associated breeder farms.
This investigation showed the importance of industry involvement in the investigation of an outbreak. Cooperation by MOH owners and staff was critical in solving this complex investigation. First, the results of the MOH industry questionnaire helped to determine the prevalence of unique business practices such as drop-shipping. In addition, poultry hatcheries periodically conduct environmental sampling throughout their hatcheries. The owner of hatchery D shared environmental sampling results with the CDC, which assisted in identifying hatchery D as the source of poultry linked with this outbreak. Continuing to partner with industry will aid in future outbreak investigations and appropriate interventions to prevent the spread of salmonellosis associated with MOHs.
Results of the MOH industry questionnaire showed while hatcheries are using disinfection procedures to reduce Salmonella, room for additional intervention and monitoring exist, such as environmental testing and vaccine use. Questionnaire results further suggest although hatchery owners are aware of the risks of Salmonella infection associated with poultry, several have yet to translate that knowledge into education to staff and consumers. CDC has convened discussions with the MOH industry to remind owners of the health risks associated with poultry contact; advise them on ways to educate their staff and customers; and enhance collaborations between the CDC, USDA-APHIS, local and state public health practitioners, and industry partners to develop and maintain comprehensive strategies to prevent and control Salmonella infection from poultry.
Of note, CDC investigated a second outbreak of the same strain of S. Braenderup from 6 May to 25 September 2013, including a total of 53 cases reported from 21 states. Thirteen (76%) out of 17 interviewed ill persons reported having exposure to live poultry in the 7 days before illness onset. The birds of 5/8 (63%) interviewees could be traced back to hatchery D. Hatchery D was notified and continued implementation of additional corrective measures. Hatchery D continues to cooperate with state public health and agriculture partners, CDC, and NPIP.
An integrated approach to preventing human Salmonella infections from contact with poultry should target MOHs, hatchery breeder flocks, agricultural feed stores, and backyard flock owners. Interventions, if implemented and maintained at all levels, could help reduce the number of human Salmonella infections. The NPIP provisions currently provide a voluntary Salmonella monitored certification programme for MOHs [Reference Barton6]. Hatcheries may choose to participate, agreeing to comply with management and sanitation practices outlined by the NPIP and enhance control of Salmonella serotypes that cause human illness [Reference Basler15]. In addition, hatcheries may refer to the Best Management Practices Handbook, published by APHIS-VS in March 2014 [16]. Biosecurity, sanitation, and quality assurance recommendations in the handbook will assist hatchery operators in reducing Salmonella contamination at their hatcheries, likely reducing the number of live poultry-associated human Salmonella infections. Feed stores and others who sell or display poultry should disseminate educational messages to consumers on the risk of illness and simple steps to reduce the risk. They should post signs requesting consumers not to touch poultry on display. They may consider offering hand washing stations or hand sanitizers next to poultry display areas instructing customers to wash hands immediately after leaving the poultry display area. Displaying birds out of reach of customers, especially children, and cleaning and disinfecting bird display enclosures between shipments of birds would further prevent the spread of disease.
At the consumer level, all persons should wash their hands with soap and warm water after touching poultry or surfaces in contact with poultry. Live poultry should not be kept in the house, especially in areas where food or drink are prepared, served, or stored, and all surfaces that come into contact with poultry (e.g. tables, floors, bird enclosures) should be properly cleaned and disinfected because they may be contaminated with Salmonella. Additional recommendations for consumers, feed stores, and MOHs, as well as free educational materials, are available from CDC [17–20].
Our investigation is subject to several limitations. For the enteric disease and live poultry questionnaires, ill persons or their proxies were asked to recall contact histories preceding their onset of illness yet were interviewed 1–4 weeks after their illness, introducing a possible recall bias. Further, only nine of the 13 involved hatcheries participated in interviews with the MOH industry questionnaire. A response bias may have been introduced by the small sample size of hatcheries.
This investigation illustrates the interconnectedness of humans, animals, and the environment and suggests a multidisciplinary approach, referred to as a ‘One Health’ approach [13], to addressing human illness linked to poultry that leverages expertise in human, animal, and environmental health. Consideration of how diseases spread among people, animals, and the environment in emerging zoonotic human illness outbreaks will enhance outbreak investigation and offer new avenues for disease control and prevention. Although the root cause of this outbreak remains unknown, this investigation highlights the importance of a ‘One Health’ approach to prevent additional human illnesses.
ACKNOWLEDGEMENTS
The authors thank state and local health departments, state departments of agriculture, the USDA-APHIS, and the owner of hatchery D for their collaboration and efforts on this investigation. Thanks are also due to Jim Walters for assistance with graphic design of Figure 3; and Michael Merchant for his communication.
This work was supported by Epi-Aid 2013–027.
DECLARATION OF INTEREST
None.








