INTRODUCTION
The control and eradication of highly pathogenic avian influenza (HPAI) have so far relied on the stamping-out of infected animals and biosecurity measures including quarantine, surveillance and hygiene. Strategies for the control of low pathogenic avian influenza (LPAI) viruses have varied from no action to control with vaccination [Reference Swayne1]. However, AI vaccination is prohibited or not used in most AI-affected countries (Table 1). To limit the risk of introducing the disease, the exportation of vaccinated animals was prohibited because up to recently it was impossible to differentiate infected from vaccinated animals [2].
Table 1. Vaccination status of countries infected with H5N1 avian influenza virus

Less than 50% of contaminated countries are authorizing vaccination as a control tool and less than 30% have started using it. [Source: OIE: (http://www.oie.int/downld/AVIAN%20INFLUENZA/A_AI-Asia.htm), January 2007.]
H5N1 HPAI outbreaks have occurred in both wild and domestic birds in some Asian countries including People's Republic of China (P.R. China), Vietnam and Indonesia, and millions of birds which might represent the only source of food supply and/or income for many households in developing countries have been culled. Measures such as culling, stamping-out, cleaning and disinfection that have been effective in Europe have not been successful in eradicating the disease in Asia [Reference Sims3]. The World Organization for Animal Health (OIE) and the United Nation's Food and Agricultural Organization (FAO) do not recommend mass culling in developing countries because such a practice is no longer acceptable for ‘ethical, ecological and economic reasons’ [Reference Buttler4]. Moreover, more countries are at a high risk of H5N1 introduction due to the existence of commercial routes and migratory waterfowl flyways between South East Asia, the Middle East, the Far East and Europe [Reference Yee, Carpenter and Cardona5–Reference Munster7]. However, the potential role of migratory birds in spreading the H5N1 virus remains controversial [Reference Wang8, Reference Causey and Edwards9]. Emergency (e.g. ring vaccination during an outbreak), preventive (e.g. at-risk birds or specific zone, free of disease or epidemic situation) or prophylactic vaccination (e.g. mass vaccination in an endemic situation), currently allowed in some countries (Table 1) should be used with effective biosecurity measures and thorough surveillance and tracing systems to better control and/or eradicate AI outbreaks [10]. The aim of the present study is to present practical information on the AI vaccination campaigns which have been implemented to date in developing countries, especially in South East Asia, covering the type of vaccine used, the vaccination strategy followed, the associated biosecurity measures and the outcome of each vaccination programme. The state of the art on AI vaccines and vaccination strategies will be described in the first two sections, with a focus on constraints of using AI vaccination in developing countries. For more in-depth information on methods to control AI and on the immunology behind AI vaccines, see the recent review by Capua & Marangon [Reference Capua and Marangon11].
AVIAN VACCINES
The ideal vaccine should be potent, safe, stable at room temperature, administered in a single dose and cheap. It should also enable differentiation between vaccinated and infected animals (DIVA). None of the AI vaccines currently licensed in the field meet all these requirements (Table 2).
Table 2. Review of the advantages and limits of avian influenza vaccines currently licensed on the market against criteria for an ideal vaccine
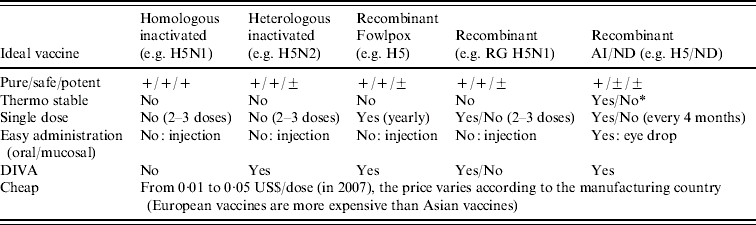
RG, Reverse genetics; AI, avian influenza; ND, Newcastle disease; DIVA, differentiation of infected from vaccinated animals.
* Depending on the ND virus strain.
There are currently two types of AI vaccines in use: inactivated whole AI virus (AIV) vaccine and live recombinant vaccines [Reference Suarez and Schultz-Cherry12]. Table 3 presents a non-exhaustive list of AI vaccines available on the market with information on their efficacy and reference to laboratory and/or field validation.
Table 3. Experimental validation of commercial inactivated and recombinant avian influenza vaccines
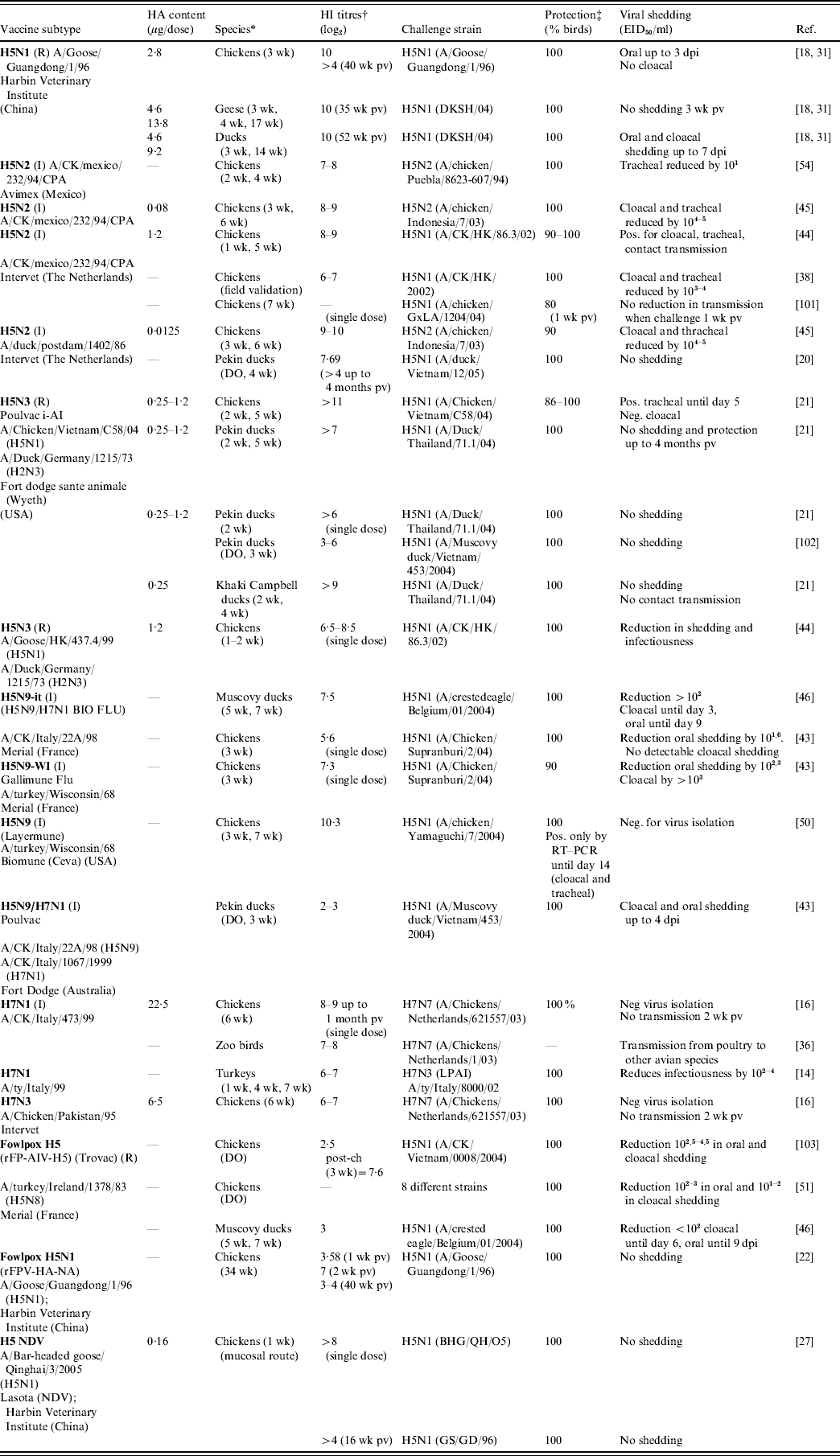
—, No information; AIV, avian influenza virus; CK, chicken; dpi, days post-infection; DO, day old; EID50, egg infectious dose (lethality 50%); FPV, fowlpox virus; HA, haemagglutinin; HI, haemagglutinin inhibition; I, inactivated; NDV, Newcastle disease virus; neg., negative; pos., positive; post-ch, post-challenge; pv, post-vaccination; R, recombinant; RT–PCR, reverse transcription–polymerase chain reaction; wk, weeks.
* Age of vaccinated birds in weeks; parenteral vaccination (intra-muscular or subcutaneous) performed in laboratory animals unless mentioned otherwise.
† As measured between 2–4 weeks post-vaccination (unless mentioned otherwise) by OIE standard HI method [10].
‡ Protection from clinical signs and mortality.
Note: This list is a non-exhaustive list and more details on producers from China, Indonesia, Iran, Israel and Pakistan may be found on public access websites (FAO: http://www.fao.org/ag/againfo/programmes/en/empres/vaccine_producers.htm and http://poultrymed.com/files/index.html; accessed May 2008).
Their protective efficacy relies on the production of neutralizing antibodies against the haemagglutinin (HA) protein of a specific subtype of avian influenza virus (AIV) [Reference Katz13]. The minimum onset of protective immunity conferred by an inactivated vaccine begins 2 weeks post-vaccination [Reference Sims3, Reference Capua14–Reference van der Goot16] and could last up to 1 year post-immunization [Reference Marangon and Capua17, Reference Tian18]. In order to confer long-term protection (from 4 months to 1 year), between two and four injections, at a minimum of 3 weeks apart, are recommended depending on vaccine type and targeted species (manufacturers' recommendations [Reference Marangon and Capua17, Reference Capua19–Reference Webster21]; Table 3). This represents a great limitation for countries with a huge proportion of backyard poultry difficult to vaccinate at multiple times (e.g. P.R. China, Indonesia, and Vietnam). Only recombinant vaccines [reverse genetics (RG) H5N1 and H5N3, H5 fowlpox and H5 NDV] are able to confer protection after single dose (Table 3): the live fowlpox recombinant vaccine can even confer early and long-lasting protection after a single dose, from 20–40 weeks [Reference Qiao22, Reference Swayne, Beck and Mickle23]. However, its practical use is limited to fowlpox-free chickens (i.e. young birds) as the immune response is impaired if animals have previously been infected with fowlpox virus [Reference Swayne1, Reference Swayne, Beck and Kinney24]. All current commercial AI vaccines are for parenteral administration only; research on new delivery routes (oral or in ovo) is ongoing [Reference Crawford25, Reference Song, Nieto and Perez26]; with the exception of the H5 AI-NDV vaccine from P.R. China (oculonasal administration) [Reference Ge27], although limited information on its field application (P.R. China only) is available.
Inactivated whole virus vaccines
Conventional AI vaccines are produced with whole AIV of a specific subtype grown in embryonating chicken eggs (infective allantoic fluid). The grown viruses are chemically inactivated by β-propiolactone or formaline, adjuvanted with mineral oil (e.g. paraffin) [10].
Inactivated AI vaccines are either homologous or heterologous, depending on the choice of the viral strain. The homologous type is prepared from epidemic isolates or standard strains possessing the same HA and neuraminidase (NA) subtypes of the circulating field virus. The heterologous type has the same HA subtype as the circulating wild virus but a different NA subtype. The use of heterologous vaccines could allow the differentiation of infected animals from vaccinated animals (DIVA strategy). However, this is not relevant for H5N1-infected developing countries where the first priority is to control the infection [Reference Sims3].
Recombinant vaccines
Recombinant vaccines are based on the expression of an AIV gene of interest after insertion into a carrier vector (no pathogenic virus). Only three recombinant vaccines are currently licensed and in use (although numerous vaccines based on vectors such as baculovirus and retrovirus are under experimental development). (i) The fowlpox recombinant AI vaccine, this is based on the insertion of a gene coding for a specific HA subtype (H5 and H7) into the fowlpox virus (Table 3). This vaccine could confer long-term protection of up to 24 weeks, after a single administration, however, no protection is obtained if birds were previously infected with fowlpox virus [Reference Swayne28]. The efficacy of the fowlpox recombinant vaccine against the H5 subtype was well reviewed by Swayne [Reference Swayne1] and is being used on 1-day-old chicks in P.R. China, Mexico and Vietnam [Reference Qiao22]. (ii) The recombinant H5N1 vaccine is based on RG technology [Reference Subbarao and Katz29] (Table 3). Circulating strains of HPAI H5N1 viruses are too pathogenic to be grown in the laboratory for vaccine production. A LPAI H5N1 reassortant virus was generated using HA and NA genes from the circulating HPAI H5N1 strain and six internal genes from a low pathogenic virus (e.g. PR8 virus) [Reference Tian18]. This vaccine was used in Hong Kong and Korea in 2003 and is currently being used in P.R. China, Vietnam and Indonesia. (iii) The Newcastle disease (ND) recombinant AI vaccine has been licensed and in use in P.R. China since 2006 (Table 3). This vaccine combines immunization against AI and ND viruses [Reference Swayne30–Reference Veits32].
Recombinant vaccines are of great interest for oral or mucosal administration because of the flexibility in the choice of a vector suitable for these administration routes [Reference Crawford25]. However, in the case of the fowlpox vaccine, only limited protection is conferred using intranasal, eye drop, or drinking-water administration methods [Reference Ariyoshi33].
AI vaccine efficacy
To ensure a successful vaccination campaign, the vaccine must protect the vaccinated animals against clinical signs of the disease and prevent mortality (defined as ‘protection’ in the following section); reduce virus shedding into the environment and increase the minimum dose of virus required to infect a bird, therefore limiting contact infection and spread of the disease.
Immunological principles for AI vaccine protection have been well reviewed recently [Reference Swayne34], only practical results from laboratory and field validation will be discussed here. A clear distinction should be made between field validation of the vaccine (pilot trial under controlled field conditions as opposed to laboratory validation) and evaluation of the vaccination campaign (post-vaccination monitoring). The field validation under controlled field conditions by means of experimental epidemiology will give information on vaccine efficacy according to potential limiting factors associated with the vaccine itself (virus serotype and level of protection induced) and the bird/flock (maternal immunity, immunosuppresssion, sanitary status. genetic factors) [Reference Marangon and Busani35]. Whereas multiple factors not directly linked to the vaccine or the animals could be interfering with the vaccine efficacy when evaluating a vaccination campaign (vaccine administration; associated biosecurity measures; awareness campaign, etc.) [Reference Marangon and Busani35]. Evaluation of a vaccination campaign (post-vaccination monitoring practices, coverage rate, level of seroconversion, virus circulation) will be discussed later in the review.
Protection against AI is mostly conferred by the production of antibodies against HA viral protein [Reference Katz13, Reference Swayne34]. Therefore, the level of seroconversion in terms of anti-HA antibodies [measured by haemagglutinin inhibition (HI) test] is used to evaluate vaccine efficacy [2]. According to the OIE International Manual, HI titres are considered positive when the inhibition of the hemagglutination occurs for a serum dilution of at least 16 (4 log2) against 4 haemagglutinin antigen units (HAU) antigen [10]. To assess vaccine efficacy, some experimental studies [Reference Philippa36] and field vaccination campaigns (Italy 2000–2002 [Reference Capua and Marangon37], Hong Kong 2002 [Reference Ellis38], Vietnam – ongoing since 2005 [Reference To39]) are following the criteria defined by the Committee for Proprietary Medicinal products (CPMP) for validation of human influenza vaccines, i.e. HI titres ⩾40 (5–6 log2) in ⩾70% of the vaccinated population [40, 41]. However, the validity of those criteria in avian species remains empirical. Regarding infection and contact transmission, a minimum goal for reduction in viral shedding has been defined as 102-fold in order to reduce or prevent contact infection [Reference Suarez, Lee and Swayne42].
All the commercial vaccines have been validated in the laboratory to meet those standards (with HI titres in chickens: primary dose >5 log2; booster dose >8 log2 [Reference van der Goot16, Reference Tian18, Reference Webster21, Reference Bublot43–Reference Swayne, Lee and Spackman45]; see Table 3) and some studies have shown a reduction in viral shedding >103-fold in vaccinated chickens compared to unvaccinated birds following challenge with homologous and heterologous viral strains [Reference Capua14, Reference Ellis38, Reference Bublot43, Reference Swayne, Lee and Spackman45]; a reduction of infectiousness and reduction of the birds' susceptibility 2 weeks' post-vaccination [Reference Capua14–Reference van der Goot16].
Although differences in HI titres could be observed between vaccines (e.g. HI response in chickens against H1N1 (11 log2); H5N1 (9 log2); H2N3 (8 log2) [Reference Liu44]; fowlpox vaccine does not induce very high level of antibodies compared to H5N9-inactivated vaccine [Reference Steensels46]); and species (antibody response in ducks or turkeys could be lower than chickens [Reference Webster21, Reference Tumpey, Kapczynski and Swayne47]), they all confer very high level of protection against clinical signs and mortality (90–100% protected birds). A limited number of studies are available on the correlation between serological titres and protection against viral challenge. A direct relationship between HI titres and protection has been demonstrated for inactivated vaccines: Kumar et al. have shown that if HI titres were <3·5 log2, chickens were not protected against clinical signs and might die; with titres between 3·5 log2 and 4·5 log2, no mortality was observed but the animals will shed the virus and only with titres >4·5 log2 was no further viral shedding observed [Reference Kumar48]; van der Goot et al. have shown that viral shedding was reduced and transmission was prevented with HI titres >4 log2 [Reference van der Goot16]; and Lee et al. have shown that no shedding was observed for HI titres >5 log2 [Reference Lee49]. Therefore experimental data confirm the threshold value of HI titres >4 log2, as defined by the OIE International Manual, to assess vaccine efficacy in terms of protection of the vaccinated animals [2]. The link between serological titres and protection against viral challenge is not so clear for recombinant vaccines: in a study from Webster and colleagues, Pekin ducks vaccinated with low antigen doses of RGH5N3 (from 0·015 μg to 0·0313 μg) did not produce detectable HI antibody titres but were fully protected against lethal challenge with H5N1 HPAI virus [Reference Webster21]; similar data have been shown for the H5 fowlpox vectored recombinant vaccine which induces a very low level of HI titres (<3 log2) but conferred full protection to the vaccinated chickens [Reference Steensels46]. However, inconsistent or low HI antibody responses following vaccination of chickens with the recombinant fowlpox H5 vaccine could be linked with the antigen used in the HI test (homologous or heterologous type) whereas this is not the case for inactivated vaccines [Reference Swayne28].
Inactivated AI vaccines might induce very high level of HI titres before challenge which seems to prevent morbidity and to some extent cloacal shedding, however, some studies have shown limited reduction in oral shedding for vaccinated birds and therefore risk of contact infection for unvaccinated birds [Reference Bublot43, Reference Swayne, Lee and Spackman45, Reference Terregino50].
Moreover, a limited cross-protection could occur between heterologous and homologous vaccines. According to the literature and manufacturers' validation data, all the commercial AI vaccines tested are able to confer 100% protection against experimental challenge with homologous viruses and a broad cross-protection exists with heterologous strains [Reference Swayne, Lee and Spackman45, Reference Steensels46, Reference Terregino50–Reference Swayne53]. However, discrepancies in viral shedding (cloacal and tracheal) have been linked to genetic variation between the vaccine and challenge virus strains [Reference Lee, Senne and Suarez54]. Some studies have demonstrated a direct correlation between the HA sequence similarity of the vaccine and challenge viruses and the ability of the vaccine to reduce tracheal shedding [Reference Swayne51, Reference Lee, Senne and Suarez54]. Lee et al. have also demonstrated that fourfold more HI titres were required to protect vaccinated animals against challenge with heterologous virus [Reference Lee49]. Moreover, Liu et al. demonstrated that despite the high level of HI titres induced by commercial H5N2 vaccine, protection was not fully achieved following challenge with high doses of H5N1 virus [Reference Liu44] which highlights possible limits of heterologous vaccines in terms of disease control and eradication.
Discrepancy between laboratory and field results have also been observed [Reference Cristalli55]. Such differences could be linked to the immune status of the vaccinated birds (immunosuppressive conditions and concurrent diseases) and the practicalities of setting up a vaccination campaign (technical issues related to vaccine storage and administration). However, limited field trial data have been available so far and international and/or national authorities should ensure that new vaccine formulations are being validated in field pilot studies prior routine use in mass vaccination campaigns.
As the minimum requirements in terms of vaccine efficacy are not yet clearly defined for laboratory experiments it is still very difficult to predict the minimum required for a vaccine to be efficient in controlling the disease in the field. Field validation trials based on standard epidemiological protocols (case-control or cohort studies) [Reference Toma56] are needed to assess vaccine efficacy according to specific epidemiological and local context (type of poultry production, species, type of circulating field virus, other circulating diseases and immune status of the animals, etc.).
IMPLEMENTATION OF AI VACCINATION
A basic rule to follow when implementing vaccination against AI is that the use of vaccines is only one of several tools to prevent or contain an outbreak spreading in unaffected flocks. Other effective control and biosecurity measures must be implemented in addition to vaccination. Vaccination against HPAI has proven to be a successful additional control measure implemented alongside controlled culling in Italy in 2000 (H7N1), in Mexico in 1995 (H5N2), and in Pakistan in 2003 (H7N3) [Reference Marangon and Capua17, Reference Naeem and Siddique57, Reference Villarreal58]. Hong Kong was also successful in eradicating H5N1 AIV after the 2002 outbreak when implementing both vaccination and strict biosecurity measures such as the ban of live animal markets and the separation of ducks from chickens on production farms [Reference Ellis15].
Stamping-out vs. vaccination
Until recently, mass culling of animals was the most efficient measure of preventing the spread of AI epidemics. After a H5N1 AIV outbreak in Hong Kong in 1997, mass culling of all poultry animals was performed within a few days and this measure prevented the spreading of the virus at that time [Reference Shortridge, Peiris and Guan59]. However, since 2003, the H5N1 epidemic has spread too far worldwide to be contained only with a stamping-out policy which would imply mass culling of millions of more birds. In most of the infected countries in Asia poultry rearing represents the main source of income and many households own backyard poultry. Most of the infection of backyard poultry (or even small farms) goes unnoticed and sick poultry are quickly sold to the local market [Reference Phan Dang, Camus, Cardinale, Dalibard, Martinez, Renard and Roger60], helping the virus to spread rapidly. The delay in putting in place measures such as awareness programmes and compensation schemes by the competent authorities along with limited level of biosecurity and local commercial practices (e.g. live bird markets) has greatly facilitated the spreading of the disease. The rationale of vaccination and mass culling should be carefully evaluated. Key elements to be taken into account by decision makers include the efficiency of veterinary surveillance systems in place, the economic impact, and the export policy of the country. An evaluation tool called ‘Performance, Vision and Strategy’ has been developed by the OIE to help in the identification of such key elements and in the design of investment programmes to reinforce them if necessary. Countries which are able to rapidly detect, contain and eradicate the disease based on efficient surveillance system and control measures should continue with the stamping-out of infected flocks, following OIE recommendations [2]. Stamping-out could also be associated with preventive vaccination depending on the risk of introduction of the disease. In February 2006, France applied a strict stamping-out policy to contain an outbreak of H5N1 AIV along with a nationwide preventive vaccination of birds that could not be contained indoors because there was a high risk of introduction of H5N1 AIV by migratory birds. Countries where the disease is becoming or likely to become endemic, should conduct a universal vaccination programme [2]. In 2005 Vietnam and P.R. China began universal vaccination programmes against H5N1 AIV following the introduction of the virus in 2003. However, because of the presence of billions of backyard poultry, vaccination remains difficult to implement for practical and economic reasons.
Advantages of vaccination
AI vaccines increase host resistance to AI disease by inducing a strong immune response through the production of neutralizing antibodies against AIV [Reference Katz13]. Moreover, AI vaccines reduce virus shedding and therefore limit contact transmission. In their transmission experiments, van der Goot et al. have shown that more virus load is needed to infect a vaccinated animal [Reference van der Goot16]. This experiment also demonstrated that vaccinated animals excreted less or no detectable viral particles (depending on the vaccine efficacy), therefore reducing the risk of contact infection within flocks [Reference van der Goot16, Reference Lee and Suarez61]. Mexico was able to contain an HPAI outbreak and eradicate the HPAI virus by using vaccination along with strict biosecurity measures in 1995 [Reference Villarreal58]; vaccination against an H5N1 AI outbreak interrupted virus transmission in Hong Kong in 2002 [Reference Ellis15]. However, most of the vaccines currently in use have only been validated in the laboratory to prove their efficacy both in terms of resistance to infection and reduction of viral shedding (Table 3). Their field efficacy could often be impaired because of the immunodeficiency of the hosts due to many circulating diseases. Efficient vaccines for waterfowl also remain an issue especially in countries such as Vietnam [Reference Cristalli and Capua62].
Issues related to vaccination
The quality of AI vaccines is affected by many factors including antigenicity of the vaccine strain, route of administration, procedures in vaccine production, vaccine formulation and antigen mass present in the vaccine and vaccine preservation [Reference Webster63, Reference Swayne64]. Although AI vaccines could protect a bird or a flock from clinical diseases, as described above AI vaccines could only partially reduce virus shedding and the bird or the flock might still spread the virus (see section on AI vaccine efficacy) [Reference Capua19, Reference Swayne64].
Furthermore, AI vaccines may promote the selection of mutation in the circulating virus and thereby perpetuate the risk of infection in the original species or in another. For example, the H5N2 vaccines that have been used in Mexico since 1995 might be, among other factors, at the origin of an antigenic drift of the field virus away from the vaccine strain [Reference Lee, Senne and Suarez54]. If this were to occur, the vaccine protective efficacy would be impaired in time and the use of this specific vaccine strain would eventually become obsolete. For these reasons an efficient monitoring of circulating virus within vaccinated flocks is essential. This represents a major constraint for developing countries due to economic and practical limitations.
Validation of vaccines in the field is often species specific, i.e. most of the AI vaccines have been validated in the field in chickens and turkeys but little is known about their field efficacy in other species such as ducks and geese. Ducks have been identified as a potential asymptotic carrier of the virus [Reference Hulse-Post65]. Most of the inactivated AI vaccines have a limited efficacy in ducks [Reference Swayne34]. However, RG vaccines are already in use in the field for vaccination of ducks but require twice the antigenic load used for chickens and/or the addition of a strong stimulator for the immune response to be effective (oil-adjuvanted recombinant vaccines) [Reference Tian18]. Vaccine quality control and pilot field trials are not widely performed within developing countries as this requires specific equipment and financial resources.
A major limitation of vaccination is the need to be able to differentiate between infected and vaccinated animals (DIVA). When homologous vaccines are used, both infected and vaccinated animals carry antibodies to the same virus subtype. However, simple practical techniques exist to distinguish infected from non-infected vaccinated birds such as the use of sentinel birds (unvaccinated animals). In the case of recombinant vaccines, they do not induce antibodies against matrix or nuclear proteins of the AIV, therefore simple ELISA or AGID tests can be performed to apply the DIVA strategy [10]. Although these techniques seems relatively simple and easy to implement in industrialized countries, their use in developing countries is greatly impaired because of practical and socio-economic issues [Reference Sims3, Reference Cristalli and Capua62].
When heterologous vaccines are used, anti-NA antibodies specific to the field virus and different from the vaccine strain may be detected. However, their use could be impaired in areas where LPAI viruses with NA similar to the vaccine strain might be circulating. To date, the DIVA method using the heterologous NA test has only been applied in Italy although heterologous vaccines have been used worldwide [Reference Capua and Marangon37].
Under the OIE initiative, a disease-free zone could be created by selective use of vaccine and the area could regain some export capacity. However, developing countries practicing AI vaccination such as Vietnam do not yet consider it to be a feasible option because thorough surveillance, traceability and sampling protocols would be required. DIVA testing is considered to be too laborious and costly to implement and recovery of export capacities is of secondary importance to the control of the disease and public health issues.
Vaccination strategies
Different vaccination strategies could be applied according to the infectivity level of a country [Reference Capua and Marangon66, Reference Bruschke, Bruckner and Vallat67].
(1) Preventive vaccination should be used in a country free of disease but at high risk of introduction of the disease. All birds at high risk should be vaccinated. France, The Netherlands and Switzerland recently vaccinated zoo birds and other birds which could not be easily contained due to the threat of H5N1 infection in migratory bird areas. In Europe preventive and emergency vaccination against HPAI are authorized under some circumstances (EU Council Directive 2005/94/EC). This strategy implies a major cost-benefit issue for developing countries where vaccination against circulating diseases such as Newcastle disease is not properly in place.
(2) Emergency vaccination should be conducted during an outbreak. All unaffected animals within and around an outbreak quarantine zone should be vaccinated. The size of the vaccination zone depends on the transmission rate and initial spread during the high-risk period and should be defined within the contingency plan by professionals and/or national and international authorities. As protective immunity takes a minimum of 2 weeks to develop, the efficacy of this strategy depends on various factors including vaccine availability and the feasibility of rapid administration. This represents a major limitation for developing countries. There is a strong need to develop vaccine banks by international bodies such as OIE and FAO, to provide efficient vaccines to developing countries and to ensure their delivery to remote areas or poor countries. Preventive vaccination should be used in coordination with emergency vaccination within a country if the risk of virus dissemination is high.
(3) Prophylactic vaccination should be applied when the disease has become endemic. Birds are vaccinated systematically against the same HA subtype of the virus circulating in poultry to obtain a minimum protective level within the ‘at-risk’ population. The final goal of the approach is to control (and to eradicate when possible) the disease within the country. This could be a long-term vaccination plan which should be applied nationwide on all commercial and backyard poultry. Strict control measures including stamping-out should be applied to affected flocks to better coordinate this approach. Once the disease is controlled, biosecurity measures and stamping-out can achieve eradication. This strategy also implies major practical and cost-benefit issues for implementation by developing countries.
Export bans linked to any of the vaccination strategies are still a limitation for some countries. To be efficient a vaccination programme should involve all the stakeholders including farmers, veterinarians and decision makers. This should be an integrated approach with awareness campaigns and proper training, especially in countries where biosecurity within the poultry production sector is limited.
Post-vaccination surveillance
Vaccine efficacy and virus circulation within vaccinated flocks should be monitored as recommended by international authorities by using virological and immunological methods described below [10].
First, the protection conferred by the vaccination campaign must be assessed. Protective immune response, i.e. antibody levels against a field virus, is measured in sera from vaccinated animals by the HI test [10]. Serum samples should be collected 2–3 weeks post-vaccination (Table 4a, Fig. 1).

Fig. 1. Flowchart of diagnosis tests for post-vaccination surveillance. (A), Virus circulation and (B), vaccine efficacy could be monitored by rapid tests and confirmed by conventional tests: agar gel immunodiffusion assay (AGID); haemagglutinin inhibition test (HI); reverse transcriptase–polymerase chain reaction test (RT–PCR).
Table 4a. Serological screening tests to measure antibody levels in poultry serum
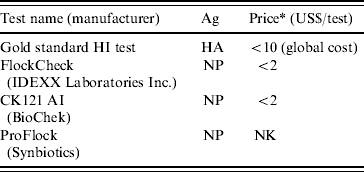
Ag, Antigen detected; HA, haemagglutinin; NK, not known; NP, nucleoprotein.
* 2007 prices, manufacturers' information.
A serological conversion of 70–80% of the vaccinated animals has been empirically considered by Charles Nicolle as sufficient to confer protection to the whole population [Reference Toma56]. Indeed, Nicolle's law analogous to the herd immunity threshold (HIT) is greatly in use but would require experimental validation under the AI context. HIT is based on the evaluation of the rate of spread of the disease measured by the basic reproductive number R 0, i.e. the average number of secondary infections produced by one infected individual introduced into a fully susceptible population [Reference Heffernan, Smith and Wahl68, Reference Gerbier69]. Models designed to calculate R 0 and therefore HIT usually meet Nicolle's value [Reference Savill70]. However, more studies are needed to assess the minimum coverage and seroconversion levels required within a zone and/or AI-vaccinated birds to confer protection against the disease; and so at multiple epidemiological units (e.g. animals, farms, villages). During the 2002 H5N2 vaccination campaign, the Hong Kong Ministry of Agriculture recommended testing 10–20 serum samples per flock with the HI test at 1 month after the second vaccine dose. A good seroconversion was achieved (>80%) and no subsequent AI outbreak or virus isolation were reported which proves that the vaccination programme was efficient and associated with other control measures enabled control of the disease [Reference Ellis38].
The main drawback to AI vaccination is that a virus might circulate undetected which increases the risk of subsequent outbreaks and/or antigenic drift of the circulating virus, away from the vaccine strain. It is recommended that unvaccinated birds or sentinels (chickens or ducks) should be kept within each flock (minimum 10–20 birds per flock) and distributed randomly inside the flock. Sentinels immediately will show morbidity/mortality when HPAI is circulating; they also will show antibodies if LPAI is in circulation or if the birds do not show any clinical signs of HPAI infection (e.g. some ducks) [diagnosis made by rapid testing, Tables 4a, 4b; Fig. 1, A(1) and B(1)] followed by confirmatory tests [Fig. 1, A(2) and B(2)]. In 2002, the Hong Kong Agricultural Fisheries and Conservation Department recommended the use of 30–60 sentinel birds per flock, and the collection and testing of sentinel serum samples every 30–45 days for as long as the vaccination campaign continued [Reference Ellis38]. A recent mathematical model has shown that a flock with 80% vaccinated birds increases the chance by almost 20% of an undetected outbreak at the end of the production cycle and cumulative infectiousness. According to the model this situation does not apply when sentinel birds are used [Reference Savill70], but this simulation model has not been validated with any historical data. Unfortunately, the use of sentinels is often limited in developing countries for economic and practical reasons associated with difficulties in identification of unvaccinated birds within a flock. Furthermore, sentinel birds can not be used with backyard poultry. All backyard chickens should be vaccinated to build a protection zone in high-risk areas. Any unvaccinated backyard birds could be a potential virus reservoir which already poses a problem in Asian countries practising vaccination and which may pose a problem in Africa. In numerous developing countries, the poultry production system is mainly backyard poultry production. Under these circumstances, virus circulation may remain unnoticed. However, the role of backyard poultry in maintaining and spreading the infection has been questioned as this production sector appears to be less at risk than the semi-commercial sector (100–10 000 heads) with poor biosecurity level [Reference Sims3].
To increase the surveillance level, swabs and/or serum samples from random animals in the flock could also be tested regularly by polymerase chain reaction (PCR) test which detects AI nucleoprotein genes or HA genes (Fig. 1). The PCR test applied to field samples has a high sensitivity (95·6%) and specificity (96·3%) compared to conventional virus isolation (VI) procedures (Table 4b) [Reference Cattoli and Capua71], but it requires special equipment, laboratory supplies, and well-trained test conductors. Rapid diagnosis tests have less sensitivity and specificity than PCR tests (Table 4b) [Reference Cattoli72] but they are convenient and easy to use when an outbreak has been declared and the virus previously identified using conventional methods such as VI followed by PCR on purified concentrated viral samples [10] (Fig. 1). At first Vietnam used such tests to diagnose AI within sick flocks, however, the high cost and sensitivity has been a real issue, therefore the Vietnamese authorities no longer recommend the use of such rapid tests.
Table 4b. Characteristics of diagnostic tests to detect presence of Influenza A virus (antigen detection) in field samples (cloacal or thracheal swabs)

Ag, Antigen detected; EID, embryo infective dose (lethality 50%); wk, weeks; HA, haemagglutinin; NK, not known; NP, nucleoprotein.
* 2007 prices, manufacturers' information.
† Independent validation study [Reference Cattoli72].
‡ Manufacturer's data.
Vaccine supply
The main AI vaccine manufacturers are located in the United States, Mexico, The Netherlands, France, and P.R. China (Table 3). To date, pharmaceutical companies in the United States produce AI vaccines based on demand and do not hold any stocks [Reference Suarez, Lee and Swayne42]. The orders are processed on a case-to-case basis and a few months' delay could occur before the delivery of the final product. This is also the case for most of the manufacturers worldwide as until now vaccines against AI were not recommended for general use. Since the beginning of the Asian epidemic in 2003, recommendations have been made by international organizations (OIE, WHO) to vaccine manufacturers and affected countries to create a vaccine bank and to allow individual states to stockpile AI vaccines for emergency use [Reference van Aarle73].
In January 2006, France ordered 30 million doses of AI vaccine from two manufacturers (Intervet, The Netherlands and Fort Dodge, USA) for preventive vaccination purposes; Russia is planning to produce 1.2 billion doses of AI vaccine annually and 6 millions doses were delivered in 2007 to provinces in southern Russia; the Nigerian government plans to acquire 1 million doses of AI vaccine because of the recent outbreaks. With the rapid spreading of the H5N1 virus since the beginning of the year 2006, the list of countries stockpiling AI vaccines constantly is increasing.
P.R. China has produced billions of doses of vaccines to be used in mainland China and for export to the South East Asian countries of Vietnam and Indonesia. Over 10 regional vaccine companies or regional veterinary research institutes coordinated and supervised by China National Harbin Veterinary Research Institute are producing AI vaccines. Local production of vaccines should be considered for countries where the disease is endemic to ensure a constant vaccine supply and financial independence from the international markets.
Economic aspects of the implementation of AI vaccination
The choice of implementing vaccination should also be considered from an economic perspective. Cost-effectiveness and cost-benefit analyses should be conducted to determine direct costs (e.g. price of doses, administration) and indirect costs (post-vaccination surveillance, commercial losses, export bans, etc.) before the implementation of vaccination [Reference McLeod74, Reference McLeod and Rushton75].
Parameters to be taken into account include:
• Development status of poultry production systems (commercial vs. backyard production; e.g. 99% backyard poultry in Ethiopia and Cambodia; 60% backyard and 40% commercial in Nigeria and Vietnam and 90% commercial in Thailand).
• Direct vaccination costs (‘needle in bird’ cost including price of vaccine per dose; administration and storage; post-vaccination monitoring; awareness campaign) [Reference McLeod74, Reference Hinrichs, Sims and McLeod76].
• Indirect vaccination costs (e.g. potential drop in poultry market prices) including economic losses due to reduction of exporting capacity. The rates of indirect costs could greatly vary from one country to another according to the development status of the poultry sector [Reference McLeod and Rushton75].
Consideration of export bans are only relevant for industrialized countries relying on poultry exports as an important income. The indirect costs linked to export bans for countries using AI vaccination could be controlled by applying vaccination based on a DIVA strategy. As previously mentioned, a country could recover its full exportation capacity if there is sufficient evidence of no contamination in vaccinated flocks, depending on the requirements of international authorities. In 2001, Italy was able to recover its poultry trade capacity while using a H7N3 heterologous vaccine against a H7N1 LPAI outbreak by applying the iFat technique to detect infected from non-infected birds within the vaccinated population [Reference Capua19]. However, this is not a priority for countries such as Vietnam or Indonesia where between 60–90% of the poultry population is reared backyard.
Most of the economic studies on AI have so far concentrated on the impact of the disease, more studies are needed to assess the impact of its control measures both in term of sustainability and efficacy [Reference Rushton77, 78]. For developing countries such as Vietnam and Indonesia, considerations are focused on small-scale poultry producers who rely on the poultry market as their main livelihood [Reference Phan Dang, Camus, Cardinale, Dalibard, Martinez, Renard and Roger60]. Epidemiological models including economic aspects are needed to estimate the cost-benefit of a vaccination campaign and to compare the cost vs. benefit of different available strategies prior to implementation. Such models would help decision makers in their choice of the best strategy to implement according to the specific socio-economic context of the country/zone [Reference Alders79].
FIELD EXPERIENCE IN RELATION TO THE USE OF AI VACCINES
AI vaccines have been used in Mexico, Italy, Pakistan, and the United States to control LPAI. Previous to the HPAI H5N1 outbreak in South East Asia (2003), only a few attempts to control HPAI outbreaks by the means of vaccination have been reported: the HPAI H5N2 outbreak in Mexico (1994) and the HPAI H7N3 outbreak in Pakistan (2003) [Reference Naeem and Siddique57, Reference Villarreal58]. Table 5 presents a synthetic review of the AI vaccination strategies used in the field according to the country and the type of AI virus (HP or LP) and a brief summary of the outcome of the vaccination campaigns.
Table 5. Avian influenza vaccination campaign established in countries
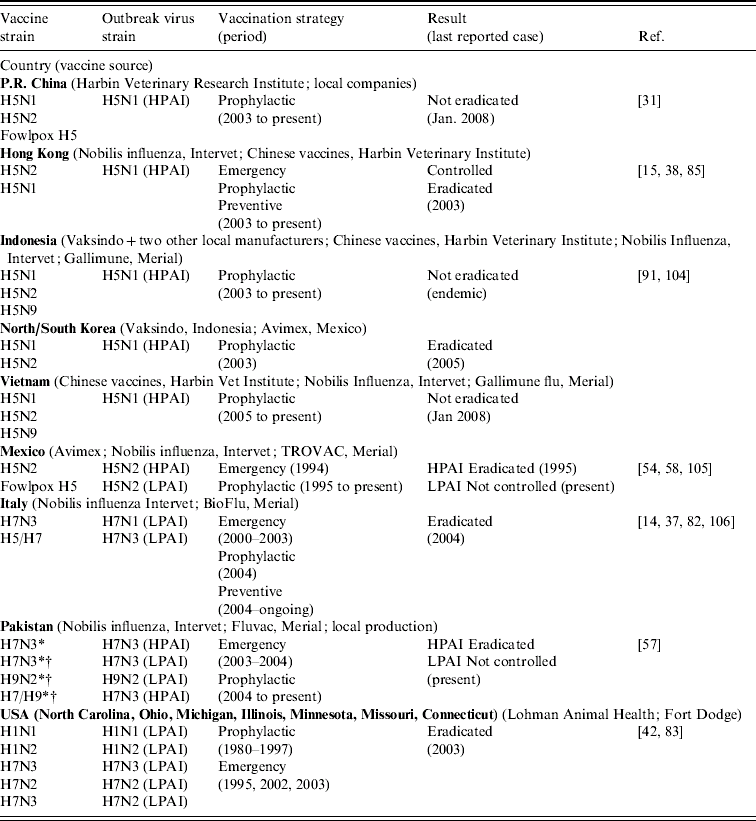
* Aqueous-based vaccine.
† Oil-based vaccine.
Homologous vs. heterologous vaccines
Homologous AI vaccines against H1 and H7 LPAI outbreaks have been used in commercial turkey flocks within localized areas in the United States since the 1980s [Reference Suarez, Lee and Swayne42]. These strategic vaccination campaigns have been successful at either controlling or eradicating the disease by prophylactic vaccination and/or emergency vaccination respectively. The choice of using vaccination or mass culling could be made easily in high-density poultry areas in which export bans were not an issue for consideration. The safe and efficient use of homologous vaccination for turkeys was monitored by veterinary services and surveillance systems that were established previously in the country [Reference Suarez, Lee and Swayne42]. A DIVA vaccination strategy was planned against the 2003 H7N2 outbreak in the United States but the disease was eradicated before the heterologous H7N3 vaccine was used. A similar scenario was successful in Italy to eradicate LPAI of H7N1 subtype by using heterologous H7N3 vaccine in 2000. In order to apply the DIVA strategy [Reference Capua, Cattoli and Marangon80], the Italian reference laboratory developed an ad hoc diagnostic test capable of detecting N1 subtype NA antibodies in serum samples based on fluorescent immunoreactions. Due to the use of heterologous vaccines and the effective implementation of a DIVA strategy, the country was able to recover its exportation capacity by practising vaccination [Reference Marangon and Capua17]. However, as previously mentioned, the use of an heterologous vaccine as part of a DIVA strategy to limit economic losses is neither a priority nor practical to implement in many developing countries as DIVA testing can be laborious, expensive and sampling procedures difficult to implement.
Moreover, as previously mentioned, the efficacy of heterologous vaccines could be limited according to the genetic similarities with the challenge strain (see section on AI vaccine efficacy). Although no published field data are yet available to confirm experimental findings one could also expect limited efficacy against challenge in the field. The current priority for developing countries infected with H5N1 AI virus is to control the disease in an efficient manner. Therefore the choice for an heterologous vaccination as part of a DIVA strategy is not a priority for most of the currently H5N1-infected countries [81].
On the other hand, when control measures are not applied properly to coordinate the vaccination campaigns, the use of homologous vaccines could have a negative impact on the genetic evolution of the field virus. Indeed, Mexico has been using extensive vaccination to control LPAI H5N2 epidemics since 1995 without any success to date [Reference Villarreal58]. Even worse, genetic studies on the evolution of the circulating virus have demonstrated an antigenic drift away from the vaccine strain [Reference Lee, Senne and Suarez54]. As the virus is undergoing mutation due to vaccine pressure the threat of LPAI virus mutation into a HPAI strain remains important [Reference Villarreal58, Reference Capua82, Reference Halvorson83]. Therefore careful attention should be paid to the genetic evolution of the field virus strain away from the vaccine strain. The vaccine strain should be updated when necessary to maintain its efficacy against challenge in the field [Reference Dung84].
Vaccination against H5N1, the Asian experience
SAR Hong Kong has been using vaccination against H5N1 since the 2002 epidemic [Reference Sims3, Reference Ellis15, Reference Ellis38, Reference Ellis85–Reference Sims87]. All chickens were vaccinated with inactivated H5N2 vaccine in 2002–2006. Unvaccinated sentinel poultry were used in commercial flocks and ducks were separated from chickens in live-bird markets. At first, the virus continued to spread within vaccinated flocks with a low mortality rate 9–18 days post-vaccination, however, after 18 days (the minimum onset time for vaccine to confer protection) there were no more deaths within vaccinated flocks. Moreover, there was no evidence of asymptomatic shedding of the virus [Reference Ellis38]. Vaccination programmes and biosecurity measures have proved to be effective as no outbreak was reported from 2004 until the present time whereas a number of outbreaks occurred in adjacent regions of mainland China, Thailand, and Vietnam [Reference Sims3].
Emergency vaccination for commercial and backyard poultry was conducted to contain the widely spread H5N1 outbreaks in P.R. China in 2004. State agricultural agencies established a plan to conduct universal vaccination in 2005 [Reference Sims3]. However, the disease was not contained and new outbreaks have occurred in several regions of China since early 2006 [88]. A study conducted in 2004–2005 showed that unvaccinated asymptomatic chickens and ducks in some Chinese markets were infected with H5N1 virus [Reference Hulse-Post65]. The existence of such ‘silent carriers or excretors’ is dangerous because they become the virus reservoir and shed the virus into their environment, causing potential outbreaks in commercial poultry and threatening human health. However, only 0·6% of the chickens were positive by virus detection which could be due to a temporary infection in the market. The findings in live-bird markets may not represent the true situation in commercial flocks. The vaccination campaign involving more than 5 billion poultry – most of all backyard poultry – with coverage of only 20–50% has been sufficient to a certain extent in containing the outbreaks or the circulation of the virus. However, the presence of a new non-pathogenic virus variant for ducks increases the risk of new outbreaks within the poultry population [Reference Hulse-Post65]. The appropriate application of the monitoring systems such as sentinel birds, serology and viral testing, is also under question. P.R. China has recognized the need to improve efficacy of the AI vaccines produced domestically in research institutes and vaccine manufacturers. Nonetheless, the Chinese government plans to provide free AI vaccines to poultry producers and farmers to increase the vaccination rate.
The situation between P.R. China and Hong Kong is very different for many reasons: the scale of territory to be controlled; the heterogeneity between Chinese regions in terms of poverty and level of industrialization, and in particular the high number of households still raising backyard poultry in mainland P.R. China, compared with Hong Kong. In Hong Kong the virus was eradicated in 1997 after the first outbreak when the authorities decided to cull all the poultry within the territory, then strict biosecurity measures where applied to prevent a new emergence of the virus [Reference Sims86]. In P.R. China the situation is very different as the virus might be circulating in the environment (wild birds) and being maintained unnoticed within unvaccinated backyard poultry [Reference Lei89]. China has only reported a small number of human cases since the start of the epidemics in 2003 (30 human cases since 2003 compared with 133 cases for Indonesia since 2005) which could reflect a controlled level of infected animals (or a low reporting rate), probably because of the large-scale vaccination campaign. However, given the low transmission rate of this disease from birds to humans, human cases act as ‘sentinels’ for avian infection and therefore still indicate a significant virus activity in China's poultry population (so far three human cases in China for 2008) [Reference Sims3].
In Indonesia, vaccination has been implemented since 2005 using mostly Chinese and locally produced inactivated H5N1 and H5N2 vaccines but a high number of H5N1 outbreaks in unvaccinated flocks have been reported since early 2006 [90]. Initially, the Indonesian government wanted to apply universal vaccination, but due to budget limitations, only strategic vaccination was conducted in regions where the disease was endemic [Reference Sawitri Siregar91]. The uncontrolled situation currently faced by the Indonesian government might be explained by the specific geographical and economic context of Indonesia. The insular organization of the country has created an ‘autonomous’ era of the veterinary services and a common policy is difficult to implement [Reference Sawitri Siregar91]. Moreover a limited supply of vaccines due to budget constraints has also impaired the vaccination campaigns [Reference Sawitri Siregar91].
Vietnam used European and Chinese vaccines for a pilot vaccination campaign in the summer of 2005. Later, only Chinese RG H5N1 and TROVAC (1-day-old chicks, industrial sector) vaccines were used for universal vaccination. Vietnamese authorities approved the import of AI vaccines from China after they conducted all required quality control tests. The first round of vaccination was completed in all chickens (around 160 millions doses, twice a year). A post-vaccination surveillance programme has been implemented and serum samples were collected in some flocks and tested in state laboratories [Reference Taylor and Dung92]. Data showed that in some areas the vaccination coverage rate was closer to 60% with only 60% of seroconversion of vaccinated animals which could bring the protection level of the poultry population down to 40% in some provinces [Reference To39, Reference Taylor and Dung92]. Low levels of seroconversion could be a result of poorly administered or preserved vaccines. From the thousands of poultry swab samples collected within the country, only a few tested positive by RRT–PCR [Reference To39]. Nonetheless, no outbreaks within the vaccinated poultry population were reported following the first year of vaccination (from December 2005 to December 2006). And no human cases were reported in Vietnam until 2007. All the new cases of H5N1 AIV infection reported recently were all in unvaccinated ducks and geese [93, 94]. This new wave of outbreaks (March–July 2007) is simultaneous with the lift of the ban on duck hatching and the end of the rice-field season when ducks are allowed to free range on field paddies [Reference Desvaux95]. These new outbreaks emphasize the need for a comprehensive waterfowl vaccination strategy and the development of waterfowl-specific efficient vaccines. Muscovy ducks have been vaccinated since 2007 with European H5N9 vaccine but no post-vaccination monitoring data have yet become available. Moreover, the vaccination campaign might have helped the virus spread from one farm to another [Reference Pfeiffer96]. A comprehensive AI surveillance system to efficiently monitor virus circulation in vaccinated flocks still is needed as the use of sentinels and virological monitoring is not properly applied [Reference Desvaux, Camus, Cardinale, Dalibard, Martinez, Renard and Roger97].
DISCUSSION
AI vaccination alone can not be used to eradicate the disease. Indeed, it is difficult to predict the efficacy of a vaccine with respect to reduction in virus shedding and therefore its impact alone on the transmission dynamics of the disease [Reference Suarez, Lee and Swayne42]. In countries where poultry is mainly backyard scavenger poultry, optimum vaccination coverage might be difficult to achieve. Furthermore, if an antigenic drift occurs due to vaccine pressure [Reference Lee, Senne and Suarez54], the vaccine may still prevent the host from showing clinical disease but fail to stop or reduce virus shedding by the host. To effectively control the virus from circulating in poultry, an efficient post-vaccination surveillance programme should be established. This appears to be the main challenge for developing countries in the use of vaccination against AI.
An eradication programme should include strict quarantine, movement controls on animals and equipment, increased biosecurity, extensive surveillance, and a comprehensive education programme for the public.
Until recently, AI vaccines have not been used extensively and research was limited (around four publications per year on AI vaccines for the past 40 years against 120 per year since 2004). Although H5N1 vaccines have been extensively used in Asia since 2004, Chinese manufacturers have been covering most of the market needs and other international companies have not felt the need to focus research on the optimization of current vaccines. The ideal vaccine for tropical countries needs to be a single dose, stable at room temperature and efficient in multiple species; none of the current AI vaccines meet any of these standards. However, numerous experimental AI vaccines are under development such as a recombinant vaccine based on baculovirus vector [Reference Crawford98] or DNA vaccines [Reference Kodihalli, Kobasa and Webster99]. Eradication of the disease within a poultry population will not be achieved if effective vaccines are not available for waterfowl.
New concerns recently have arisen regarding the illegal use of vaccines obtained from ‘underground’ or ‘black’ markets. The illegal use of AI vaccines has been reported in some countries where vaccination against AI is still prohibited [90]; fake vaccines containing only cows' milk were sold to farmers and used on chickens in some countries [100]. A thorough control and communication programme on the benefits and limits of AI vaccination needs to be developed quickly and put in place.
AI vaccine markets are expanding. More developing countries are planning to produce their own vaccine to meet domestic need. Vietnam is making and testing its own AI vaccine to become independent on supply and to ensure the availability of the vaccine for a rapid response to an outbreak. However, it is difficult for a small country to compete with international pharmaceutical companies in the AI vaccine markets.
CONCLUSION
Through the Asian experience, it has become clear that AI vaccination would play an essential part in the control of the actual H5N1 pandemic in developing countries. To prevent any negative impact of illegal vaccination, international and national authorities must react quickly by implementing national vaccination strategies, especially in Africa where the first H5N1 outbreaks were declared in January 2006. The risk that the poultry industry and consumers will lose trust in vaccine safety and efficacy also is a concern. This would make the application of field vaccination even more difficult. Uncontrolled vaccination, including the improper distribution and administration of a vaccine and/or the use of bad vaccines, poses a greater threat in further outbreaks and raises the possibility of the potential mutation of the virus to become a pandemic pathogen.
ACKNOWLEDGEMENTS
The authors thank Les Sims for his useful comments and acknowledge the constructive comments of the anonymous reviewers of this paper. This work was partially supported by the FSP GRIPAVI project funded by the French Ministry of Foreign and European Affairs (MAEE).
DECLARATION OF INTEREST
None.









