Percutaneous closure of perimembranous ventricular septal defects has been gaining popularity as an off-label treatment of small and medium-sized defects. Reference Jiang, Han and Zhao1–Reference Mandal, Su and Pang3 Operators have been using various occluders in the past decade such as patent ductus arteriosus occluders, Konar Multifunctional ventricular septal defect occluders (Lifetech Scientific Corporation, China), Nit-Occlud Le VSD (PFM Medical, Germany), and Amplatzer vascular plug II (Abbott Cardiovascular System, USA) with encouraging results in most of the devices. Reference Yang, Yang and Yu2,Reference Cen, Peng, Li, Chen and Sun4,Reference Hua, Aquino and Owada5 Amplatzer Vascular Plug IV (Abbott Cardiovascular System, USA) is usually used for the occlusion of abnormal tortuous vessels and has not been tried for this application. In this report, we present three successful cases of transcatheter closure of wind-sock-like perimembranous ventricular septal defects using Amplatzer Vascular Plug IV with encouraging results.
Clinical cases
Case 1
An 8.6-year-old (23 kg / 126 cm) girl presented with a small-size perimembranous ventricular septal defect and mild left heart overload. The closure procedure was performed under general anaesthesia, transesophageal echocardiography, and fluoroscopy. A left ventriculogram was performed at 40° left anterior oblique/20° cranial projection and showed a solitary ventricular septal defect with suitable measurements for Amplatzer Vascular Plug IV (Table 1). Ultrasound showed that the perimembranous ventricular septal defect was partially closed by tricuspid valve aneurysmal tissue. The tissue formed a long, narrowed tunnel, mimicking a wind-sock appearance. The perimembranous ventricular septal defect was crossed retrogradely from the femoral artery using a 4-Fr Judkins right catheter (Technowood Corporation, Japan) and 0.035” Terumo guidewire (Terumo Corporation, Japan). After positioning the 4-Fr Amplatzer long delivery sheath in the right ventricle, the Amplatzer Vascular Plug IV was deployed sequentially in the defect’s aneurysmal sac. The device was released successfully after confirming the absence of a residual shunt and valvular complications.
Table 1. Summary of the characteristics of the patient and perimembranous ventricular septal defect at the time of intervention

Fr = French; FU = follow up; LVED = left ventricular entry diameter; PA = pulmonary artery; pmVSD = perimembranous ventricular septal defect; RVED = right ventricular exit diameter; Qp/Qs = ratio of the pulmonary to systemic flow.
Case 2
An 11.1-year-old boy (30.8kg/ 143 cm) presented with a hemodynamically significant perimembranous ventricular septal defect. A left ventricle angiogram revealed a solitary perimembranous ventricular septal defect with a size of 4 mm at its exit. Again, the morphology was that of a long-wind-sock aneurysmal sac (Fig. 1). The perimembranous ventricular septal defect was crossed retrogradely from the left ventricle using the 4F Judkins right and Terumo guide wire. A 6 mm Amplatzer Vascular Plug IV was deployed at the perimembranous ventricular septal defect sac. Upon releasing the device, angiography and transesophageal echocardiography showed no residual shunt, no aortic valve regurgitation, and no pericardial effusion. However, there was a mild de novo tricuspid regurgitation.

Figure 1. ( a ) Pre-procedure echocardiogram showing a ventricular septal defect with a windsock morphology which has a small mouth (4.8 mm in diameter) at the left ventricular end and a smaller 3.2 mm right ventricular exit. [Thick arrow]. The tricuspid valve is shown by the thin arrow. ( b ) Color Doppler image profiling the tricuspid valve aneurysmal sac. ( c ) Left ventricular angiography showing a perimembranous ventricular septal defect with an aneurysmal tissue which is mildly tortuous. ( d ) Left ventriculogram showing retrograde device implantation. The device was away from the aortic valve (white line). ( e ) The left ventricular lobe of the device was minimally protruding into the left ventricle. ( f ) Transesophageal echocardiogram showing the right ventricular lobe of the device protruding mildly into the right ventricle (*) while the left ventricular outflow tract (#) is not obstructed by the device.
Case 3
A 6.8-year-old boy (18.1kg/118 cm) presented with a perimembranous ventricular septal defect with mild symptoms. On the transesophageal echocardiogram, the perimembranous ventricular septal defect was 4 mm at the exit into the right ventricle with aneurysmal tissue. (Fig. 2) A ventricular septal aneurysm was present as well. A retrograde approach was adopted. A 6 mm Amplatzer Vascular Plug IV was used. Post-procedural echocardiogram revealed no complications, residual shunt or new onset of aortic or tricuspid valve regurgitation.
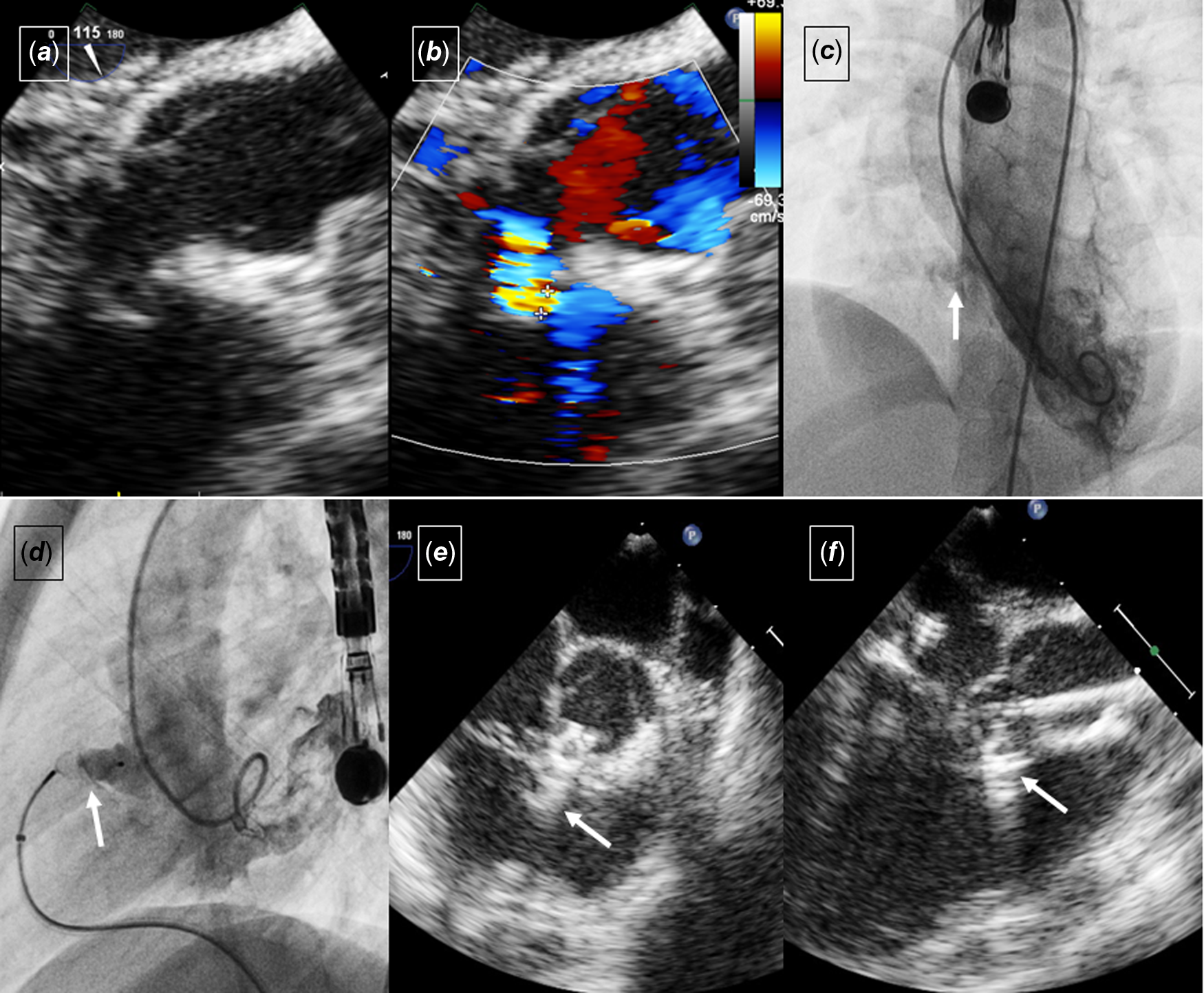
Figure 2. ( a ) Transesophageal echocardiography revealed a perimembranous ventricular septal defect with the morphology of a long windsock aneurysmal sac. ( b ) Color-compare image profiling the morphology of the perimembranous ventricular septal defect. ( c ) The left ventriculogram showed a long, narrowed tunnel aneurysmal sac (arrow). ( d ) Left ventricular angiography showed the device (arrow) sitting snugly in the sac without tension. ( e ) Transesophageal echocardiogram in the short axis showing the device in the sac (arrow) without protruding significantly into the right ventricle. ( f ) Transthoracic echocardiography of the left ventricular outflow tract revealed the left ventricular lobe of the Amplatzer Vascular Plug IV not protruding into the outflow tract.
The clinical features of the three cases are summarised in Table 1. All the patients did not have gross hematuria. However, urine analysis was not performed. The patients were followed up for a median duration of 47.2 months (range: 27.3–56.4 months). During follow up, the patients were noted to be well. They were free from syncope or palpitation Holter was not performed in the absence of symptoms. The regular ECGs during the follow up visits did not show the appearance of a new arrhythmia. There was no residual perimembranous ventricular septal defect shunt and no worsening or new appearance of the tricuspid or aortic valve regurgitation.
Discussion
Device closure of perimembranous ventricular septal defects has been widely performed, especially in developing countries. Reference Jiang, Han and Zhao1,Reference Yang, Yang and Yu2,Reference Wang, Zhuang and Zhang6,Reference Haddad, Daou and Saliba7 Most of these procedures were performed using “off-label” devices, which were designed for indications other than perimembranous ventricular septal defect closure. Device selection in perimembranous ventricular septal defects is a crucial aspect of the procedure. The device must be chosen based on several factors—the size, morphology of the perimembranous ventricular septal defect and the patient’s physiology. Unlike other ventricular septal defects, perimembranous ventricular septal defects are frequently associated with tricuspid valve aneurysm, which can lead to tricuspid valve regurgitation, partial closure of the perimembranous ventricular septal defect, and alteration of the defect morphology. Often, the tricuspid valve aneurysm has a broad base at the left ventricular end and tapers to a smaller exit at the right ventricular end. Such a morphology has a large base to accommodate a wide-body device and is suitable for devices with a retention disk. However, rarely, the tricuspid valve aneurysm forms a windsock-like tunnel, which has a small left ventricular entry with a long, narrow tunnel which mimics a wind-sock. The long narrow tunnel has limited space for a device with a wide retention disk such as the Konar-MF or the ductal occluder. Placing a bulky device may stretch the aneurysmal sac and potentially worsen the tricuspid valve regurgitation, hence an alternative device that conforms to the morphology of the sac should be used. Instead, the Amplatzer Vascular Plug IV is long, slender, and devoid of a retention disk and it conforms to the windsock-like tricuspid valve aneurysm. In addition, if the tricuspid valve aneurysm has multiple exits, one may attempt to occlude the whole sac with the device, obliterating the sac rather than merely the exit point. In order to accomplish this, the sac needs to be small enough to fit the occluder snugly.
The Amplatzer Vascular Plug IV is a good option for the closure of perimembranous ventricular septal defects with such morphology. Firstly, its spindle-shaped bilobed design is devoid of a retention disk. The conventional devices used in perimembranous ventricular septal defect closure have a retention disk, which is 2–3 mm larger than the body of the device. The absence of a retention disk averted the possibility of aortic cusp trapping in a perimembranous ventricular septal defect with an absent aortic rim and clapping onto the conduction fibres by the retention disk causing a complete atrioventricular heart block. Secondly, it is soft and can easily conform to the shape of the long tunnel tricuspid valve aneurysm. Thirdly, it is able to conform to a long tunnel with an angulated exit, a feature which is not uncommonly seen in perimembranous ventricular septal defect with tricuspid valve aneurysm. The device is suitable for the tortuous course of the tricuspid valve aneurysm. Lastly, Amplatzer Vascular Plug IV has the lowest profile and can be delivered through a 4–5F diagnostic catheter, which makes it an excellent device for retrograde deployment in very young children.
However, some limitations of the Amplatzer Vascular Plug IV exist. The size available is limited to 4 mm to 8 mm. As our strategy has been to either occlude the right ventricular exit point of the defect or to stent the whole sac, a device with a size at least 1.5 times larger than the defect would be needed. This implies that Amplatzer Vascular Plug IV can only be used in defects smaller than 5 mm given that the device has to be at least 50% larger in diameter than the target defect diameter. Ferro et al. Reference Ferro, Rossi, Bovio, Petrocelli and Seitun8 reported that Amplatzer Vascular Plug IV of 7 mm and 8 mm are a little challenging to manoeuvre within the delivery cable when angulated shapes are met. To ease this situation, the friction between the plug and the catheter wall can be reduced by continuously injecting normal saline through the delivery sheath or the delivery sheath can be upsized. All the cases were closed retrogradely. However, if the device were to be deployed from anterogradely, there is a potential challenge in estimating the precise spot to deploy the right ventricular lobe of the Amplatzer Vascular Plug IV. The issue can be resolved by defining the interventricular septum’s outline using right and left ventriculograms, and transesophageal echocardiography. Lastly, the absence of the retention disk may, hypothetically, increase the risk of device embolisation compared to an occluder with a retention disk.
Conclusion
In this report, we demonstrate that the closure of perimembranous ventricular septal defects with centrally narrowed aneurysmal sacs can be performed effectively with Amplatzer Vascular Plug IV. These preliminary results pave the way for the novel use of this occluder in this application. The plugs are, however, not suitable for aneurysmal perimembranous ventricular septal defects with an exit diameter larger than 5 mm.
Acknowledgement
The above authors take responsibility for all aspects of the reliability and freedom from bias of the data presented and their discussed interpretation.
Financial support
None.
Competing interests
XYH is a medical student attached to the Paediatric & Congenital Heart Centre for her elective posting.






