It is now estimated that over 20 % of adults in many Western countries have the metabolic syndrome with numbers continuing to increase(Reference Balkau, Vernay and Mhamdi1, Reference Golden, Robinson and Saldanha2). The metabolic syndrome is classified as a combination of risk factors such as obesity, atherogenic dyslipidaemia, hypertension, glucose intolerance and insulin resistance, which significantly increases the risk of developing CVD and type 2 diabetes mellitus(Reference Grundy, Brewer and Cleeman3, Reference Egger and Dixon4). Many studies, including the National Health and Nutrition Examination Survey (1999–2004), and the Coronary Artery Risk Development In Young Adults study have suggested that higher dairy consumption reduces the risk of obesity(Reference Beydoun, Gary and Caballero5, Reference Pereira, Jacobs and Van Horn6). It has been proposed that specific components of dairy, such as Ca, other minerals, casein or whey proteins(Reference Huth, DiRienzo and Miller7), may have beneficial effects on metabolic risk factors.
Milk contains two major protein groups: caseins and whey proteins. Caseins account for almost 80 % and whey accounts for 20 % of total protein in bovine milk(Reference Aimutis8). Casein and whey are both heterogeneous groups of proteins containing all the common amino acids, and are especially rich in the essential ones. Whey proteins contain globular proteins that can be isolated from whey, a by-product of cheese manufactured from cows' milk(Reference Aimutis8).
A study by Bowen et al. (Reference Bowen, Noakes and Clifton9) suggested that the whey and casein protein components of dairy appear to be more important for weight loss than Ca in overweight adults due to their high concentrations of branched-chain amino acids. However, there is little evidence of beneficial effects of casein beyond it being a good quality source of protein and having a possible hypotensive effect(Reference Yamamoto and Takano10–Reference Pal and Ellis12). In a separate article, we have shown that both whey and casein proteins consumed over 12 weeks significantly reduced diastolic and systolic blood pressure from baseline in overweight individuals; however, whey protein consumption also significantly reduced arterial stiffness(Reference Pal and Ellis12). Some studies demonstrate that dairy whey proteins have a better effect on appetite control than other protein sources such as egg and casein(Reference Hall, Millward and Long13–Reference Uhe, Collier and O'Dea15). In addition, convincing evidence indicates that dairy whey proteins and their bioactive components such as lactalbumin, angiotensin-converting enzyme inhibitor and branched-chain amino acids may have an insulinotrophic effect(Reference Belobrajdic, McIntosh and Owens16–Reference Nilsson, Holst and Björck20), hypotriacylglycerolaemic effect(Reference Kawase, Hashimoto and Hosoda21), muscle-sparing effect(Reference Layman and Baum22–Reference Layman24) and cholesterol-lowering effect(Reference Zhang, Guo and LeBlanc25). However, most of these studies using whey proteins have been conducted in healthy individuals or animals, with limited studies in overweight/obese individuals. Given the effect of whey proteins on appetite control, muscle sparing and lipid metabolism demonstrated previously in healthy adults, our hypothesis was that whey protein consumption would also have a beneficial effect on metabolic risk factors in overweight and obese individuals, a population highly susceptible to the metabolic syndrome. Therefore, the aim of the present study was to compare the effect of whey and casein consumption on lipids, insulin, glucose and body composition in overweight/obese individuals.
Methods and procedure
Subjects
Overweight and obese individuals between the ages of 18–65 years with a BMI between 25 and 40 kg/m2 were recruited through advertisements in the local newspapers and television exposure. Of the 380 who responded, 97 were eligible to participate and 89 commenced the study. The suitability assessments were conducted via telephone and online followed by screenings in person. Exclusion criteria included taking regular medications (such as lipid-lowering agents and hypertensives), kidney and liver diseases, cancer within the last 5 years, smoking, pregnancy or lactation, over two standard alcoholic drinks per day, type 1 or type 2 diabetes, liver or kidney diseases, and cardiovascular events in the last 6 months.
Study design and methodology
This was a randomised, parallel design study conducted over a 12-week period where subjects were randomised to one of the three groups, whey protein group, casein protein group or the control group (glucose), using a simple randomisation table. The subjects had no knowledge of the supplement type as they were all closely matched for taste and appearance. After randomisation, subjects were asked to consume one of the following supplement sachets mixed with 250 ml water twice a day for 12 weeks: whey protein isolate, sodium caseinate (both containing 27 g protein) or glucose control (27 g glucose; MG Nutritionals, Brunswick, Vic, Australia). The protein supplement compositions are given in Table 1. The kJ content of all supplement sachets was equal to 525 kJ per sachet. Subjects were instructed to take one sachet within 30 min before breakfast and one within 30 min before their evening meal. To monitor compliance, subjects were asked to record their consumption of the supplements by completing tailored calendar tick boxes and bringing their empty sachets to their 6- and 12-week visits.
Table 1 Protein supplement typical composition in two sachets per day (60 g)

All subjects were asked to maintain their usual dietary intake and physical activity levels during the study. Weighed food records were completed by subjects before commencement of the study to gain baseline intake data, and every 2 weeks of the study (weeks 2, 4, 6, 8 and 12) on two weekdays and one weekend day to assess compliance. To maintain isoenergetic diets, subjects were given individual instruction by the study dietitian to reduce their usual dietary intake by about 1050 kJ per day (equal to two sachets of supplements) which they recorded on the weighed food diary. This was monitored, and subjects were contacted if they were under- or over-compensating. All subjects were instructed to maintain dairy intake at one serve per day for 4 weeks leading up to and during the study period, to limit alcohol to two or less standard drinks for men and one or less standard drinks for women, and to not take any multivitamins or herbal supplements during the study period. All other aspects of their dietary intake were to remain unchanged. Energy and nutrient compositions were calculated using FoodWorks 2007 (Xyris Software, Highgate Hill, QLD, Australia) based on the Australian food composition tables.
Assessments
Subjects were assessed at baseline, 6 and 12 weeks. Body weight (UM-018 Digital Scales; Tanita Corporation, Tokyo, Japan) was recorded in light clothing without shoes. Height was measured to the nearest 0·1 cm using a stadiometer (26SM 200 cm SECA, Hamburg, Germany) without shoes. Waist circumference was measured three times in the standing position at the narrowest area between the lateral lower rib and the iliac crest, and the measures were averaged. Total body fat, lean mass, android fat (fat around the abdomen and trunk) and gynoid fat (fat around the hips, thighs and buttocks) as well as percentage fat were assessed by whole body dual-energy X-ray absorptiometry (Lunar Prodigy; Lunar, Madison WI, USA) at baseline and at 12 weeks.
Measurement of plasma lipids, insulin and homeostasis model assessment
An 8·5 ml fasting blood sample was taken via venepuncture from subjects in the fasting state, at baseline, 6 and 12 weeks, to measure circulating insulin, glucose and lipids (TAG, NEFA, HDL, LDL and apo B). Serum was isolated by centrifugation at 3000 rpm at 4 °C for 10 min, and was stored at − 80 °C until the end of the study. Serum TAG and total cholesterol (TC) were measured using enzymatic colorimetric kits (TRACE Scientific Limited, Noble Park, Vic, Australia). Serum HDL cholesterol was determined after precipitation of apo B-containing lipoproteins with phosphotungstic acid and MgCl2; HDL cholesterol present in the supernatant was determined by enzymatic colorimetry (TRACE Scientific Limited). Serum LDL cholesterol was determined using a modified version of the Friedewald equation(Reference Bairaktari, Hatzidimou and Tzallas26). NEFA were determined using WAKO NEFA C kit. Apo B was analysed using an ELISA kit obtained from Mabtech AB (Nacka Strand, Sweden). Plasma glucose levels were measured using the Randox glucose GOD–PAP kit (Antrim, UK), and plasma insulin was measured using an ELISA kit (Dako Diagnostic, Kyoto, Japan). The homoeostasis model of assessment index 2 was used to assess insulin resistance (homeostasis model assessment of insulin resistance) from fasting glucose and insulin concentrations(Reference Wallace, Levy and Matthews27).
Statistical analysis
All subjects who completed the study were included in the data analysis. Statistical analysis was conducted using SPSS 17 for Windows (SPSS, Inc., Chicago, IL, USA). Data are expressed as mean values with their standard errors, and were assessed for normality. Comparison of baseline characteristics between each group was undertaken by one-way ANOVA. Differences within groups were determined using a two-sided paired t test. Using one-way analysis of covariance with the baseline data as the covariate, differences between groups were determined at week 6 to assess immediate effects of intervention and at week 12 to assess longer term effects of supplementation. Statistical differences were analysed further by post hoc analysis using the least square differences method. Percentage change between groups was calculated based on raw values of group means. Statistical significance was considered at P < 0·05.
The present study was conducted according to the guidelines laid down in the Declaration of Helsinki, and all procedures involving human subjects/patients were approved by the Curtin University Human Research Ethics Committee (Approval Number HR 149/2007). Written informed consent was obtained from all subjects.
This clinical trial has been registered with the Australian New Zealand Clinical Trials Registry. The registration number is ACTRN12609000175279, and trial Web address is http://www.ANZCTR.org.au/ACTRN12609000175279.aspx
Results
Eighty-nine individuals were randomly assigned to either a control, casein or whey protein supplement for 12 weeks. Nineteen subjects withdrew from the study within 4 weeks after randomisation due to non-compliance for various reasons as shown in Fig. 1. Seventy subjects completed the 12-week study (control group: n 25, twenty-two women and three men; casein group: n 20, seventeen women and three men; whey protein group: n 25, twenty-one women and four men). Subject characteristics in the three treatment arms at baseline were not significantly different.

Fig. 1 Details of subject recruitment and withdrawal.
The self-reported composition of the study diets consumed during the 3-month study period is presented in Table 2. There were no significant differences in total energy, total fat, saturated fat, monounsaturated fat, polyunsaturated fat, Ca and fibre between control, casein and whey protein groups (Table 2). The contribution of carbohydrate to total energy intake was lower in the casein group (P < 0·001) and whey group (P < 0·001) compared with the control group. The contribution of protein to total energy intake was higher in subjects in the casein group (P < 0·001) and whey group (P < 0·001) compared with the control group.
Table 2 Nutritional data recorded in 3 d food diaries
(Mean values with their standard errors, n 70)
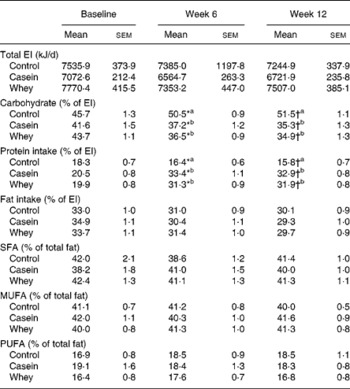
EI, energy intake.
a,b Mean values with unlike superscript letters were significantly different between groups at 6 and 12 weeks (P < 0·05).
* Mean values were significantly different within group (time 0 v. time 6 weeks, P < 0·05).
† Mean values were significantly different within group (time 0 v. time 12 weeks, P < 0·05).
Changes in body composition (Table 3) were assessed by body weight, waist and hip circumference measurements and body composition scans by dual-energy X-ray absorptiometry. There was no overall effect of supplements on body weight, BMI, waist, waist:hip ratio, total body fat, android fat, gynoid fat and lean body mass between groups at 6 or 12 weeks, with no difference within groups.
Table 3 Body composition measurements*
(Mean values with their standard errors)

* There were no significant differences at baseline between diets. There were no significant differences between groups.
Table 4 shows the mean fasting levels of all blood measurements from each intervention group at the three visits, and summarises the significant within-group and between-group differences. Fasting TAG concentrations decreased by 13 % at week 6 (P = 0·008) and week 12 (P = 0·003) compared with baseline in the whey protein group. Fasting TAG levels were lowered by 22 % in the whey protein group compared with the control group at 6 weeks (P = 0·025) and by 22 % at 12 weeks (P = 0·035).
Table 4 Concentration of plasma lipids, lipoproteins, glucose and NEFA*
(Mean values with their standard errors)

TC, total cholesterol.
a,b Mean values with unlike superscript letters were significantly different between groups at 12 weeks (P < 0·05).
* There were no significant differences at baseline between diets.
† Mean values were significantly different within group (time 0 v. time 6 weeks, P < 0·05).
‡ Mean values were significantly different within group (time 0 v. time 12 weeks, P < 0·05).
TC levels in plasma (Table 4) were decreased by 7 % at week 12 in the whey group compared with baseline (P < 0·001). There was a lowering in TC at week 6 in the whey protein group compared with the control group (P = 0·044). There was a decrease in TC by 9 % at week 12 in the whey protein group compared with the casein group (P = 0·026) and by 11 % compared with the control group (P < 0·001). Plasma LDL was reduced by 7 % at week 12 in the whey group compared with baseline (P = 0·007). There was an attenuation in plasma LDL levels at week 12 in the whey protein group compared with the casein (P = 0·045) and control groups (P = 0·003).
There was an 11 % reduction in plasma insulin levels (Fig. 2(a)) in the whey protein group at 12 weeks (41·71 (sem 3·80) pmol/l) compared with baseline (46·96 (sem 3·76) pmol/l; P = 0·012). There was also a lowering in fasting insulin levels in the whey protein group (41·71 (sem 3·80) pmol/l) compared with the control group (54·77 (sem 5·185); P = 0·049) at 12 weeks. There was a decrease in homeostasis model assessment of insulin resistance scores at 12 weeks in the whey protein group (0·82 (sem 0·08)) compared with baseline (0·91 (sem 0·07); P = 0·046) (Fig. 2(b)). There was a decrease in homeostasis model assessment of insulin resistance scores in the whey treatment group (0·82 (sem 0·08)) compared with the control group at 12 weeks (1·01 (sem 0·10); P = 0·034).

Fig. 2 Changes in insulin (a) and homeostasis model assessment of insulin resistance (HOMA-IR, b) scores. The change in insulin and HOMA-IR from baseline to 6 and 12 weeks following consumption of control, casein or whey protein supplement. Data are mean values with their standard errors. * Mean value was significantly different from that at baseline (P < 0·05). a,b Mean values with unlike letters were significantly different (P < 0·05). ![]() , Control;
, Control; ![]() , casein;
, casein; ![]() , whey.
, whey.
Discussion
Previous studies have demonstrated the effect of whey proteins on appetite control, muscle sparing and lipid metabolism in healthy adults, but limited data are available for the effect of whey protein consumption on metabolic risk factors in overweight and obese individuals. In the present study, supplementation with whey protein for 12 weeks decreased TC and LDL cholesterol levels compared with the supplementation with control and casein. Whey protein also decreased plasma TAG, insulin and homeostasis model assessment of insulin resistance scores compared with the control. There was no effect of casein supplementation on any metabolic risk parameter compared with control supplementation. Overall, the present study demonstrated that whey protein supplementation can significantly improve metabolic risk factors associated with chronic diseases in overweight and obese individuals.
Supplementation with whey protein significantly lowered plasma TC and LDL cholesterol after 12 weeks compared with the supplementation with casein and control. Interestingly, a decrease in LDL cholesterol levels with whey protein was observed without changes in apo B levels, indicating a change in LDL particle size rather than in particle number. Lipid-lowering agents such as statins, fibric acid and nicotinic acid have been shown to lower LDL levels from 5 to 55 %, resulting in a CHD risk reduction of 25–45 % with 5 years of treatment in randomised, placebo-controlled clinical trials(28). A meta-analysis by Baigent et al. (Reference Baigent, Keech and Kearney29) found that over 5 years, every mmol/l reduction in LDL cholesterol by statin therapy resulted in a 12 % reduction in all-cause mortality and a 19 % reduction in coronary mortality. Similarly, a 20–24 % reduction in TAG levels has been shown to reduce the progression of CHD(Reference Miller30). Therefore, a reduction in serum TC by 11 %, in LDL cholesterol by 9·6 % and in TAG by 22 % after 12 weeks of whey supplementation in the present study may also be clinically significant. The effect of whey proteins on lipid levels in the present study is consistent with earlier animal experiments mostly undertaken in rats(Reference Kawase, Hashimoto and Hosoda21, Reference Sautier, Dieng and Flament31, Reference Nagaoka, Kanamaru and Kuzuya32) and healthy men(Reference Kawase, Hashimoto and Hosoda21).
In the present study, whey protein consumption did not change body composition. Therefore, the beneficial changes in TC, LDL cholesterol and TAG from whey supplementation seem to have occurred independently of changes in body weight or fat mass. The mechanisms behind the favourable effects on lipids may be related to the effect of whey proteins on de novo cholesterol biogenesis in the liver(Reference Zhang and Beynen33), the inhibition of cholesterol absorption in the intestine mediated by β-lactoglobulin(Reference Nagaoka, Kanamaru and Kuzuya32), the inhibition of the expression of genes involved in intestinal fatty acid and cholesterol absorption and synthesis(Reference Chen and Reimer34) and/or the increase in faecal steroid excretion(Reference Lovati, West and Sirtori35).
The differential effects on lipid metabolism between whey and casein may be related to their specific influence on digestion and absorption and their amino acid content(Reference Hall, Millward and Long13, Reference Boirie, Dangin and Gachon36). Whey proteins have been shown to have a fast rate of digestion and absorption producing a rapid peak in plasma amino acids compared with casein. Whey also has a higher branched-chain amino acids content compared with casein. Unlike whey, casein is a coagulating protein and exhibits a slower rate of gastric emptying and also mediates lower postprandial excursions of plasma amino acid concentrations(Reference Hall, Millward and Long13, Reference Boirie, Dangin and Gachon36). Casein has been shown numerous times to produce higher levels of serum cholesterol and LDL when compared with soya proteins, and this is thought to be due to its unusually low content of cysteine(Reference Castiglioni, Manzoni and D'Uva37, Reference Atwal, Kubow and Wolynetz38).
In the present study, whey protein supplementation decreased plasma insulin levels by 11 % at 12 weeks and improved insulin sensitivity. Insulin-resistant rats were fed a high-protein diet for 6 weeks, either whey or red meat protein; the whey protein concentrate reduced plasma insulin by 40 % and increased insulin sensitivity(Reference Belobrajdic, McIntosh and Owens16). These changes were explained by the reduction in visceral fat in the rats fed the whey protein diet, because visceral obesity is strongly correlated with insulin resistance. However, in the present study, there were no changes in body fat to explain changes in insulin sensitivity. Interestingly, postprandial studies have demonstrated that whey proteins have a stimulating effect on insulin secretion in healthy subjects(Reference Nilsson, Stenberg and Frid18) and in diabetics(Reference Frid, Nilsson and Holst17). In healthy subjects, ingestion of a mixture of leucine, isoleucine, valine, lysine and threonine resulted in glycaemic and insulinaemic responses similar to those resulting after whey ingestion(Reference Nilsson, Holst and Björck20), suggesting that branched-chain amino acids are the major determinants of insulinaemia as well as lowered glycaemia. The key mechanism is not known, although elevated concentrations of specific insulinogenic amino acids as well as bioactive peptides, either originally present in whey or formed during digestion, are possible. The short-term insulinotrophic effect of whey proteins may be a valuable tool in the management of type 2 diabetes or the metabolic syndrome. Today, sulphonylurea agents are commonly used to stimulate insulin secretion and to attenuate postprandial blood glucose for the purpose of facilitating normoglycaemia in diabetic subjects. However, further studies are required to resolve why whey proteins can have an insulinotrophic effect in the short term, but can decrease plasma insulin levels and improve insulin sensitivity in the long term.
A study limitation involved the self-reporting of energy intake by the study participants. Mean intakes of about 7000 kJ as recorded in the present study would not represent the true energy intake of overweight or obese individuals on an isoenergetic diet. However, it is well established that underreporting of energy intake in this population is prevalent(Reference Singh, Martin and Hickey39–Reference Voss, Kroke and Klipstein-Grobusch41), and is an accepted study limitation. Despite the underreporting, analysis of the weighed food records enabled monitoring of any changes in energy intake and macronutrient as well as dairy product intake.
In the present study, most subjects had a waist circumference greater than that specified by the ATP III Clinical Identification of Metabolic Syndrome(Reference Grundy, Brewer and Cleeman3). In addition, most subjects already had at least one or two other metabolic syndrome risk factors or were borderline candidates. Many in this group will eventually progress to establish the metabolic syndrome if their obesity continues. As the consumption of whey proteins in this group has beneficial effects on serum lipids and insulin, regular whey protein supplementation may, at the very least, slow down the progression to the metabolic syndrome, CVD or diabetes, and/or may delay the need for medications. Collectively, whey protein has the potential to be used as an added component in dietary plans and in functional foods aimed at the management of chronic diseases in overweight and obese individuals.
Acknowledgements
The present study is a Dairy Australia initiative funded by the Dairy Service Levy and the Australian Government (grant no. CUT11913). We thank Murray Goulburn Nutritionals (Vic, Australia) for providing the supplements. The authors declare that they have no competing interests. The authors' responsibilities were as follows: V. E. coordinated the trial, data collection and input for the manuscript. S. P. conceived and designed the study, wrote the manuscript, and supervised the study and the statistical analysis; S. D. provided support with the statistical analysis and input for the manuscript. All authors have read and approved the final manuscript.








