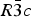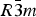Since Boeke's finding of a reversible phase transition of calcite (calcium carbonate, CaCO3) at elevated temperatures [Boeke, H. E. (1912). Neues Jahrb. Mineral. 1, 91–121], and following W. L. Bragg's determination of the structure of the room-temperature Phase I [Bragg, W. L. (1914). Proc. R. Soc. Lond. A 89, 468–489.], the high-temperature Phase V of calcite has been an enduring mystery. Here, we summarize a paper on the structure of Phase V [Ishizawa, N., Setoguchi, H. and Yanagisawa, K. (2013). Sci. Rep. 3, 2832], as well as the intermediate Phase IV which exists between Phases I and V, and add new aspects. An in situ single-crystal X-ray diffraction study revealed that the I–IV and IV–V transitions occurred reversibly at approximately 985 and 1240 K, respectively, in a carbon dioxide atmosphere. Phase V was stable only over a narrow temperature range between 1240 and 1275 K. The crystal decomposed immediately at temperatures above 1275 K, leaving a nanoporous calcium oxide reaction product which retained the shape of the parent calcite crystal. The I–IV transition can be described as an orientational order/disorder transition of the carbonate group, occurring within the same space group 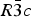 $R\bar 3c$. In Phase V, the oxygen sublattice is melted. The joint-probability density function obtained from the anharmonic atomic displacement parameters of the oxygen atoms revealed that the oxygen triangles of the carbonate group in Phase V do not sit still at specified Wyckoff positions in the space group
$R\bar 3c$. In Phase V, the oxygen sublattice is melted. The joint-probability density function obtained from the anharmonic atomic displacement parameters of the oxygen atoms revealed that the oxygen triangles of the carbonate group in Phase V do not sit still at specified Wyckoff positions in the space group 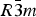 $R\bar 3m$, but are instead distributed with equal probability along the undulated circular orbital about the central carbon. The carbonate group in Phase V is no longer flat on the basal plane when the oxygen triangle comes to troughs or peaks in the undulated orbital, but is instead deformed like an umbrella. Assuming that the oxygen triangle migrates about carbon, the carbonate group should repeat the umbrella inversion in Phase V as a function of time. Finally, possible thermal decomposition mechanisms of calcite are briefly discussed.
$R\bar 3m$, but are instead distributed with equal probability along the undulated circular orbital about the central carbon. The carbonate group in Phase V is no longer flat on the basal plane when the oxygen triangle comes to troughs or peaks in the undulated orbital, but is instead deformed like an umbrella. Assuming that the oxygen triangle migrates about carbon, the carbonate group should repeat the umbrella inversion in Phase V as a function of time. Finally, possible thermal decomposition mechanisms of calcite are briefly discussed.