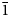Introduction
Divanadate minerals are not numerous in Nature. They are mainly represented by pyrovanadates, the oxysalts with anionic [V2O7]4– groups isolated from each other and composed of two V5+-centred tetrahedra which share a bridging O atom. Among a dozen such minerals only volborthite, known since the 1830s, is relatively widespread. Typically, pyrovanadates (and natural divanadates in general) are hydrous minerals formed in supergene environments or in late, low-temperature hydrothermal assemblages. Except for volborthite Cu3V2O7(OH)2⋅2H2O (Kashaev and Bakakin, Reference Kashaev and Bakakin1968; Vladimirova et al., Reference Vladimirova, Siidra, Ugolkov and Bubnova2021 and references therein) and the related minerals martyite Zn3V2O7(OH)2⋅2H2O (Kampf and Steele, Reference Kampf and Steele2008) and karpenkoite Co3V2O7(OH)2⋅2H2O (Kasatkin et al., Reference Kasatkin, Plášil, Pekov, Belakovskiy, Nestola, Čejka, Vigasina, Zorzi and Thorne2015), there are fianelite Mn2(V,As)2O7⋅2H2O (Brugger and Berlepsch, Reference Brugger and Berlepsch1996), engelhauptite KCu3(V2O7)(OH)2Cl (Pekov et al., Reference Pekov, Siidra, Chukanov, Yapaskurt, Britvin, Krivovichev, Schüller and Ternes2015), mesaite CaMn2+5(V2O7)3⋅12H2O (Kampf et al., Reference Kampf, Nash, Marty and Hughes2017) and donowensite CaFe3+2(V2O7)2⋅3H2O (Kampf et al., Reference Kampf, Hughes, Nash and Smith2022). For pintadoite the formula Ca2V2O7⋅9H2O is suggested, however, this mineral is poorly studied and has the IMA status Q – questionable (Pasero, Reference Pasero2023). The H-free natural pyrovanadates are represented by chervetite Pb2V2O7 (Bariand et al., Reference Bariand, Chantret, Pouget and Rimsky1963; Shannon and Calvo, Reference Shannon and Calvo1973) and two modifications of Cu2V2O7 – blossite and ziesite (Hughes and Birnie, Reference Hughes and Birnie1980; Robinson et al., Reference Robinson, Hughes and Malinconico1987; Hughes and Brown, Reference Hughes and Brown1989; Krivovichev et al., Reference Krivovichev, Filatov, Cherepansky, Armbuster and Pankratova2005); kainotropite Cu4Fe3+O2(V2O7)(VO4) is the only known mineral containing both pyrovanadate (V2O7)4– and orthovanadate (VO4)3– anions (Pekov et al., Reference Pekov, Zubkova, Yapaskurt, Polekhovsky, Britvin, Turchkova, Sidorov and Pushcharovsky2020). Blossite, ziesite and kainotropite are endemics of volcanic fumaroles, as well as the new anhydrous calcium vanadate bakakinite (Cyrillic: бакакинит) described in the present paper. Bakakinite has the ideal, end-member formula Ca2V2O7 and is a divanadate in terms of chemistry, however, from the crystal chemical viewpoint, it is not a pyrovanadate. This mineral contains more complex vanadate anionic groups – isolated tetramers [V4O14]8– built from 4-fold (tetrahedra) and 5-fold V5+-centred polyhedra.
The new mineral is named in honour of the outstanding Russian crystallographer and crystal chemist Professor Vladimir Vasilievich Bakakin (born 1933) who works in Nikolaev Institute of Inorganic Chemistry of the Siberian Branch of the Russian Academy of Sciences, Novosibirsk. Prof. Bakakin made a significant contribution to the field of structural mineralogy. In particular, Kashaev and Bakakin (Reference Kashaev and Bakakin1968) first determined the crystal structure of volborthite and showed that this mineral is a pyrovanadate and not an ortho-vanadate as it was assumed before.
Both the new mineral and its name (symbol Bkkn) have been approved by the Commission on New Minerals, Nomenclature and Classification of the International Mineralogical Association (IMA2022-046, Pekov et al., Reference Pekov, Agakhanov, Koshlyakova, Zubkova, Yapaskurt, Britvin, Vigasina, Turchkova and Nazarova2022a). The holotype specimen is deposited in the systematic collection of the Fersman Mineralogical Museum of the Russian Academy of Sciences, Moscow with the catalogue number 98012.
Occurrence and general appearance
The specimens with the new mineral were collected by us in July 2021 from the Arsenatnaya fumarole, Second scoria cone of the Northern Breakthrough of the Great Tolbachik Fissure Eruption, Tolbachik volcano, Kamchatka peninsula, Far-Eastern Region, Russia, 55°41’N, 160°14’E, 1200 m elevation. This active fumarole that contains very rich and diverse high-temperature sublimate mineralisation has been described by Pekov et al. (Reference Pekov, Koshlyakova, Zubkova, Lykova, Britvin, Yapaskurt, Agakhanov, Shchipalkina, Turchkova and Sidorov2018) and Shchipalkina et al. (Reference Shchipalkina, Pekov, Koshlyakova, Britvin, Zubkova, Varlamov and Sidorov2020).
Bakakinite was found in several open pockets at the deepest (depths of 3–4 m under the day surface) and hottest zone of Arsenatnaya. The temperatures measured using a chromel–alumel thermocouple in these pockets during sampling varied from 430 to 490°C. We believe that bakakinite crystallised at temperatures not lower than 500°C. It can be deposited directly from hot gas as a volcanic sublimate, however, in our opinion, it seems more probably that the mineral was formed as a result of the interaction between fumarolic gas and basalt scoria. The latter could be a source of calcium which has very low volatility in such post-volcanic systems (Symonds and Reed, Reference Symonds and Reed1993).
Bakakinite occurs, sporadically in significant amounts, as a constituent of polymineralic exhalation incrustations together with anhydrite, svabite, pliniusite, schäferite, berzeliite, diopside, hematite and powellite. Minor amounts of baryte, fluorapatite, calciojohillerite, ludwigite, magnesioferrite, anorthite, titanite and esseneite also occur in this mineral assemblage. Cavernous polymineralic aggregates containing up to 25 vol.% bakakinite form thin (usually not thicker than 0.02 mm) crusts up to several cm2 in area on anhydrite crystal crusts that cover basalt scoria altered by fumarolic gas to aggregates mainly consisting of diopside and hematite.
Bakakinite forms flattened crystals typically not larger than 10 μm, rarely up to 30 μm across and up to 5 μm thick. Some crystals are well-formed and complicated in shape (Fig. 1a), however, commonly bakakinite crystals are crude and distorted. The crystals, even with a well-developed outer shape, have skeletal (Fig. 1b) and/or blocky characters. Epitactic bakakinite overgrowths on svabite were observed (Fig. 1b). Clusters of bakakinite crystals are up to 0.1 mm across; in such clusters, bakakinite is intimately intergrown with other minerals, usually with anhydrite and svabite–pliniusite series members.

Figure 1. Morphology of crystals and aggregates of bakakinite and its relations with associated minerals: (a) flattened crystals of bakakinite (1) on pliniusite (2) with crystals of powellite (3) and diopside (4); (b) epitactic overgrowths of bakakinite skeletal crystals (1) on svabite (5) in association with anhydrite crusts (6) and schäferite crystals (7). Scanning electron microscopy image – back-scattered electron mode.
Physical properties and optical data
Bakakinite is transparent, colourless or pale yellow, with a white streak and strong vitreous lustre. It is brittle, cleavage or parting was not observed. The fracture is uneven (observed under the scanning electron microscope). The density value calculated using the averaged empirical formula is 3.463 g cm–3.
The new mineral is transparent and optically anisotropic, however, its optical studies were carried out in reflected light due to high refractive indices. The mean refractive index calculated on the basis of the Gladstone–Dale equation is 1.93.
Under the microscope in reflected light, bakakinite is grey; pleochroism was not observed. Bireflectance is weak, ΔR = 1.2% (589 nm). Anisotropy is very weak; internal reflections were not observed. The reflectance values measured in air using the SiC standard (Zeiss, No. 545) are given in Table 1.
Table 1. The reflectance data of bakakinite.

The values for wavelengths (λ) recommended by the IMA Commission on Ore Mineralogy are marked in boldtype.
Chemical composition
The composition of bakakinite was studied by electron microprobe using a Jeol JSM-6480LV scanning electron microscope equipped with an INCA-Wave 500 wavelength-dispersive spectrometer (Laboratory of Analytical Techniques of High Spatial Resolution, Dept. of Petrology, Moscow State University), with an acceleration voltage of 20 kV, a beam current of 20 nA and a 3 μm beam diameter. The standards used are listed in Table 2. Contents of other elements with atomic numbers >6 were below detection limits.
Table 2. Chemical composition of bakakinite (in wt.%).

S.D. = standard deviation
The chemical composition of bakakinite in wt.% is given in Table 2. The empirical formula calculated on the basis of 7 O atoms per formula unit is (Ca1.99Sr0.01)Σ2.00(V1.64As0.28P0.06Si0.01S0.01)Σ2.00O7. The simplified formula is Ca2(V,As)2O7. The ideal, end-member formula of bakakinite is Ca2V2O7 which requires CaO 38.14, V2O5 61.86, total 100 wt.%.
X-ray crystallography
Attempts to obtain single-crystal X-ray diffraction (XRD) data for bakakinite were unsuccessful due to the small size and low quality (skeletal and/or blocky character) of crystals.
Powder XRD data (Table 3) were collected with a Rigaku R-AXIS Rapid II single-crystal diffractometer equipped with cylindrical image plate detector (radius 127.4 mm) using Debye-Scherrer geometry, CoKα radiation (rotating anode with VariMAX microfocus optics), 40 kV, 15 mA and exposure of 15 min. Angular resolution of the detector is 0.045 2θ (pixel size 0.1 mm). The data were integrated using the software package Osc2Tab (Britvin et al., Reference Britvin, Dolivo-Dobrovolsky and Krzhizhanovskaya2017).
Table 3. Powder X-ray diffraction data (d in Å) of bakakinite and synthetic Ca2V2O7.

* The powder X-ray diffraction pattern also contains lines with d = 3.492 [a], 2.904 [s], 2.842 [a,s], 2.822 [s] and 1.875 [a,s] Å which are overlapped reflections of bakakinite and anhydrite [a], svabite [s] or both these minerals [a,s]. The strongest reflections are marked in boldtype.
Despite the absence of single-crystal XRD data, it is clear that bakakinite is a natural analogue of a well-known synthetic calcium divanadate, Ca2V2O7, of which the crystal structure was reported by Trunov et al. (Reference Trunov, Velikodnyi, Murasheva and Zhuravlev1983) and Tong et al. (Reference Tong, Luo, Jin and Lin2011). Intimate intergrowths of bakakinite with other minerals hampered the Rietveld refinement of the crystal structure, however, its structural identity with synthetic Ca2V2O7 found from powder XRD data is clear: see Discussion and Tables 3 and 4. Bakakinite is triclinic, with space group P $\bar{1}$![]() . The unit-cell parameters are reported in Table 4.
. The unit-cell parameters are reported in Table 4.
Table 4. Crystal data of bakakinite and synthetic Ca2V2O7 and Ca2As2O7.

Raman spectroscopy
The Raman spectrum of bakakinite (Fig. 2) was obtained on a randomly oriented crystal using an EnSpectr R532 instrument with a green laser (532 nm) at room temperature. The output power of the laser beam was ~6 mW. The spectrum was processed using the EnSpectr expert mode program in the range from 4000 to 100 cm–1 with the use of a holographic diffraction grating with 1800 lines mm–1 and spectral resolution was 6 cm–1. The diameter of the focal spot on the sample was ~10 μm. The back-scattered Raman signal was collected with a 60× objective; signal acquisition time for a single scan of the spectral range was 1000 ms and the signal was averaged over 30 scans.

Figure 2. The Raman spectrum of bakakinite.
The assignment of bands in the Raman spectrum of bakakinite can be performed based on the data reported by Griffith and Wickins (Reference Griffith and Wickins1966), Nakamoto (Reference Nakamoto1986), Hardcastle and Wachs (Reference Hardcastle and Wachs1991), Russu (Reference Russu2008) and Chong et al. (Reference Chong, Aksenov, Dal Bo, Perry, Dimakopoulou and Burns2019) and taking into account the presence of a complex anionic group [V4O14]4– in Ca2V2O7 (Trunov et al., Reference Trunov, Velikodnyi, Murasheva and Zhuravlev1983; Tong et al., Reference Tong, Luo, Jin and Lin2011), a synthetic end-member analogue of the mineral.
The Raman spectrum of synthetic Ca2V2O7 (= Ca4[V4O14]: for the structure data see below) reported by Russu (Reference Russu2008) is similar to the spectrum of bakakinite in its general pattern. However, unlike this synthetic vanadate, bakakinite contains admixed arsenic which partially substitutes vanadium. We cannot clearly identify the bands corresponding to As5+–O vibrations due to overlap with bands of V5+–O vibrations (Nakamoto, Reference Nakamoto1986), however, it is necessary to take into account the effect of the As admixture on the Raman spectrum. This probably results in the broadening of some bands in comparison with the spectrum of synthetic Ca2V2O7 (Russu, Reference Russu2008), but also other features. The position of an intense sharp band corresponding to IVM–O–VM (superscript Roman numerals mean coordination numbers) stretching vibrations, which is at 944 cm–1 in a pure vanadate synthetic compound (M = V5+) and at 928 cm–1 in bakakinite (M = V5+ > As5+). A band at 928 cm–1 is related to ν(VV5+–O–IVAs5+) and ν(VV5+–O–IVV5+), wide bands with maxima at 872, 845 cm–1 and at 789 and 775 cm–1 are assigned to symmetric (ν1) and asymmetric (ν3) vibrations, respectively, in (VO4)3–, [V2O8]6– and (AsO4)3– groups. A strong band at 695 cm–1 and a weak band at 527 cm–1 correspond to vibrations of the (V–O–V) and (V–O–As), and the low-frequency shoulder of a band at 695 cm–1 may be connected with vibrations in [V2O8]6–. The group of bands in the region of 480–300 cm–1 corresponds to overlapping split bending modes δ2 and δ4 of (VO4)3– and (AsO4)3– tetrahedra and bending modes in [V2O8]6– groups. The bands with frequencies below 300 cm–1 are assigned to translational modes of calcium cations (Ca–O) and lattice modes.
The Raman spectrum of bakakinite was obtained from a small (20 μm) flattened grain which overgrows pliniusite Ca5(VO4)3F. The strongest band in the Raman spectra of pliniusite occurs at 868–873 cm–1 and the next in intensity band is situated at 350–356 cm–1 (Pekov et al., Reference Pekov, Koshlyakova, Zubkova, Krzątała, Belakovskiy, Galuskina, Galuskin, Britvin, Sidorov, Vapnik and Pushcharovsky2022b). In the above-described bakakinite spectrum, we observe a strong broad band at 872 cm–1 and a weak low-frequency shoulder of a broad band with maximum at 386 cm–1. We cannot exclude that the broadening of these bands may be a result of, in addition to the effect of admixed As5+, the overlap of spectral bands of bakakinite (prevailing) and pliniusite (admixed).
Discussion
The ideal formula of bakakinite is Ca2V2O7. However, all electron-microprobe analyses demonstrate some arsenic admixture. Synthetic Ca2V2O7 (Trunov et al., Reference Trunov, Velikodnyi, Murasheva and Zhuravlev1983; Tong et al., Reference Tong, Luo, Jin and Lin2011) and Ca2As2O7 (Pertlik, Reference Pertlik1980) are not isotypic. They significantly differ from one another in symmetry, unit-cell metrics (Table 4) and crystal structure (Fig. 3). In both structures the layers of Ca cations are connected via anionic units built by V5+- or As5+-centred polyhedra. Noteworthy, in the papers on synthetic Ca2V2O7, the descriptions of Ca-centred polyhedra are slightly different: Trunov et al. (Reference Trunov, Velikodnyi, Murasheva and Zhuravlev1983) characterised them as nine- and eight-fold polyhedra whereas Tong et al. (Reference Tong, Luo, Jin and Lin2011) described both crystallographically non-equivalent Ca polyhedra as nine-fold ones. The analysis of interatomic Ca–O distances shows that strongly elongated Ca–O distances (more than 2.9 Å) were included in the coordination spheres of Ca in both papers. For clarity and better comparison with Ca2As2O7, we omit strongly elongated Ca–O distances in the description below and in drawings given in Fig. 3. Following this approach, Ca cations in Ca2V2O7 have distorted octahedral and seven-fold oxygen coordination whereas in Ca2As2O7 calcium cations are located only in distorted octahedra. In both structures edge-sharing Ca-centred polyhedra form layers, but these layers in Ca2V2O7 and Ca2As2O7 strongly differ topologically (Figs 3c,d). The anionic units in these compounds are also quite different in arrangement and coordination of V and As atoms. In Ca2As2O7, As5+ has only tetrahedral coordination whereas V5+ cations in Ca2V2O7 centre tetrahedra and five-fold polyhedra (distorted trigonal bipyramids). In the structure of Ca2V2O7 we see a linear anionic cluster, tetramer [V4O14]8–, which is built of two edge-connected distorted trigonal bipyramids VO5 (core of the cluster) and two VO4 tetrahedra connected with these trigonal bipyramids via common vertices (‘wings’ of the cluster) (Fig. 3e). For this reason, Tong et al. (Reference Tong, Luo, Jin and Lin2011) wrote the formula of this vanadate as Ca4V4O14 rather Ca2V2O7. In the structure of Ca2As2O7, arsenate tetrahedra form pyrogroups [As2O7]4–, isolated from each other (Fig. 3f), i.e. it is a pyroarsenate.

Figure 3. The crystal structures of synthetic analogue of bakakinite, Ca2V2O7 (left column: drawn after Trunov et al. (Reference Trunov, Velikodnyi, Murasheva and Zhuravlev1983); the unit cell is outlined) and Ca2As2O7 (right column: drawn after Pertlik (Reference Pertlik1980); the unit cell is outlined): general view (a, b), the layers of Ca-centred polyhedra (c, d) and the arrangement of anionic [V4O14]8– tetramer units (e) and [As2O7]4– pyrogroups (f).
The presence of pentacoordinated V5+ in Ca2V2O7 prevents its isotypism with the formula analogues, numerous natural and synthetic compounds Me 2T 2O7 (T = P, As and Si) in which T has only tetrahedral coordination. At the same time, our electron-microprobe analyses show that bakakinite contains a distinct As admixture (Table 2) and, thus, we suggest that Ca2V2O7 and Ca2As2O7 can form a solid-solution series, at least, in the V-dominant region. The structure determinations were performed only for synthetic end-members (Pertlik, Reference Pertlik1980; Trunov et al., Reference Trunov, Velikodnyi, Murasheva and Zhuravlev1983; Tong et al., Reference Tong, Luo, Jin and Lin2011) and, thus, we do not know which chemical composition in this hypothetic series corresponds to the point of transition from the Ca2V2O7 structure type (Fig. 3a) to the Ca2As2O7 type (Fig. 3b). Unfortunately, we also know nothing on the distribution of V and admixed As between structure positions in bakakinite. If vanadium and arsenic occupy the sites with different coordination, then the increase of As5+ content could result in the formation of the hypothetical V–As-ordered bakakinite-type compound with the ideal formula Ca2VAsO7.
In the powder XRD data, bakakinite and synthetic Ca2V2O7, being very close to one another (Table 3), differ strongly from Ca2As2O7. In particular, the characteristic, low-angle region (d > 2.5 Å) of the powder XRD pattern of Ca2As2O7 contains strong lines with d = 3.37, 3.34 and 2.77 Å and intensities I = 53, 100 (the strongest reflection of Ca2As2O7) and 35%, respectively (Pertlik, Reference Pertlik1980), which are absent in the powder XRD diagrams of bakakinite and synthetic Ca2V2O7 (Table 3).
Of note, the crystal structure, unit-cell metrics and powder XRD pattern of another natural divanadate with large cations, chervetite Pb2V2O7 (Shannon and Calvo, Reference Shannon and Calvo1973) are quite different from those of both Ca2V2O7 and Ca2As2O7. All known polymorphs of Cu2V2O7 including the minerals blossite and ziesite (Krivovichev et al., Reference Krivovichev, Filatov, Cherepansky, Armbuster and Pankratova2005), also possess quite different structures.
Acknowledgements
We thank Evgeny Galuskin and two anonymous referees for their valuable comments. The mineralogical and spectroscopic studies of bakakinite and crystal chemical analysis by IVP, NVZ and MFV were supported by the Russian Science Foundation, grant no. 19-17-00050. The technical support by the SPbSU X-Ray Diffraction Resource Center in the powder XRD study is acknowledged.
Competing interests
The authors declare none.