Introduction
Histology is considered the assessment of the microscopic structures of animal and plant cells and characteristic tissues through staining, sectioning, and examining them under a microscope (electron or light microscope). Various methods are applied, with histological studies used extensively, not only in medicine for the study of diseased tissues to aid treatment but also in forensic investigations and/or autopsy (Alturkistani et al., Reference Alturkistani, Tashkandi and Mohammedsaleh2015).
In humans, cardiac and vascular gross and microscopic anatomy has been appreciated for many years, which was followed by ultrastructural anatomy and physiology. However, pathogenesis was only clarified long after the disease was noted, and this realization is still humbling. For this reason, learning about both normal and abnormal heart function is an ongoing task (Buja & Butany, Reference Buja and Butany2015).
Faced with this growing knowledge, the pathologist has become the data integrator, interpreting the clinical information, imaging, and finally, the gross and histopathological findings to make a clinically relevant diagnosis. Therefore, the pathologist must understand the normal cardiac anatomy before appreciating pathology since the consideration of gross and microscopic anatomy allows the pathologist to understand the range of normal findings (Buja & Butany, Reference Buja and Butany2015).
Cetacean hearts largely follow the gross general appearance found in other mammalian species, but they have some noteworthy differences. For example, the heart of dolphins is larger and flatter than that of most terrestrial mammals due to the shape of the thorax and the pressure it withstands. Therefore, it has an oval, almost circular outline, with wider (but shorter) ventricles, a slightly rotated axis, and auricles that are flat, with their ventral edges surpassing the coronary sulcus separating atria from ventricles. Histologically, the cardiac muscle in cetaceans is no different from that of terrestrial mammals. Cardiac muscle fibers (cardiomyocytes) are short, thinner than skeletal striated and uninucleated fibers, with the nucleus in the center of the cells. Three typical regions are distinguished: the epicardium, myocardium, and endocardium (Fernández et al., Reference Fernández, Caballero and Jaber2005a; Eurell & Frappier, Reference Eurell and Frappier2013; Cozzi et al., Reference Cozzi, Huggenberger and Oelschläger2017).
Considerable knowledge has been gained over the last few decades on several disease processes afflicting free-ranging cetaceans from a pathologist's point of view (Arbelo et al., Reference Arbelo, Monteros, Herráez, Andrada, Sierra, Rodríguez, Jepson and Fernández2013; Díaz-Delgado et al., Reference Díaz-Delgado, Fernández, Sierra, Sacchini, Andrada, Vela, Quesada-Canales, Paz, Zucca, Groch and Arbelo2018; Groch et al., Reference Groch, Díaz-Delgado, Marcondes, Colosio, Santos-Neto, Carvalho, Boos, Meirelles, Ramos, Guimarães, Borges, Vergara-Parente, St Leger, Fernández and Catão-Dias2018). For example, several studies have proven that deep-diving cetaceans, such as beaked whales, are threatened by anthropogenic activities. These include bycatch, debris ingestion, ship collisions, and any activity producing high-intensity noise (e.g., military sonars), which has even been the cause of several unusual stranding events (Fernández et al., Reference Fernández, Edwards, Rodríguez, Monteros, Herráez, Castro, Jaber, Martín and Arbelo2005b; Carrillo & Ritter, Reference Carrillo and Ritter2010; Virgili et al., Reference Virgili, Authier, Boisseau, Cañadas, Claridge, Cole, Corkeron, Dorémus, David, Di-Méglio, Dunn, Dunn, García-Barón, Laran, Lauriano, Lewis, Louzao, Mannocci, Martínez-Cedeira, Palka, Panigada, Pettex, Roberts, Ruiz, Saavedra, Santos, Van Canneyt, Bonales, Monestiez and Ridoux2019; Puig-Lozano et al., Reference Puig-Lozano, Fernández, Sierra, Saavedra, Suárez-Santana, Fuente, Díaz-Delgado, Godinho, García-Álvarez, Zucca, Xuriach, Arregui, Felipe-Jiménez, Consoli, Díaz-Santana, Segura-Göthlin, Câmara, Rivero, Sacchini, Bernaldo de Quirós and Arbelo2020). However, there is still a dearth of knowledge on cardiovascular lesions in these species, but, for the most part, the cardiac responses to different types of injury appear to follow similar patterns to those observed in humans and other animal species.
For this reason, this study aimed to partially fill in a gap of knowledge regarding histological findings in the cardiac muscle of free-ranging stranded cetaceans and, more specifically, of deep-diving Cuvier's beaked whales (Ziphius cavirostris).
Materials and Methods
One thousand, two hundred and ninety-nine cetaceans of 26 different species, including small and large odontocetes and mysticetes, were stranded on the coast of the Canary Islands from 1992 to 2020. Of these, 106 animals (8.16%) were initially included in this study and identified as being part of the Family Ziphiidae and were then categorized into the following species: Sowerby's beaked whales (Mesoplodon bidens) (3/106–2.83%), Blainville's beaked whales (Mesoplodon densirostris) (11/106–10.38%), Gervais' beaked whales (Mesoplodon europaeus) (16/106–15.09%), 1 True's beaked whale (Mesoplodon mirus) (1/106–0.94%), and Cuvier's beaked whale (Ziphius cavirostris) (73/106–68.87%). Since Cuvier's beaked whale was the species with the most individuals, we decided to focus our study exclusively on this species. A total of 49 out of the 73 animals (67.12%) were necropsied following the basic protocol for the performance of a cetacean necropsy outlined and published by the European Cetacean Society by Thijs Kuiken and Manuel García Hartmann (Proceedings of the first ECS workshop on Cetacean Pathology: Dissection Techniques and Tissue Sampling. Leiden, Netherlands, 13–14 September 1991. Newsletter # 17 special issue) (Kuiken & Hartmann, Reference Kuiken and Hartmann1991). This protocol has undergone some modifications of the procedures referenced in the manual published by Geraci and Lounsbury in 2005 (Marine mammals ashore: a field guide for strandings), along with a more recent update and innovation by the European Cetacean Society by ASCOBANS and ACCOBAMS in 2019 (Proceedings of the ECS workshop on Cetacean Pathology: Necropsy Technique and Tissue Sampling. Funchal, Madeira, Portugal, April 2016). Tissue samples of approximately 2–3 cm3 were collected during the necropsy and stored in 10% buffered formaldehyde for further histopathological analysis (Kuiken & Hartmann, Reference Kuiken and Hartmann1991; Geraci & Lounsbury, Reference Geraci and Lounsbury2005; ASCOBANS/ACCOBAMS, 2019). After being fixed for at least 24 h, the samples were cut (approximately 1.5 cm × 0.3 cm) and placed in cassettes for automatic processing and later inclusion in paraffin.
Thus, out of the initial 49 necropsied individuals we selected for our research, 15 animals (30.61%) presented very fresh, fresh, moderate, and advanced autolysis conservation states, which were also determined following the parameters and classifications established by this protocol (Kuiken & Hartmann, Reference Kuiken and Hartmann1991; Geraci & Lounsbury, Reference Geraci and Lounsbury2005; ASCOBANS/ACCOBAMS, 2019). Additionally, the pathological findings in the different organs and the causes of death were described in PhD theses, published as scientific articles, and presented in annual reports deliverable to the Government of the Canary Islands annually (Arbelo et al., Reference Arbelo, Monteros, Herráez, Andrada, Sierra, Rodríguez, Jepson and Fernández2013; Díaz-Delgado et al., Reference Díaz-Delgado, Fernández, Sierra, Sacchini, Andrada, Vela, Quesada-Canales, Paz, Zucca, Groch and Arbelo2018).
All the individuals (15–100%) were deceased when the stranding notification was received. Nevertheless, a total of six animals (40.00%) showed a clear evidence of being alive when stranded, eight animals (53.33%) had no signs of live stranding, and the status of one animal (6.67%) was unclear.
Concerning the causes of death, the individuals died due to natural pathology associated with a good/moderate body condition (4/15–26.67%), natural pathology associated with a significant loss of body condition (3/15–20.00%), foreign-body ingestion pathology (3/15–20.00%), perinatal/neonatal pathology (2/15–13.33%), and ship strike (1/15–6.67%). It was impossible to determine the cause of death in two animals (13.33%).
The Veterinary Histology and Pathology Department of the Institute of Animal Health and Food Safety (IUSA) of the Veterinary School of the University of Las Palmas of Gran Canaria (ULPGC) is known at an international level. For this reason, on some occasions, the department acts as a consultant in specific cases. Consequently, different tissue samples of stranded cetaceans in different countries are also stored in the tissue bank of the department. As a result, we included a total of five Cuvier's beaked whales, that is, 4 (80.00%) stranded in Almería, Spain, in 2006 and 2011 and 1 (20.00%) stranded in Corfu, Greece, in 2011, which were part of atypical mass stranding (AMS) episodes and died due to gas embolism.
In conclusion, a total of 20 individuals were analyzed in this study.
All the cardiac tissue samples included in this study consisted mainly of left and/or right ventricles that were fixed and processed following standard histological procedures. Specifically, 4 μm thick sample sections were used for hematoxylin and eosin (HE) and periodic acid-Schiff staining, and 5 μm sections were used for phosphotungstic acid hematoxylin (PTAH) and Masson's trichrome techniques (Ovalle & Nahirney, Reference Ovalle and Nahirney2008). The microscopic findings, identified during the cardiac examination under an optical microscope, were reviewed blindly by four veterinarians and/or pathologists (NC, ES, PH, and AF). The extent of the cardiac lesions was judged subjectively as follows: absent, mild, mild to moderate, moderate, moderate to severe, and severe.
The procedure described previously is summarized in Figure 1. Moreover, the basic epidemiological data and causes of death of the cetaceans included in this research are presented in Supplementary Table S1.

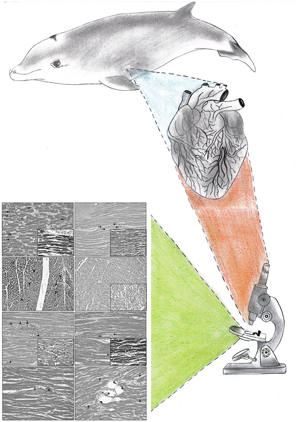
Fig. 1. This graphical abstract provides the readers with a visual representation of the conclusions and an efficient way to appreciate the work's critical findings and central message. This illustration describes the consecutive steps of the research, starting with the necropsy of stranded Cuvier's beaked whales (Ziphius cavirostris), in which a macroscopic assessment and sample collection of the cardiac muscle was performed. Then, the samples were processed employing a standard protocol for the blind evaluation of histological findings by veterinarians and/or pathologists using an optical microscope. Finally, the microscopical images resulting from the histological evaluation are used in the forthcoming figures of this article and the feature sizes may be seen from the micron markers in those later figures.
Results
The significant lesions described histologically in the cardiac muscle of the animals analyzed were (1) vascular changes (Supplementary Table S2), such as congestion, edema, hemorrhages, leukocytosis, and intravascular coagulation; (2) acute degenerative changes (Supplementary Table S3), which consisted of contraction band necrosis, wavy fibers, cytoplasmic hypereosinophilia, and perinuclear vacuolization; (3) infiltration of inflammatory cells (Supplementary Table S4); and (4) the presence and/or deposition of different substances (Supplementary Table S5), such as interstitial myoglobin globules, lipofuscin pigment, polysaccharide complexes, and intra- and/or extravascular gas emboli.
Regarding vascular changes, vascular congestion was observed in 13 out of the 20 animals (65.00%), and this phenomenon was identified by swelling and light occlusion of capillaries with blood. Eleven out of the 20 animals (55.00%) had interstitial edema, represented histologically by fibers separated by spaces. Hemorrhages, which consist of blood escaping from ruptured blood vessels, were identified in 5 out of the 20 animals (25.00%). These could be visualized in the endocardial, myocardial, and epicardial portions of the heart. Twelve out of the 20 animals (60.00%) presented leukocytosis and intravascular coagulation, which is a condition in which blood clots form and block small blood vessels, together with the presence of inflammatory cells, more specifically, leukocytes.
Regarding acute degenerative changes, contraction band necrosis was observed in 8 out of the 20 animals (40.00%), consisting of dense hypereosinophilic bands that ran transversely through the myocytes (Fig. 2). Fifteen out of the 20 animals (75.00%) exhibited long and thin undulated fibers, termed wavy fibers (Fig. 3). Hypereosinophilia was easily identified since the necrotic fibers presented more eosinophilic, blue, or red staining (using HE, PTAH, and Masson's trichrome techniques, respectively) than did adjacent normal fibers (Fig. 4). The above finding was detected in 15 out of the 20 animals (75.00%), being more pronounced in some cases at the subendocardial and subepicardial levels. Seventeen out of the 20 animals (85.00%) exhibited vacuolar degeneration (Fig. 5), morphologically characterized by the intracellular accumulation of fluid and the lysis of myofibrils. The liquid increase exerted pressure on the different parts of the cell, resulting in most of the fibers with vacuoles having a pyknotic nucleus, and in some cases, this lesion was more noticeable at both the subendocardial and the subepicardial levels.

Fig. 2. (a) Animal 16 (ref: CET 771 – i414/15). Contraction band necrosis running (arrows) transversely through the cardiomyocytes and identified through the higher intensity of color with different histochemical techniques (in this case, with the HE technique). (b) Animal 10 (ref: CET 471 – i319/08). Details of the contraction band necrosis (arrows) analyzed with the PTAH technique.

Fig. 3. (a) Animal 17 (ref: CET 855 – i197/17). Wavy fibers (arrows) consisting of long and thinned undulated fibers. HE technique. (b) Animal 17 (ref: CET 855 – i197/17). Details of the wavy fibers (arrows) analyzed with the PTAH technique.

Fig. 4. Animal 15 (ref: CET 720 – i185/14). Hypereosinophilia consisting of increased staining of necrotic cardiac cells (arrowheads) with different techniques (in this case, with the PTAH technique). The presence of cytoplasmic vacuolization was observed in some injured cardiomyocytes (thin arrows).

Fig. 5. (a) Animal 3 (ref: CET 236 – i34/04). Intracytoplasmic vacuoles (arrows) of cardiomyocytes identified with the HE technique. (b) Animal 1 (ref: CET 103-258/00). Vacuolar degeneration (arrows) in cardiac cells. HE.
In 9 out of the 20 animals (45.00%), different types of cells (Fig. 6) were identified as infiltrating zones where hemorrhages, fibrillary rupture of fibers, degeneration, and necrosis of cardiac cells were present. Of these, 3 out of the 20 animals (15.00%) displayed infiltration of mononuclear cells, another 4 out of the 20 animals (20.00%) presented infiltration by polymorphonuclear cells, and, finally, 5 out of the 20 animals (25.00%) exhibited infiltration of macrophages. It is also important to mention that 3 out of the 20 animals (15.00%) demonstrated infiltration with polymorphonuclear cells and macrophages.

Fig. 6. (a) Animal 11 (ref: i76/11). The presence of inflammatory cells (arrows) in the interstitial space, with congestion (*). HE. (b) Animal 2 (ref: CET 113-384/00). Details of the infiltration of mononuclear cells (arrows) in a zone with ruptured cardiac fibers. Additionally, there is congestion (*) of the blood vessels. HE.
Additionally, we verified the deposition of diverse substances, such as myoglobin, released by the ruptured cardiac fibers and accumulated as globules inside the blood vessels and in the interstitial spaces (Fig. 7). Myoglobin globules were observed in 14 out of the 20 animals (70.00%). Lipofuscin is one of the other substances that was detected in 5 out of the 20 animals (25.00%). It is a yellow–brown pigment resulting from the breakdown and absorption of damaged blood cells. In 4 out of the 20 animals (20.00%), polysaccharide complexes (Fig. 8) were present in the interior of the cardiomyocytes, defined as basophilic material inclusions. Vessel dilation and intra- and/or extravascular gas bubbles (Fig. 9), consisting of round empty spaces among the blood cells inside a vessel and/or the tissue cells inside the interstitium, were found in 7 out of the 20 animals (35.00%).

Fig. 7. (a) Animal 6 (ref: i17/06). Myoglobin globules (arrows) observed in the interstitial space. HE. (b) Animal 13 (ref: i384/11). Details of the myoglobin globules (arrows) stained with the PTAH technique.

Fig. 8. Animal 6 (ref: i17/06). Basophilic material described as polysaccharide complexes (arrow) in the interior of the cardiomyocytes. Congestion (*) of the blood vessels is present. HE.

Fig. 9. Animal 6 (ref: i17/06). Round empty spaces compatible with extravascular gas bubbles (arrowheads) surrounding a blood vessel (*). Detection of myoglobin globules (arrows) in the interstitial space. HE.
Discussion
Before entering a discussion of the results, the authors would like to emphasize that this research was conducted in stranded free-ranging Cuvier's beaked whales. Since we are working with wild animals, stranding networks, and the general public, in a stranding event, it is often clinically and pathologically challenging to obtain the correct information and/or the complete history of each animal prior to its death (Arbelo et al., Reference Arbelo, Monteros, Herráez, Andrada, Sierra, Rodríguez, Jepson and Fernández2013; Díaz-Delgado et al., Reference Díaz-Delgado, Fernández, Sierra, Sacchini, Andrada, Vela, Quesada-Canales, Paz, Zucca, Groch and Arbelo2018). For this reason, the diagnosis of the pathological entity and/or cause of death, which may be considered a complete puzzle, is based on the sum of the gross and/or histopathological findings or “puzzle pieces” identified during the pathological evaluation of the different organs. Nevertheless, it is essential to highlight that the observation of specific lesions may or may not be, direct or indirectly, associated with the cause of death. Therefore, the pathological entities and/or causes of death should be determined by an experienced veterinarian and/or pathologist (Arbelo et al., Reference Arbelo, Monteros, Herráez, Andrada, Sierra, Rodríguez, Jepson and Fernández2013; Díaz-Delgado et al., Reference Díaz-Delgado, Fernández, Sierra, Sacchini, Andrada, Vela, Quesada-Canales, Paz, Zucca, Groch and Arbelo2018).
In this investigation, the authors centered their attention exclusively on the histopathological findings of the hearts of stranded Ziphius cavirostris, which, in some cases, may be directly related to the cause of death.
Recent studies have characterized cardiac lesions associated with stressful situations, such as live stranding, ship collisions, and fishery interactions, as being comparable to the Stress Cardiomyopathy (SCMP) described in humans. These morphological alterations consist of vascular changes (i.e., congestion, interstitial edema, and hemorrhages) and acute degenerative lesions (i.e., contraction band necrosis, wavy fibers, hypereosinophilia, and cytoplasm vacuolization) with a perivascular distribution, indicative of ischemia–reperfusion damage. Additionally, infiltration of inflammatory cells, myoglobin globules and fibrotic tissue are also detected in this pathology (Herráez et al., Reference Herráez, Sierra, Arbelo, Jaber, Monteros and Fernández2007, Reference Herráez, Espinosa de los Monteros, Fernández, Edwards, Sacchini and Sierra2013; Câmara et al., Reference Câmara, Sierra, Fernández-Maldonado, Monteros, Arbelo, Fernández and Herráez2019a, Reference Câmara, Sierra, Fernández, Suárez-Santana, Puig-Lozano, Arbelo and Herráez2019b, Reference Câmara, Sierra, Fernández, Arbelo, Andrada, Monteros and Herráez2020a, Reference Câmara, Sierra, Fernández, Arbelo, Bernaldo de Quirós, Arregui, Consoli and Herráez2020b; Puig-Lozano et al., Reference Puig-Lozano, Fernández, Sierra, Saavedra, Suárez-Santana, Fuente, Díaz-Delgado, Godinho, García-Álvarez, Zucca, Xuriach, Arregui, Felipe-Jiménez, Consoli, Díaz-Santana, Segura-Göthlin, Câmara, Rivero, Sacchini, Bernaldo de Quirós and Arbelo2020).
There is a chronological sequence in the changes of acute ischemic injuries, such as those described in SCMP syndrome, that were identified in this study; these changes can be observed as early as the first 5 min, starting with the presence of long, thinned, wavy fibers (15/20–75.00%) separated by spaces representing edema (11/20–55.00%) and microvascular congestion (13/20–65.00%) at the borders of the ischemic myocardium. In the following 15 min, cell death may start to occur. Within 2 to 3 h, early changes in cardiomyocyte coagulation necrosis with nuclear pyknosis, color change, more specifically “red brick change” or cytoplasm hypereosinophilia (15/20–75.00%), focal contraction bands (8/20–40.00%) and subtle interstitial edema become evident. After 3 to 6 h, hypereosinophilia and edema become more pronounced and easily recognizable. Six to 12 h later, an increased number of neutrophils line up in capillaries, as well as accelerated changes and more extensive contraction band necrosis with reperfusion, become noticeable. In the next 12-h period and completing the 24-h cycle after the ischemic injury, extravazation into the interstitial space of neutrophils (4/20–20.00%) occurs, along with obvious vascular congestion, interstitial edema, and focal areas of hemorrhage (5/20–25.00%). Subsequently, the subacute period starts.
A heavy influx of neutrophils is detected in the periphery, and lymphocytes (3/20–15.00%) start to appear at the edge of the infarction at 1 to 3 days. Degeneration of neutrophils becomes clear from 3 to 7 days, with basophilic granular debris produced as “nuclear dust”. The beginning of the disintegration of necrotic myocytes with phagocytosis and removal of necrotic fibers, along with the evident presence of lymphocytes and the influx of macrophages (5/20–25.00%), is also observed.
Moreover, the onset of vascular and connective (granulation) tissue proliferation and early fibrosis has begun at the edge of the injury. Afterward, the chronic phase starts. After 2 weeks, chronic inflammation and the removal of peripheral muscle fibers occur, with increased numbers of mononuclear cells (macrophages) at the margins of the injury, where vascular and connective tissue proliferation has become more prominent. Eosinophils, lymphocytes, and plasma cells are also present, with infrequent neutrophils. Three to 4 weeks after ischemic injury, the continued removal of necrotic fibers and proliferation of granulation tissue extends throughout the injured area. Macrophages are still numerous, and plasma cells, eosinophils and lymphocytes are still present, but there is more substantial fibroblastic proliferation with collagen formation that is recognizable as a pink, fibrillary extracellular material. From 5 to 6 weeks, the removal of residual necrotic fibers and continued gradual scar formation with collagen deposition are identifiable. Inflammatory cells and vascularity decreases, but a few macrophages, plasma cells, and lymphocytes persist and remain in the scar that eventually forms. Finally, after 3 months, the fibrous scar is fully developed (Buja, Reference Buja2005; Buja & Butany, Reference Buja and Butany2015; Câmara et al., Reference Câmara, Sierra, Fernández, Arbelo, Andrada, Monteros and Herráez2020a).
As mentioned before, these alterations have been associated with the cardiac pathology designated SCMP in cetaceans. Nevertheless, it is essential to highlight that these changes can sometimes imply a bias in the cases analyzed. For instance, some live-stranded animals tend to strand themselves due to previous pathological processes, such as infectious diseases, which are not always identifiable (Câmara et al., Reference Câmara, Sierra, Fernández, Arbelo, Andrada, Monteros and Herráez2020a). Therefore, an example of this situation would be the animals that presented leukocytosis and intravascular coagulation (12/20–60.00%), which can be evaluated either as a case of disseminated intravascular coagulation (DIC) or as simply a phase of the acute ischemic injury described previously.
DIC is characterized by disseminated and often uncontrolled coagulation activation associated with a high risk of macro- and microvascular thrombosis and progressive consumption coagulopathy, which leads to an increased bleeding risk. Sepsis, cancer, trauma, and obstetric emergencies are ranked among the most frequent pathological conditions that may trigger DIC (Levi et al., Reference Levi, Cate, van der Poll and van Deventer1993; Okajima, Reference Okajima1999; Okabayashi et al., Reference Okabayashi, Wada, Ohta, Shiku, Nobori and Maruyama2004; Semeraro et al., Reference Semeraro, Ammollo, Semeraro and Colucci2010; Papageorgiou et al., Reference Papageorgiou, Jourdi, Adjambri, Walborn, Patel, Fareed, Elalamy, Hoppensteadt and Gerotziafas2018). The generalized Shwartzman reaction was the first sign demonstrating the relationship between leukocytes and coagulation abnormalities. Leukocytes have also been shown to have a crucial role in intravascular coagulation in patients with sepsis. Cytokines activate neutrophils, thus releasing various inflammatory mediators capable of damaging endothelial cells. This damage and thrombus formation may play a role in the development of organ failure in DIC associated with sepsis (Okajima, Reference Okajima1999).
Lipofuscin (5/20–25.00%) results from the accumulation of the end-product of intracellular lipid peroxidation or cellular metabolism during ageing (life stage) (Nakano et al., Reference Nakano, Oenzil, Mizuno and Gotoh1995). This pigment is widely observed in postmitotic cells, especially in long-lived cells such as neurons and cardiomyocytes. Therefore, lipofuscin is a pigment that accumulates during ageing (Kakimoto et al., Reference Kakimoto, Okada, Kawabe, Sasaki, Tsukamoto, Nagao and Osawa2019). To date, the pathogenic roles of lipofuscin have been strongly suggested in various diseases, such as the end stage of heart failure with dilated cardiomyopathy and ischemic cardiomyopathy (Rayment et al., Reference Rayment, Haven, Madden, Murday, Trickey, Shipley, Davies and Katz1999; Radu et al., Reference Radu, Bold, Pop, Mălăescu, Gheorghişor and Mogoantă2012; Nozynski et al., Reference Nozynski, Zakliczynski, Konecka-Mrowka, Zakliczynska, Pijet, Zembala-Nozynska, Lange and Zembala2013; Kakimoto et al., Reference Kakimoto, Okada, Kawabe, Sasaki, Tsukamoto, Nagao and Osawa2019).
Complex polysaccharide inclusions (4/20–20.00%) within skeletal muscle fibers occur in various human and animal disorders. In humans, such inclusions have been classified as incidental, age-related changes, such as in corpora amylacea in neural tissue or basophilic degeneration of the myocardium, and as pathological accumulations as a critical feature of the wide-ranging polyglucosan body disease (Cavanagh, Reference Cavanagh1999; Longué et al., Reference Longué, Dagleish, McGovern, Brownlow and Baily2020). Occasionally, these inclusions occur in skeletal or cardiac muscle in conditions such as Lafora body disease or as a primary lesion at sites of myopathies, including glycogen storage disorders (Cavanagh, Reference Cavanagh1999; Sierra et al., Reference Sierra, Fernández, Monteros, Jaber, Andrada and Herráez2012; Hedberg-Oldfors & Oldfors, Reference Hedberg-Oldfors and Oldfors2015; Longué et al., Reference Longué, Dagleish, McGovern, Brownlow and Baily2020). Similar inclusions have also been reported in animals and are most thoroughly studied in equine polysaccharide storage myopathy (Valentine et al., Reference Valentine, Credille, Lavoie, Fatone, Guard, Cummings and Cooper1997; Sierra et al., Reference Sierra, Fernández, Monteros, Jaber, Andrada and Herráez2012; Longué et al., Reference Longué, Dagleish, McGovern, Brownlow and Baily2020). In cetaceans, these intramyofiber Pas-positive, diastase-resistant aggregates and granules have already been described as present within the skeletal muscles of several cetacean species and within the myocardium of pilot whales. Nevertheless, the pathogenesis and clinical significance remain undetermined (Scotti, Reference Scotti1962; Cowan, Reference Cowan1966; Sierra et al., Reference Sierra, Fernández, Monteros, Jaber, Andrada and Herráez2012; Longué et al., Reference Longué, Dagleish, McGovern, Brownlow and Baily2020).
The formation of intravascular and extravascular gas bubbles (7/20–35.00%) are the main findings of forensic investigations related to a widely known clinical syndrome in human medicine, mainly occurring in divers, called Decompression Sickness (Vann et al., Reference Vann, Butler, Mitchell and Moon2011; Velázquez-Wallraf et al., Reference Velázquez-Wallraf, Fernández, Caballero, Møllerløkken, Jepson, Andrada and Quirós2021). Gas embolism and decompression-like sickness have also been described in wild animals, first in cetaceans, more specifically with mass strandings of beaked whales, which occurred in 2003 and were coincidental in time and space with naval maneuvres (Jepson et al., Reference Jepson, Arbelo, Deaville, Patterson, Castro, Baker, Degollada, Ross, Herráez, Pocknell, Rodríguez, Howie, Espinosa, Reid, Jaber, Martin, Cunningham and Fernández2003). Since then, many other cases have been diagnosed in cetaceans and other species, such as turtles (Fernández et al., Reference Fernández, Edwards, Rodríguez, Monteros, Herráez, Castro, Jaber, Martín and Arbelo2005b, Reference Fernández, Sierra, Díaz-Delgado, Sacchini, Sánchez-Paz, Suárez-Santana, Arregui, Arbelo and Quirós2017; García-Párraga et al., Reference García-Párraga, Crespo-Picazo, de Quirós, Cervera, Martí-Bonmati, Díaz-Delgado, Arbelo, Moore, Jepson and Fernández2014). More recently, the decompressive pathology in cetaceans has been studied through an experimental pathological model, where the presence of not only gas bubbles but also other gross and histological lesions, such as vascular changes (e.g., interstitial edema, hemorrhages) and/or acute degenerative changes (e.g., contraction band necrosis, wavy fibers, hypereosinophilia, intracytoplasmic vacuolization), was described (Velázquez-Wallraf et al., Reference Velázquez-Wallraf, Fernández, Caballero, Møllerløkken, Jepson, Andrada and Quirós2021). Although these changes have been reported in previous investigations, it is essential to remark that in our study, there was no apparent difference and/or relationship between the extension and severity of the acute degenerative lesions and the atypical mass stranding involving beaked whales with gas embolism when compared with the corresponding findings related to other causes of death, such as live stranding or ship collision, with a stress component associated in both deep- and shallow-diving cetaceans.
Summary or Conclusion
Herein, we present the prevalence and types of cardiac histopathological findings in stranded free-ranging Cuvier's beaked whales. Although some of these observed features were considered incidental or of little clinical and/or pathological relevance, others could have resulted in severe cardiovascular dysfunction, such as those injuries compatible with the damage occurring with the SCMP described in humans and cetaceans. In conclusion, these results advance our understanding of cetacean pathology, may prove to be of value to diagnosticians, and may guide future comparative pathologic studies, which can improve our medical and conservation efforts by reducing the mortality of these animals.
Availability of data and material
All data reported in this work are classified and stored in the tissue bank of the Veterinary Histology and Pathology Department of the Institute of Animal Health and Food Safety (IUSA) of the Veterinary School of the University of Las Palmas of Gran Canaria (ULPGC) (Campus Universitario Cardones de Arucas, Trasmontaña s/n, 35413 Arucas, Las Palmas of Gran Canaria, Spain).
Supplementary material
To view supplementary material for this article, please visit https://doi.org/10.1017/S1431927622000605.
Acknowledgments
We appreciate all the people who indirectly participated in the elaboration of this work; therefore, we extend very special thanks mainly to our laboratory staff and to all associated nongovernmental organizations (Marisa Tejedor, SECAC and Canarias Conservación) and to the members and volunteers of the Canary Islands Cetacean Stranding Network who participated in the necropsies. We also want to express our gratitude to the scientific personnel of the Stranding Network in Almeria (Spain) and Corfu (Greece) for the international collaboration between our institutions.
Author contributions statement
Nakita Câmara: This author wrote the article, performed the necropsy of the animals, and contributed to the gross, histological and histochemical description and diagnosis of the cases. Antonio Fernández: This author performed the necropsy of the animals, contributed to the histological and histochemical descriptions and diagnosis of the cases, and guided the first author during the drafting and publication process. Pedro Herráez: This author contributed to the histological and histochemical description and diagnosis of the cases. Manuel Arbelo: This author performed the necropsy of the animals and contributed to the histological and histochemical descriptions and diagnosis of the cases. Marisa Andrada: This author contributed to the histological and histochemical description and diagnosis of the cases. Cristian M. Suárez-Santana: This author performed the necropsy of the animals and contributed to the histological and histochemical descriptions and diagnosis of the cases. Eva Sierra: This author performed the necropsy of the animals and contributed to the histological and histochemical descriptions and diagnosis of the cases. All authors read and approved the final manuscript.
Financial support
This study is part of a postdoctoral research project, which was conducted at the Universidad de Las Palmas de Gran Canaria (ULPGC), supported by the Ministerio de Economía y Competitividad (MINECO) through a year of postdoctoral orientation offered by a predoctoral grant for training of research personnel (Contrato Predoctoral para Formación de Personal Investigador, año 2016) with reference number BES-2016-076907. Furthermore, part of this research was supported through a national project titled Patología Embólica (Gaseosa/Grasa) en Cetáceos supported by the Ministerio de Ciencia, Innovación y Universidades with reference number PGC2018-101226-B-I00. Moreover, the Canary Islands Government has funded and provided support to the Cetacean Stranding Network, which is part of the Canary Island Surveillance Network (Red Vigía Canarias).
Conflict of interest
The authors declare none.
Ethical standards
Regarding the management of stranded cetaceans, required permission was issued by the environmental department of the Canary Islands Government and the Spanish Ministry of Environment.