Axenfeld–Rieger syndrome is a rare autosomal dominant disorder that leads to abnormal development of the anterior segment of the eye. Reference Seifi and Walter1,Reference French2 A number of extraocular manifestations have been described including craniofacial and dental anomalies, umbilical abnormalities, pituitary abnormalities, sensorineural hearing loss, and CHDs. Reference Balikov, Jacobson and Prasov3,Reference Zhang, Liang and Yue4 Approximately half of all cases are due to mutations in Forkhead Box C1 or paired-like homeodomain transcription factor 2. Reference French2,Reference Balikov, Jacobson and Prasov3 Both Forkhead Box C1 and paired-like homeodomain transcription factor 2 encode transcription factors that play key roles during embryonic development. Reference Balikov, Jacobson and Prasov3,Reference Du, Huang, Fan, Li, Xia and Xiang5,Reference Ahmed, Sethna, Krueger, Yang and Hufnagel6 Insight into the mechanisms by which mutations in Forkhead Box C1 and paired-like homeodomain transcription factor 2 lead to Axenfeld–Rieger syndrome has been largely derived from mouse and zebrafish models, highlighting abnormalities of neural crest cell migration as well as variation in the expression of genes critical to embryonic development. Reference French2,Reference Zhang, Liang and Yue4,Reference Campione, Steinbeisser and Schweickert7–Reference Lambers, Arnone, Fatima, Qin, Wasserstrom and Kume12 Both genes have been associated with determination of the left-right axis, with clear potential implications for cardiovascular development. Reference Campione, Steinbeisser and Schweickert7,Reference Chrystal, French and Jean8,Reference Ai, Liu and Ma13–Reference Yu, St Amand and Wang16 Importantly, transcriptional activity of Forkhead Box C1 is negatively regulated by paired-like homeodomain transcription factor 2, helping to explain the overlapping phenotypes. Reference Tumer and Bach-Holm17,Reference Berry, Lines and Oas18
Axenfeld–Rieger syndrome has been associated with a variety of CHDs. The most frequently described cardiac lesions associated with Axenfeld–Rieger syndrome include valvular heart disease (including both atrioventricular and semilunar valve abnormalities), atrial septal defects, patent arterial duct, outflow tract abnormalities, tetralogy of Fallot, and ventricular hypoplasia. Reference Du, Huang, Fan, Li, Xia and Xiang5,Reference Ahmed, Sethna, Krueger, Yang and Hufnagel6,Reference Zhao, Peng and Li19–Reference Bekir and Gungor23 To date, there has not been a comprehensive report of the incidence and heterogeneity of CHD associated with Axenfeld–Rieger syndrome. This project aims to report the incidence and spectrum of congenital heart lesions in patients with Axenfeld–Rieger syndrome using the electronic medical record of a large academic medical centre.
Patients with a diagnosis of Axenfeld–Rieger syndrome were identified from our institutional electronic medical record (Epic Systems, Verona, WI) using keyword searches for Axenfeld–Rieger syndrome, Rieger syndrome, and Axenfeld anomaly over the period from November 2017 to May 2021. Dates were chosen based on the timing that Epic was implemented at our institution, allowing such a query. Medical records were reviewed to confirm the diagnosis of Axenfeld–Rieger syndrome and for the presence and type of CHD. This study was approved as an exempt study by the Vanderbilt University Medical Center Institutional Review Board.
A total of 60 patients were identified from our electronic medical record search, of which 2 (3.3%) were excluded, as the diagnosis of Axenfeld–Rieger syndrome could not be confirmed. Patient demographics are shown in Table 1. A total of 14 patients (24.1%) had documented CHD. Of the patients with CHD, no familial relationship existed between patients. Specific cardiac lesions are shown in Table 2. Of note, some patients had more than one cardiac defect. Importantly, a number of these cardiac lesions were haemodynamically significant, with some requiring intervention. Of the patients with cardiac defects, six patients had genetic defects that were reported in the electronic medical record, including trisomy 2, partial chromosome 9 deletion, and chromosome 6 ring, each in one patient, and Forkhead Box C1 mutation or variants in three patients. Of note, not all patients underwent a formal genetic evaluation.
Table 1. Demographics and clinical characteristics (N = 58)

Table 2. Cardiac diagnoses among patients with Axenfeld–Rieger syndrome
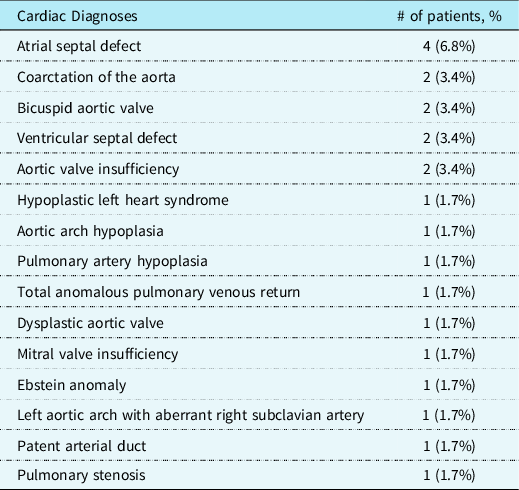
*Some patients had multiple diagnoses.
Axenfeld–Rieger syndrome is a rare disorder characterised most commonly by abnormalities of the anterior segment of the eye. While prior studies have noted an association between Axenfeld–Rieger syndrome and CHDs anomalies, our analysis provides the first estimate of prevalence in this population. Our case series is the first of its kind to characterise the heterogeneity of cardiac lesions associated with Axenfeld–Rieger syndrome at a single large academic centre, including a case of hypoplastic left heart syndrome, not previously reported in the literature. Notably, many left-sided obstructive lesions were associated with Axenfeld–Rieger syndrome in our case series, which indicates that this type of defect may be more prevalent in patients with Axenfeld–Rieger syndrome than previously recognised. In line with previous studies, our patients had genetic defects contributing to their presentation, although some had other genetic abnormalities identified, such as chromosomal abnormalities that did not involve the genes commonly reported in the literature (Forkhead Box C1, paired-like homeodomain transcription factor 2).
In summary, patients with Axenfeld–Rieger syndrome have a diverse presentation. There are many cardiac lesions associated with Axenfeld–Rieger syndrome. Although the most common are atrial septal defects and mitral valve defects, it is important to know others that could be associated with Axenfeld–Rieger syndrome as well, such as left-sided obstructive lesions. Therefore, cardiovascular screening should be pursued in patients diagnosed with Axenfeld–Rieger syndrome, and consideration of ophthalmology referral may be beneficial in patients with CHD and evidence of multi-system involvement when Axenfeld–Rieger syndrome is suspected.
Financial support
This research received no specific grant from any funding agency, commercial, or not-for-profit sectors.
Conflicts of interest
None.





